Document
... • Always choose the longest chain that contains both atoms of the double bond. • In cycloalkenes, the double bond is located between C1 and C2, and the “1” is usually omitted in the name. • The ring is numbered clockwise or counterclockwise to give the first substituent the lower number. • Compounds ...
... • Always choose the longest chain that contains both atoms of the double bond. • In cycloalkenes, the double bond is located between C1 and C2, and the “1” is usually omitted in the name. • The ring is numbered clockwise or counterclockwise to give the first substituent the lower number. • Compounds ...
2nd Semester Practice Chemistry Final 2009
... heat of gold is 0.13 J/g·ºC. a. 26 J c. 0.0006 J b. 26 J/g·ºC d. 0.0006 J/g·ºC 57. A system that changes spontaneously without an enthalpy change a. is impossible. c. becomes more disordered. b. becomes more ordered. d. releases heat. 58. What is the symbol for entropy? a. T c. G b. H d. S 59. What ...
... heat of gold is 0.13 J/g·ºC. a. 26 J c. 0.0006 J b. 26 J/g·ºC d. 0.0006 J/g·ºC 57. A system that changes spontaneously without an enthalpy change a. is impossible. c. becomes more disordered. b. becomes more ordered. d. releases heat. 58. What is the symbol for entropy? a. T c. G b. H d. S 59. What ...
Postprint
... Discovered in 1804 by Smithson Tennant, iridium is amongst the least abundant elements in the Earth’s crust.3 It knew little use until the synthesis by Vaska of his eponymous complex: [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum ...
... Discovered in 1804 by Smithson Tennant, iridium is amongst the least abundant elements in the Earth’s crust.3 It knew little use until the synthesis by Vaska of his eponymous complex: [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum ...
Novel Methods and Materials in Development of Liquid Carrier
... within the frame of PhD studies in the industry. Therefore, I was looking for an advisor, who would take over the supervision of my work. I led quite long e-mail correspondence with Prof. Melin whom I knew from Bayer AG, where I did my first industrial working experience in summer 1996. Meanwhile, I ...
... within the frame of PhD studies in the industry. Therefore, I was looking for an advisor, who would take over the supervision of my work. I led quite long e-mail correspondence with Prof. Melin whom I knew from Bayer AG, where I did my first industrial working experience in summer 1996. Meanwhile, I ...
Edita Pusvaškienė
... toluene, ethylbenzene and o-xylene. As it was shown above, the coating can be operated without any damage up to the 280oC temperature so, for the further experiments this desorption temperature was selected. Desorption time from 10 to 180 s was investigated. A standard solution containing 10 mg L-1 ...
... toluene, ethylbenzene and o-xylene. As it was shown above, the coating can be operated without any damage up to the 280oC temperature so, for the further experiments this desorption temperature was selected. Desorption time from 10 to 180 s was investigated. A standard solution containing 10 mg L-1 ...
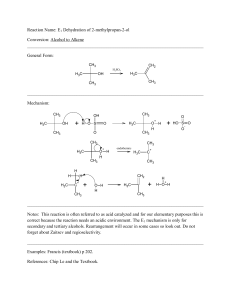
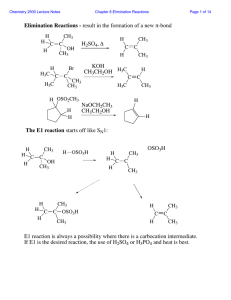
Reactions
... OH H2O H2SO4 The regiochemistry is determined by the relative stability of the intermediate carbocation. But what if you want 1-propyl alcohol? What could you do to “trick” the regiochemistry? ...
... OH H2O H2SO4 The regiochemistry is determined by the relative stability of the intermediate carbocation. But what if you want 1-propyl alcohol? What could you do to “trick” the regiochemistry? ...
Chm 2
... For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many moles of iron(II ...
... For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many moles of iron(II ...
Grignard Reagents brochure
... H.M.Walborsky, Chemie in unserer Zeit 1991, 25, 108-116. a) H.M.Walborsky, Acc.Chem.Res. 1990, 23,286; H. M. Walborsky, C. Zimmermann, J. Am. Chem. Soc. 1992, 114, 4996; b) J.F.Garst, Acc.Chem.Res,1991,24,95, c), C.Walling, Acc.Chem.Res. 1991 24, 255. Ultrasound: C. Horst, U. Hoffmann, U. Kunz, Chem ...
... H.M.Walborsky, Chemie in unserer Zeit 1991, 25, 108-116. a) H.M.Walborsky, Acc.Chem.Res. 1990, 23,286; H. M. Walborsky, C. Zimmermann, J. Am. Chem. Soc. 1992, 114, 4996; b) J.F.Garst, Acc.Chem.Res,1991,24,95, c), C.Walling, Acc.Chem.Res. 1991 24, 255. Ultrasound: C. Horst, U. Hoffmann, U. Kunz, Chem ...
Sam P. de Visser,* Jan-Uwe Rohde,* Yong
... The catalytic cycle of one representative enzyme, taurine:˛ketoglutarate dioxygenase (TauD) [7,28–34], is shown in Fig. 2 and starts from a resting state where the three remaining Fe coordination sites are occupied by water molecules, and upon co-substrate binding, namely ␣KG, two water molecules ar ...
... The catalytic cycle of one representative enzyme, taurine:˛ketoglutarate dioxygenase (TauD) [7,28–34], is shown in Fig. 2 and starts from a resting state where the three remaining Fe coordination sites are occupied by water molecules, and upon co-substrate binding, namely ␣KG, two water molecules ar ...