PowerPoint for Cornell Notes
... and salt. Have you ever been unlucky enough to be stung by a wasp or a bee? Bee stings are acidic in nature, which is why a household remedy for a bee sting is baking soda or sodium bicarbonate, which is a basic substance. A wasp sting, on the other hand, is mildly basic, so a household remedy for t ...
... and salt. Have you ever been unlucky enough to be stung by a wasp or a bee? Bee stings are acidic in nature, which is why a household remedy for a bee sting is baking soda or sodium bicarbonate, which is a basic substance. A wasp sting, on the other hand, is mildly basic, so a household remedy for t ...
South Pasadena • AP Chemistry
... The chemical elements are the building blocks of matter, which can be understood in terms of arrangements of atoms. Molecules & elements Chemical analysis The mole Electron configuration Periodicity Quantum mechanical model Atomic models Mass spectrometry Light & matter Conservat ...
... The chemical elements are the building blocks of matter, which can be understood in terms of arrangements of atoms. Molecules & elements Chemical analysis The mole Electron configuration Periodicity Quantum mechanical model Atomic models Mass spectrometry Light & matter Conservat ...
Oxidation-reduction reactions and electrochemistry
... Oxidation-reduction, precipitation, formation of weak electrolytes (especially water), Ch. 4 [Ch.4] seen as competition processes (little attention given to this notion in textbook). Qualitative notion of “driving forces” of competing reactions - eg, in the case of pHdependent solubilities of salts ...
... Oxidation-reduction, precipitation, formation of weak electrolytes (especially water), Ch. 4 [Ch.4] seen as competition processes (little attention given to this notion in textbook). Qualitative notion of “driving forces” of competing reactions - eg, in the case of pHdependent solubilities of salts ...
Unit 2: Chemical Reactions
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
Document
... (d) If you break a piece of glassware, you should pick up all the pieces with your hands. ...
... (d) If you break a piece of glassware, you should pick up all the pieces with your hands. ...
Deconstructed HS-PS1-2
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...
Electron Discovery (PowerPoint)
... At the end of the 19th century it was known that when a very high voltage was applied between the cathode (negative electrode) and the anode (positive electrode), the end of the gas filled tube would glow. A hole in one or more screens restricts the glow to a tiny spot. It seemed as if something bei ...
... At the end of the 19th century it was known that when a very high voltage was applied between the cathode (negative electrode) and the anode (positive electrode), the end of the gas filled tube would glow. A hole in one or more screens restricts the glow to a tiny spot. It seemed as if something bei ...
S3 Chemistry - eduBuzz.org
... Identify whether a species has an equal or unequal number of protons and electrons and use this to state whether it is an atom or ion. State the charge of an ion. Calculate the charge on a ion using nuclide notation Use the periodic table to identify whether an element is a metal or non-meta ...
... Identify whether a species has an equal or unequal number of protons and electrons and use this to state whether it is an atom or ion. State the charge of an ion. Calculate the charge on a ion using nuclide notation Use the periodic table to identify whether an element is a metal or non-meta ...
Learning Outcomes for Chemical Reactions and
... • Identify whether a species has an equal or unequal number of protons and electrons and use this to state whether it is an atom or ion. • State the charge of an ion. • Calculate the charge on a ion using nuclide notation • Use the periodic table to identify whether an element is a metal or non-meta ...
... • Identify whether a species has an equal or unequal number of protons and electrons and use this to state whether it is an atom or ion. • State the charge of an ion. • Calculate the charge on a ion using nuclide notation • Use the periodic table to identify whether an element is a metal or non-meta ...
Chemical Kinetics - Review
... S2O82-(aq) + 2 I–(aq) → 2 SO42-(aq) + I2 (s) Ba2+(aq) + SO42-(aq) → BaSO4 (s) ...
... S2O82-(aq) + 2 I–(aq) → 2 SO42-(aq) + I2 (s) Ba2+(aq) + SO42-(aq) → BaSO4 (s) ...
Topic 9 - uaschemistry
... because they involve a more reactive metal or non-metal displacing the reactive one from its salt. ...
... because they involve a more reactive metal or non-metal displacing the reactive one from its salt. ...
Pre- AP & NET IONIC EQUATIONS
... single, double, synthesis, decomposition, combustion of hydrocarbons Anhydrides taught with synthesis and decomposition reactions Calculation of oxidation numbers Oxidation, reduction, oxidizing agents, and reducing agents. Net ionic equations for single and double replacement (taught with s ...
... single, double, synthesis, decomposition, combustion of hydrocarbons Anhydrides taught with synthesis and decomposition reactions Calculation of oxidation numbers Oxidation, reduction, oxidizing agents, and reducing agents. Net ionic equations for single and double replacement (taught with s ...
NOTES: 2.1 - Intro to Chemistry
... *IONIC BOND: bond formed by the attraction of a positive ion to a negative ion -anion: negatively charged ion; has gained 1 or more electrons -cation: positively charged ion; has lost 1 or more electrons ...
... *IONIC BOND: bond formed by the attraction of a positive ion to a negative ion -anion: negatively charged ion; has gained 1 or more electrons -cation: positively charged ion; has lost 1 or more electrons ...
AS Unit F321 Unit 1 Side A check list
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
Activity Series Unit
... the previous problem? The oxidation numbers decrease. This term is known as reduction. 24. Let’s called this trend reduction. Use the following equation as another example to consider this term. What happens to chlorine in reaction four? Is there a general trend that can be stated for any species th ...
... the previous problem? The oxidation numbers decrease. This term is known as reduction. 24. Let’s called this trend reduction. Use the following equation as another example to consider this term. What happens to chlorine in reaction four? Is there a general trend that can be stated for any species th ...
students - Teach-n-Learn-Chem
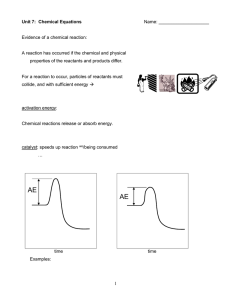
... A reaction has occurred if the chemical and physical properties of the reactants and products differ. ...
... A reaction has occurred if the chemical and physical properties of the reactants and products differ. ...
Chemical Equations
... the arrow) and the products (on the right of the arrow). C. The law of conservation of mass and energy must be satisfied. Therefore the same number of atoms of each element must appear on each side of a correct chemical equation. ...
... the arrow) and the products (on the right of the arrow). C. The law of conservation of mass and energy must be satisfied. Therefore the same number of atoms of each element must appear on each side of a correct chemical equation. ...
chemistry important question i
... (a) How many moles of mercury will be produced by electrolysing 1.0 M. Hg(NO3)2 solution with a current of 2.00 A for 3 hours? (b) A voltaic cell is set up at 25° C with the following half-cells Al3+ (0.001 M) and Ni2+ (0.50 M). Write an equation for the reaction that occurs when the cell generates ...
... (a) How many moles of mercury will be produced by electrolysing 1.0 M. Hg(NO3)2 solution with a current of 2.00 A for 3 hours? (b) A voltaic cell is set up at 25° C with the following half-cells Al3+ (0.001 M) and Ni2+ (0.50 M). Write an equation for the reaction that occurs when the cell generates ...
acids and bases - No Brain Too Small
... Have same atomic # but different mass # Have a slightly different mass to each other because of different numbers of neutrons in the ...
... Have same atomic # but different mass # Have a slightly different mass to each other because of different numbers of neutrons in the ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.