SCH4U TEXT BOOK
... your eye. “Certified organic” and “all natural” are stamped on the labels of some foods. Other labels claim that the foods are “chemical free.” As a chemistry student, you are aware that these labels may be misleading. Are all “chemicals” harmful in food, as some of the current advertising suggests? ...
... your eye. “Certified organic” and “all natural” are stamped on the labels of some foods. Other labels claim that the foods are “chemical free.” As a chemistry student, you are aware that these labels may be misleading. Are all “chemicals” harmful in food, as some of the current advertising suggests? ...
chapter 18 - HCC Learning Web
... Magnesium is an alkaline earth metal; Mg will oxidize to Mg2+. The oxidation state of hydrogen in HCl is +1. To be reduced, the oxidation state of H must decrease. The obvious choice for the hydrogen product is H2(g), where hydrogen has a zero oxidation state. The balanced reaction is Mg(s) + 2HCl(a ...
... Magnesium is an alkaline earth metal; Mg will oxidize to Mg2+. The oxidation state of hydrogen in HCl is +1. To be reduced, the oxidation state of H must decrease. The obvious choice for the hydrogen product is H2(g), where hydrogen has a zero oxidation state. The balanced reaction is Mg(s) + 2HCl(a ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... • so to describe solutions accurately, we must describe how much of each component is present – we saw that with pure substances, we can describe them with a single name because all samples identical Burns 4/e Chap 4 ...
... • so to describe solutions accurately, we must describe how much of each component is present – we saw that with pure substances, we can describe them with a single name because all samples identical Burns 4/e Chap 4 ...
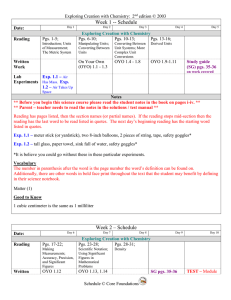
Week 1 -- Schedule
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
Equilibrium - pedagogics.ca
... The effect of a temperature change on a system at equilibrium can be now considered in terms of Le Chatelier’s principle. Going back to the first reaction, the value of ∆H here refers to the forward direction; so in this case the forward reaction is endothermic and the reverse reaction is exothermic ...
... The effect of a temperature change on a system at equilibrium can be now considered in terms of Le Chatelier’s principle. Going back to the first reaction, the value of ∆H here refers to the forward direction; so in this case the forward reaction is endothermic and the reverse reaction is exothermic ...
5 Steps to a 5 AP Chemistry, 2008-2009 Edition
... Welcome to the AP Chemistry Five-Step Program. The fact that you are reading this preface suggests that you will be taking the AP exam in chemistry. The AP Chemistry exam is constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exa ...
... Welcome to the AP Chemistry Five-Step Program. The fact that you are reading this preface suggests that you will be taking the AP exam in chemistry. The AP Chemistry exam is constantly evolving and so this guide has evolved. In this edition, we have updated the book to match the new AP Chemistry exa ...
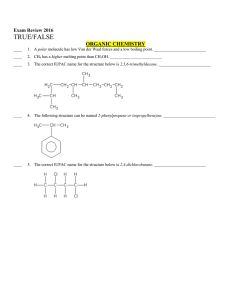
Chemistry Final Exam Review
... ____ 24. A reversible chemical reaction means that the reaction can travel forwards or backwards. ____ 25. Subscripts are used to balance chemical reactions. ____ 26. A synthesis reaction contains two products. ____ 27. A decomposition reaction contains at least two products. ____ 28. A combustion r ...
... ____ 24. A reversible chemical reaction means that the reaction can travel forwards or backwards. ____ 25. Subscripts are used to balance chemical reactions. ____ 26. A synthesis reaction contains two products. ____ 27. A decomposition reaction contains at least two products. ____ 28. A combustion r ...
Chem Course Desc2. New
... Pedagogical Philosophy: Chemistry is all around you--from the toothpaste you use in the morning, to the additives in your food, to the gasoline you put in your cars. All of these products share in common that a chemist has manipulated all their molecules to make your life more comfortable. Chemistry ...
... Pedagogical Philosophy: Chemistry is all around you--from the toothpaste you use in the morning, to the additives in your food, to the gasoline you put in your cars. All of these products share in common that a chemist has manipulated all their molecules to make your life more comfortable. Chemistry ...
Study Guide for Content Mastery - Student Edition
... upper part of the stratosphere. The ozone forms a layer around Earth, which absorbs ...
... upper part of the stratosphere. The ozone forms a layer around Earth, which absorbs ...
Stoichiometry Worksheet
... 3. Convert all amounts of products and/or reactants in the question into moles. Balance the following equation and use it to work the problems! NH3 + O2 → NO + H2O 4NH3 + 5O2 → 4NO + 6H2O a. How many grams of NO were needed to produce 30.2 g of water? NO = 30 amu 30g NO = 1 mole NO H2O = 18.02 amu 1 ...
... 3. Convert all amounts of products and/or reactants in the question into moles. Balance the following equation and use it to work the problems! NH3 + O2 → NO + H2O 4NH3 + 5O2 → 4NO + 6H2O a. How many grams of NO were needed to produce 30.2 g of water? NO = 30 amu 30g NO = 1 mole NO H2O = 18.02 amu 1 ...
Application of Novel Phosphine Ligands in Palladium
... catalysts have advantages compared to their homogeneous counterparts. However, being less selective, for many applications they are not suitable. The construction of complex molecules possessing various functional groups, for instance, requires mild reaction conditions, selective reagents and theref ...
... catalysts have advantages compared to their homogeneous counterparts. However, being less selective, for many applications they are not suitable. The construction of complex molecules possessing various functional groups, for instance, requires mild reaction conditions, selective reagents and theref ...
IIT-JEE (Advanced) - Brilliant Public School Sitamarhi
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
Experiments in General Chemistry: Featuring MeasureNet
... The second edition of this manual offers several new features: new digital pictures and illustrations, all concept/technique experiments converted to a guided inquiry format, the addition of three new self-directed experiments, and one new Capstone experiment. Virtually all the artwork and illustrat ...
... The second edition of this manual offers several new features: new digital pictures and illustrations, all concept/technique experiments converted to a guided inquiry format, the addition of three new self-directed experiments, and one new Capstone experiment. Virtually all the artwork and illustrat ...
PDF - Chemistry - University of Canterbury
... to their other studies such as biochemistry, biology, earth sciences, engineering, environmental science, forestry, geology, health sciences or physics. The Department of Chemistry offers a variety of courses to cater for all types of professional development and the CHEM 281 laboratory course is pa ...
... to their other studies such as biochemistry, biology, earth sciences, engineering, environmental science, forestry, geology, health sciences or physics. The Department of Chemistry offers a variety of courses to cater for all types of professional development and the CHEM 281 laboratory course is pa ...
Grade XII Unit 1 - Ethiopian Ministry of Education
... substance in a given solvent. What this expression means is that two substances with intermolecular forces of similar type and magnitude are likely to be soluble in each other. For example, in a solution formed between benzene (C6H6) and carbon tetrachloride (CCl4), both species are non-polar and ex ...
... substance in a given solvent. What this expression means is that two substances with intermolecular forces of similar type and magnitude are likely to be soluble in each other. For example, in a solution formed between benzene (C6H6) and carbon tetrachloride (CCl4), both species are non-polar and ex ...
Chapter 2 1.Certain gases in the 293K and 9.97 × 104Pa when the
... Why do some non-metallic elements (such as F, O, etc.) have become abnormal? Answer: In general, the electron affinity and energy with the decrease of atomic radius increases because the radius of hours, the nuclear charge of the electron cited large, therefore, electron affinity and energy left to ...
... Why do some non-metallic elements (such as F, O, etc.) have become abnormal? Answer: In general, the electron affinity and energy with the decrease of atomic radius increases because the radius of hours, the nuclear charge of the electron cited large, therefore, electron affinity and energy left to ...
evaluation copy
... 1. MODELING CHEMISTRY / STUDENT HANDOUT 18. Use the remaining solutions in Beakers A–D to repeat the reactions carried out above by combining the solutions in Beakers A and B and making measurements, and then combining the solutions in Beakers C and D and making measurements. Record observations of ...
... 1. MODELING CHEMISTRY / STUDENT HANDOUT 18. Use the remaining solutions in Beakers A–D to repeat the reactions carried out above by combining the solutions in Beakers A and B and making measurements, and then combining the solutions in Beakers C and D and making measurements. Record observations of ...
Chemistry - Bulletin < Brown
... The Chemistry concentration offers courses and research opportunities that range from fundamental studies involving the characterization and preparation of synthetic and naturally occurring molecules, to interdisciplinary studies at the interfaces of chemistry with biology, medicine, physics, engine ...
... The Chemistry concentration offers courses and research opportunities that range from fundamental studies involving the characterization and preparation of synthetic and naturally occurring molecules, to interdisciplinary studies at the interfaces of chemistry with biology, medicine, physics, engine ...
Multiple Choice Exam Review June 2016
... ____ 34. Catalysts can be used to speed up a reaction. _________________________ ____ 35. The value of the rate constant, k, is valid only for a specific reaction at a specific temperature. _________________________ ____ 36. An ineffective collision is one that has sufficient energy and correct orie ...
... ____ 34. Catalysts can be used to speed up a reaction. _________________________ ____ 35. The value of the rate constant, k, is valid only for a specific reaction at a specific temperature. _________________________ ____ 36. An ineffective collision is one that has sufficient energy and correct orie ...
KCl + O KClO 3 → However, this equation is not balanced, since
... In order to write a chemical equation, it is essential to know what substances are reacting and what substances are formed. It is only through experiment that a knowledge of what occurs can be discovered ─ either your experiment or the experiments of others (which can be found in textbooks). As more ...
... In order to write a chemical equation, it is essential to know what substances are reacting and what substances are formed. It is only through experiment that a knowledge of what occurs can be discovered ─ either your experiment or the experiments of others (which can be found in textbooks). As more ...