Chapter 4 – Reactions in Aqueous Solutions
... • Example-1: Balance the following oxidation-reduction reaction in aqueous solution: ...
... • Example-1: Balance the following oxidation-reduction reaction in aqueous solution: ...
Chemistry Notes for the Whole Year Powerpoint
... • Electron configurations contain the energy level, orbital, and number of electrons in that orbital. For example, 1s1, the first number means energy level one, s is the orbital, and the superscript number means that there is one electron in this orbital. • Start at hydrogen and fill electrons from ...
... • Electron configurations contain the energy level, orbital, and number of electrons in that orbital. For example, 1s1, the first number means energy level one, s is the orbital, and the superscript number means that there is one electron in this orbital. • Start at hydrogen and fill electrons from ...
AP® Chemistry
... A 10-minute break should be provided after Section I is completed. Students should be given a 10-minute warning prior to the end of each of the Parts A and B in Section II of the exam. The actual AP Exam is administered in one session. Students will have the most realistic experience if a complete m ...
... A 10-minute break should be provided after Section I is completed. Students should be given a 10-minute warning prior to the end of each of the Parts A and B in Section II of the exam. The actual AP Exam is administered in one session. Students will have the most realistic experience if a complete m ...
EQUILIBRIUM
... 1 point for calculation of Q with correct mass action expression consistent with part a ii) Since Q > Kp, the numerator must decrease and the denominator must increase, so the reaction must proceed from right to left to establish equilibrium 1 point for direction or for stating that Q > Kp 1 point f ...
... 1 point for calculation of Q with correct mass action expression consistent with part a ii) Since Q > Kp, the numerator must decrease and the denominator must increase, so the reaction must proceed from right to left to establish equilibrium 1 point for direction or for stating that Q > Kp 1 point f ...
A flask contains 0
... o they are generally lined up by decimal points, even though this does not make the number list “straight” allows you to see decimal points and significant figures easier. Look for the word approximate in the question, if there, you can use estimation to help arrive at the answer. Strategies on ...
... o they are generally lined up by decimal points, even though this does not make the number list “straight” allows you to see decimal points and significant figures easier. Look for the word approximate in the question, if there, you can use estimation to help arrive at the answer. Strategies on ...
Section 3_Energetics
... Direct determination of lattice energy is very difficult because it is very difficult to get isolated sodium and chloride ions. Therefore the values are usually calculated from other experimentally determined data by applying the Hess Law. The Born-Haber Cycle is a technique of applying Hess‘s Law t ...
... Direct determination of lattice energy is very difficult because it is very difficult to get isolated sodium and chloride ions. Therefore the values are usually calculated from other experimentally determined data by applying the Hess Law. The Born-Haber Cycle is a technique of applying Hess‘s Law t ...
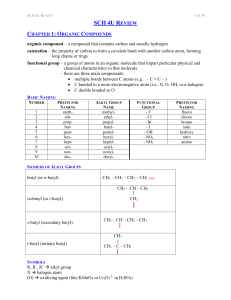
12 U Chem Review
... toluene / phenyl methane methyl benzene acetate ethanoate acetamide ethanamide ...
... toluene / phenyl methane methyl benzene acetate ethanoate acetamide ethanamide ...
SQA Advanced Higher Chemistry Unit 2 Principles of Chemical
... a) statement 1 only b) statements 1 and 2 c) statements 1, 2 and 3. Q14: When real quantities need to be weighed out in the laboratory or industry, which statement is most helpful? a) statement 1 b) statement 2 c) statement 3 ...
... a) statement 1 only b) statements 1 and 2 c) statements 1, 2 and 3. Q14: When real quantities need to be weighed out in the laboratory or industry, which statement is most helpful? a) statement 1 b) statement 2 c) statement 3 ...
PRE-LABORATORY ASSIGNMENT EXPERIMENT 6 1. Is the sign of
... Figure 1 shows a set of data taken for the determination of a heat of solution. The time of mixing was at 190 seconds and data were recorded for 330 seconds. In order to determine the temperature change, ∆T, which would have occurred if the mixing was instantaneous, it is assumed that the loss of he ...
... Figure 1 shows a set of data taken for the determination of a heat of solution. The time of mixing was at 190 seconds and data were recorded for 330 seconds. In order to determine the temperature change, ∆T, which would have occurred if the mixing was instantaneous, it is assumed that the loss of he ...
Multiple Choice Math Practice File
... o they are generally lined up by decimal points, even though this does not make the number list “straight” Allows you to see decimal points and significant figures easier. Look for the word approximate in the question, if there, you can use estimation to help arrive at the answer. Strategies on ...
... o they are generally lined up by decimal points, even though this does not make the number list “straight” Allows you to see decimal points and significant figures easier. Look for the word approximate in the question, if there, you can use estimation to help arrive at the answer. Strategies on ...
Chapter 4: Reactions in Aqueous Solution
... 1) Water is a very common solvent due to its wide availability and low cost (most of our world is water). 2) Many reactions take place in aqueous solution. The term aqueous means dissolved in water. 3) Hydration of solids in Water A) Solid dissolves (falls apart) through interaction of ions with wat ...
... 1) Water is a very common solvent due to its wide availability and low cost (most of our world is water). 2) Many reactions take place in aqueous solution. The term aqueous means dissolved in water. 3) Hydration of solids in Water A) Solid dissolves (falls apart) through interaction of ions with wat ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.