Chemical Equations and Reactions notes File
... MnO2 or Pt: catalyst 25°C :specific Temp requirement 2 atm: specific Pressure ...
... MnO2 or Pt: catalyst 25°C :specific Temp requirement 2 atm: specific Pressure ...
Ivan Lomachenkov
... The electromagnetic rotation of water The rotation of water in magnetic and electrical fields demonstrates the effect of a magnetic force on charged particles. It’s a simple equipment. ...
... The electromagnetic rotation of water The rotation of water in magnetic and electrical fields demonstrates the effect of a magnetic force on charged particles. It’s a simple equipment. ...
Ivan Lomachenkov
... the ions of Na. The result is vr~ I/n, I- the current, n- the concentration of the ions. For the current I~ 0.1 A we have vr~ 10-7m/s. • We can also estimate the circular component of the velocity: v~ nvrB/, where - the viscosity of the solution. For inductance B~10-2 T and ~10-3kg/(m·s) the re ...
... the ions of Na. The result is vr~ I/n, I- the current, n- the concentration of the ions. For the current I~ 0.1 A we have vr~ 10-7m/s. • We can also estimate the circular component of the velocity: v~ nvrB/, where - the viscosity of the solution. For inductance B~10-2 T and ~10-3kg/(m·s) the re ...
sch3u unit 1 test: matter
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...
Chapter 20 - public.asu.edu
... Consider the Zn/Cu2+ reaction if more Cu2+ is added to the cell. The voltage becomes greater than 1.10 V. What is E of the Zn/Cu2+ reaction if [Cu2+] = 0.010 M and [Zn2+] =1.99 M? Note that this corresponds to starting with standard conditions and changing to 99% completion of reaction. Eo = 1.10 V ...
... Consider the Zn/Cu2+ reaction if more Cu2+ is added to the cell. The voltage becomes greater than 1.10 V. What is E of the Zn/Cu2+ reaction if [Cu2+] = 0.010 M and [Zn2+] =1.99 M? Note that this corresponds to starting with standard conditions and changing to 99% completion of reaction. Eo = 1.10 V ...
Unit 5 Study Guide
... Unit 5 Study Guide: Chemical Reactions 1. What are the 7 diatomic molecules? ...
... Unit 5 Study Guide: Chemical Reactions 1. What are the 7 diatomic molecules? ...
Exam practice answers 5
... (d) There are two equally viable ways of doing this: using a Hess’s triangle or a Born-Haber type cycle. Hess’s triangle method: ...
... (d) There are two equally viable ways of doing this: using a Hess’s triangle or a Born-Haber type cycle. Hess’s triangle method: ...
CVB101 – Lecture 3 Chemical Bonding • Chemical bonding
... Reduction and Oxidation (redox) reactions Definition of redox reaction – a reaction in which electrons are transferred Half reactions are used to show electron transfer. Oxidation – loss of electrons Reduction – gain of electrons Common redox reactions Combination reactions – a reaction in ...
... Reduction and Oxidation (redox) reactions Definition of redox reaction – a reaction in which electrons are transferred Half reactions are used to show electron transfer. Oxidation – loss of electrons Reduction – gain of electrons Common redox reactions Combination reactions – a reaction in ...
Cl -1
... 2. The oxidation number of a monatomic ion equals the charge on the ion. 3. The more-electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in a compound is always -1. 5. Oxygen has an oxidation num ...
... 2. The oxidation number of a monatomic ion equals the charge on the ion. 3. The more-electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in a compound is always -1. 5. Oxygen has an oxidation num ...
lecture 13
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
ch5_f08
... keep track of electron transfers : 1) The oxidation number of any free element is zero. 2) The oxidation number of any simple, monoatomic ion is equal to the charge on the ion. 3) The sum of all oxidation numbers of the atoms in a molecule or polyatomic ion must equal the charge on the particle. 4) ...
... keep track of electron transfers : 1) The oxidation number of any free element is zero. 2) The oxidation number of any simple, monoatomic ion is equal to the charge on the ion. 3) The sum of all oxidation numbers of the atoms in a molecule or polyatomic ion must equal the charge on the particle. 4) ...
1. A [1] 2. B [1] 3. Dilute sodium chloride: 2H 2 O → O 2 + 4H + + 4e
... sodium is a very powerful reducing agent/high in electrochemical series; any chemical reducing agent would need to be even higher in ...
... sodium is a very powerful reducing agent/high in electrochemical series; any chemical reducing agent would need to be even higher in ...
2013 Final Exam Answers
... No energy change is associated with the reaction. Not enough information is given to determine the energy change. None of these. ...
... No energy change is associated with the reaction. Not enough information is given to determine the energy change. None of these. ...
S8 + ___ F2 → ___ SF6 - Canvas by Instructure
... __ H+ + __ Cr2O72- + __C2H5OH __ Cr3+ + __ H2O + __ CO2 (Note: this is a net ionic equation – number of atoms & CHARGE must be balanced.) ...
... __ H+ + __ Cr2O72- + __C2H5OH __ Cr3+ + __ H2O + __ CO2 (Note: this is a net ionic equation – number of atoms & CHARGE must be balanced.) ...
Practical, Asymmetric Redox-Neutral Chemical Synthesis via Borrowing Hydrogen
... “redox economy” which focuses on minimizing synthetic steps that only adjust the oxidation state of the intermediates without generating structural complexity is an important consideration at the strategic level for chemical synthesis, and redox-neutral transformations that circumvent such redundant ...
... “redox economy” which focuses on minimizing synthetic steps that only adjust the oxidation state of the intermediates without generating structural complexity is an important consideration at the strategic level for chemical synthesis, and redox-neutral transformations that circumvent such redundant ...
Gas-Forming reactions Reactions that form a
... But they end up with the same number of electrons they start with. Every atom, ion or polyatomic ion has a formal oxidation number associated with it. This value compares the number of protons in an atom (positive charge) and the number of electrons assigned to that atom (negative charge). In many c ...
... But they end up with the same number of electrons they start with. Every atom, ion or polyatomic ion has a formal oxidation number associated with it. This value compares the number of protons in an atom (positive charge) and the number of electrons assigned to that atom (negative charge). In many c ...
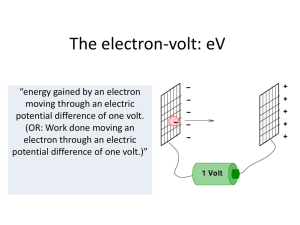
The electron-volt - Hockerill Students
... moving through an electric potential difference of one volt. (OR: Work done moving an electron through an electric potential difference of one volt.)” ...
... moving through an electric potential difference of one volt. (OR: Work done moving an electron through an electric potential difference of one volt.)” ...
Chapter 4 - Aqueous Reactions
... Some observations on the series Lead (Pb) is above H, so is Al. But these metals are not attacked by 6M HCl. They form very protective oxides. Cu reacts with nitric acid (HNO3) because that acid is a strong oxidizing agent in addition to being an acid. Gold (Au) and platinum (Pt) are valuable b ...
... Some observations on the series Lead (Pb) is above H, so is Al. But these metals are not attacked by 6M HCl. They form very protective oxides. Cu reacts with nitric acid (HNO3) because that acid is a strong oxidizing agent in addition to being an acid. Gold (Au) and platinum (Pt) are valuable b ...
Chapter 13 Notes
... In this reaction, carbon is losing electrons so it is undergoing oxidation. Iron is gaining back electrons it had lost to become a free element so it is undergoing reduction. This is called an oxidation-reduction reaction or redox for short. Although this type of reaction is named for oxygen, many o ...
... In this reaction, carbon is losing electrons so it is undergoing oxidation. Iron is gaining back electrons it had lost to become a free element so it is undergoing reduction. This is called an oxidation-reduction reaction or redox for short. Although this type of reaction is named for oxygen, many o ...
The Cathode Ray Tube (CRT)
... Between anode and cathode, potential energy is converted to kinetic energy so that q∆V = ...
... Between anode and cathode, potential energy is converted to kinetic energy so that q∆V = ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
OXIDATION NUMBERS
... surface of the metal and the liquid; this is called the electrode potential. The further to the left the above equilibrium lies, the greater the potential difference will be. For a given metal, the position of the equilibrium depends on the concentration of metal ions already present in the solution ...
... surface of the metal and the liquid; this is called the electrode potential. The further to the left the above equilibrium lies, the greater the potential difference will be. For a given metal, the position of the equilibrium depends on the concentration of metal ions already present in the solution ...
Document
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.











![1. A [1] 2. B [1] 3. Dilute sodium chloride: 2H 2 O → O 2 + 4H + + 4e](http://s1.studyres.com/store/data/011637084_1-98d57769d10f8d697c9e5678e340d457-300x300.png)