Chapter 10. Chemical Bonding II. Molecular Geometry and
... Valence Bond Theory ("simple" but somewhat limited) ...
... Valence Bond Theory ("simple" but somewhat limited) ...
Chapter 2 Outline
... Compounds and mixtures A. When two or more atoms bond covalently, they form a molecule B. A compound is formed when two or more different atoms bond chemically C. A mixture occurs when compounds can be separated by non-chemical means ...
... Compounds and mixtures A. When two or more atoms bond covalently, they form a molecule B. A compound is formed when two or more different atoms bond chemically C. A mixture occurs when compounds can be separated by non-chemical means ...
Nature of Atoms Atomic Structure
... amounts • Four elements make up 96.3% of human body weight – Carbon, hydrogen, oxygen, nitrogen ...
... amounts • Four elements make up 96.3% of human body weight – Carbon, hydrogen, oxygen, nitrogen ...
History of the Atom
... The History of the Atomic Model In other words, the old guys who came up with the atom. ...
... The History of the Atomic Model In other words, the old guys who came up with the atom. ...
Hydrogen Bonding
... Electronegativity – The capacity of an atom to attract electrons from a neighboring atom measured on a scale from 4 (fluorine, the most electronegative element) to a hypothetical 0 Covalent bonds between two atoms with comparable electronegativity correspond to an equal sharing of electrons betwee ...
... Electronegativity – The capacity of an atom to attract electrons from a neighboring atom measured on a scale from 4 (fluorine, the most electronegative element) to a hypothetical 0 Covalent bonds between two atoms with comparable electronegativity correspond to an equal sharing of electrons betwee ...
Democritus - davis.k12.ut.us
... • Formulated the first modern version of atomic theory: 1. All matter is made of indivisible units called atoms. 2. All atoms of the same element are identical. 3. Compounds (molecules) are formed when two or more atoms bond together. 4. Chemical reactions are the rearrangement of atomic bonds. • Co ...
... • Formulated the first modern version of atomic theory: 1. All matter is made of indivisible units called atoms. 2. All atoms of the same element are identical. 3. Compounds (molecules) are formed when two or more atoms bond together. 4. Chemical reactions are the rearrangement of atomic bonds. • Co ...
Molecular Geometry and Chemical Bonding Theory
... traditional unit, the debye (D, 1 D = 3.34 x 10–30 C·m), is often used. Depending on the spatial arrangement of the bonds, a molecule containing highly polar bonds can be nonpolar … the vector addition of the dipole moments yields a net dipole moment of zero for the overall molecule. Bond order is t ...
... traditional unit, the debye (D, 1 D = 3.34 x 10–30 C·m), is often used. Depending on the spatial arrangement of the bonds, a molecule containing highly polar bonds can be nonpolar … the vector addition of the dipole moments yields a net dipole moment of zero for the overall molecule. Bond order is t ...
Atomic combinations: Electronegativity and ionic
... 2.1 The nature of the ionic bond You will remember that when atoms bond, electrons are either shared or they are transferred between the atoms that are bonding. In covalent bonding, electrons are shared between the atoms. There is another type of bonding, where electrons are transferred from one ato ...
... 2.1 The nature of the ionic bond You will remember that when atoms bond, electrons are either shared or they are transferred between the atoms that are bonding. In covalent bonding, electrons are shared between the atoms. There is another type of bonding, where electrons are transferred from one ato ...
Grade 11 Chemistry E.. - hrsbstaff.ednet.ns.ca
... 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then balance the equation. For each reaction, tell what type of reaction it is. a) lead (II) ...
... 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then balance the equation. For each reaction, tell what type of reaction it is. a) lead (II) ...
Honors Chemistry
... 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happens when the excited electron returns to the grou ...
... 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happens when the excited electron returns to the grou ...
Ch. 3: “Atoms & the Periodic Table”
... 1. Summarize the main ideas of Dalton’s atomic theory. Elements are made of tiny, unique particles called atoms. Atoms cannot be divided. Atoms of the same element are identical. Atoms of different elements can join to form molecules. ...
... 1. Summarize the main ideas of Dalton’s atomic theory. Elements are made of tiny, unique particles called atoms. Atoms cannot be divided. Atoms of the same element are identical. Atoms of different elements can join to form molecules. ...
biol 1406 chapter 3: water
... Determine if the statement is true. If it is not, rewrite the italicized part to make it true. 1. An element is a substance that can be broken down into simpler substances. ______________________ 2. On Earth, 90 elements occur naturally. ________________________________________ 3. Only four elements ...
... Determine if the statement is true. If it is not, rewrite the italicized part to make it true. 1. An element is a substance that can be broken down into simpler substances. ______________________ 2. On Earth, 90 elements occur naturally. ________________________________________ 3. Only four elements ...
Chapter 6.2 Notes
... Metals form metallic bonds – bonds between metal cations and the sea of electrons around them - the nuclei form a closest packing structure - the electrons flow around them and do not belong to any one atom - there is a sea of freely moving electrons - this allows metals to flex into sheets or wires ...
... Metals form metallic bonds – bonds between metal cations and the sea of electrons around them - the nuclei form a closest packing structure - the electrons flow around them and do not belong to any one atom - there is a sea of freely moving electrons - this allows metals to flex into sheets or wires ...
Chapter 2—Atoms, Molecules, and Ions
... The table in one sentence • The periodic table is an arrangement of elements in horizontal order of increasing atomic number and vertical order of chemical similarity • The modern periodic table got its start with Dmitri Mendeleev, who first began grouping elements in order of similar chemical prop ...
... The table in one sentence • The periodic table is an arrangement of elements in horizontal order of increasing atomic number and vertical order of chemical similarity • The modern periodic table got its start with Dmitri Mendeleev, who first began grouping elements in order of similar chemical prop ...
The Chemistry of Life
... 2 electrons 1st energy level, 4 electrons on outer energy level 6 protons, 6 neutrons Bonds well with other carbon atoms, as well as with other elements. • Single, double and triple bonds ...
... 2 electrons 1st energy level, 4 electrons on outer energy level 6 protons, 6 neutrons Bonds well with other carbon atoms, as well as with other elements. • Single, double and triple bonds ...
atomic number
... If there are vacancies, the atom is reactive/unstable If there are no vacancies, the atom is nonreactive/stable If the vacancies are filled due to bonding, the molecule is stable ...
... If there are vacancies, the atom is reactive/unstable If there are no vacancies, the atom is nonreactive/stable If the vacancies are filled due to bonding, the molecule is stable ...
download
... Since electrons have a negative charge, the atom that gains electrons becomes a negatively charged ions (aka anion) because it now has more electrons than protons. Alternately, an atom that looses electrons becomes a positively charged ion (aka cations). The particles in an ionic compound are held t ...
... Since electrons have a negative charge, the atom that gains electrons becomes a negatively charged ions (aka anion) because it now has more electrons than protons. Alternately, an atom that looses electrons becomes a positively charged ion (aka cations). The particles in an ionic compound are held t ...
Chemistry 201/211 - Department of Chemistry | Oregon State
... d.) can have a maximum of 3 unpaired electrons for an atom in the ground state yes, there can be six electrons total in a p sublevel, if there are only three, they will be arranged so that their spin is maximized () 3.) The first two quantum numbers (n,l) of the highest energy electron of scandiu ...
... d.) can have a maximum of 3 unpaired electrons for an atom in the ground state yes, there can be six electrons total in a p sublevel, if there are only three, they will be arranged so that their spin is maximized () 3.) The first two quantum numbers (n,l) of the highest energy electron of scandiu ...
Name: Date: Period: _____ Unit 2 Notes, Part 1 – The Basics of
... 2. Atoms are the smallest unit of matter. Each different type of atom represents an element (ex: hydrogen, oxygen, carbon). Scientists have created a chart called the periodic table of elements to organize elements by their atomic properties. 3. Four elements—carbon (C), oxygen (O), hydrogen (H), an ...
... 2. Atoms are the smallest unit of matter. Each different type of atom represents an element (ex: hydrogen, oxygen, carbon). Scientists have created a chart called the periodic table of elements to organize elements by their atomic properties. 3. Four elements—carbon (C), oxygen (O), hydrogen (H), an ...
document
... share to form compounds. 5. Ionic Bond I E. States that all elements want either a full outer shell or eight 6. Subscript H electrons in their outer electron shell. 7. Polyatomic Ion J F. A multiplier. It is used to balance equations. 8. Synthesis Reaction L G. A reaction in which two reactant compo ...
... share to form compounds. 5. Ionic Bond I E. States that all elements want either a full outer shell or eight 6. Subscript H electrons in their outer electron shell. 7. Polyatomic Ion J F. A multiplier. It is used to balance equations. 8. Synthesis Reaction L G. A reaction in which two reactant compo ...
Medical Physics and Statistics
... Forty three (43) signed immediately. Six (6) asked for time to think. ...
... Forty three (43) signed immediately. Six (6) asked for time to think. ...
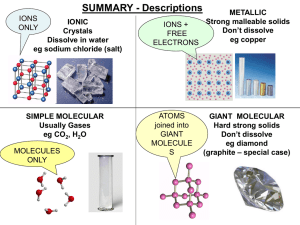
smart_materials_1 - Aldercar High School
... HIGH mpt/bpt. Hard & strong Strong bonds between IONS Malleable Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...
... HIGH mpt/bpt. Hard & strong Strong bonds between IONS Malleable Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions ...