Kinetics of Particles: Newton`s Second Law
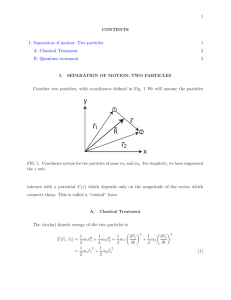
... This is a diagram showing some object and the forces applied to it. It contains only forces and coordinate information, nothing else. There are only two kinds of forces to be considered in mechanics: ...
... This is a diagram showing some object and the forces applied to it. It contains only forces and coordinate information, nothing else. There are only two kinds of forces to be considered in mechanics: ...
Contemporary Quantum Optics
... Consequences of the semiclassical theory • Photoelectric, Compton effects can be understood with a classical wave • Pulses recorded in the photomultiplier are due to quantum jumps inside the material and not to the granular structure of light same for the photographic plate in Taylor ’s experiment ...
... Consequences of the semiclassical theory • Photoelectric, Compton effects can be understood with a classical wave • Pulses recorded in the photomultiplier are due to quantum jumps inside the material and not to the granular structure of light same for the photographic plate in Taylor ’s experiment ...
chapter 7: atomic structure and periodicity
... Hund’s Rule – orbitals of ___________ energy (degenerate orbitals) are each occupied by a ____________ Electron (with parallel spins) before any one orbital is occupies by a ______________ electron. Paramagnetic – contains one or more _____________ electrons – attraction towards a magnetic field. Di ...
... Hund’s Rule – orbitals of ___________ energy (degenerate orbitals) are each occupied by a ____________ Electron (with parallel spins) before any one orbital is occupies by a ______________ electron. Paramagnetic – contains one or more _____________ electrons – attraction towards a magnetic field. Di ...
An introduction to Quantum Optics
... 1) Problem of interpretation 2) Problem of formalism : many diverging quantities e.g. Vacuum energy : 3) Problem of "concurrence" : the more simple semiclassical theory gives (generally) the same results • 2) was solved in 1947 (Feynman, Schwinger & Tomonaga) : ...
... 1) Problem of interpretation 2) Problem of formalism : many diverging quantities e.g. Vacuum energy : 3) Problem of "concurrence" : the more simple semiclassical theory gives (generally) the same results • 2) was solved in 1947 (Feynman, Schwinger & Tomonaga) : ...
WAVE MECHANICS (Schrödinger, 1926)
... WAVE MECHANICS * The energy depends only on the principal quantum number, as in the Bohr model: En = -2.179 X 10-18J /n2 * The orbitals are named by giving the n value followed by a letter symbol for l: l= 0,1, 2, 3, 4, 5, ... s p d f g h ... * All orbitals with the same n are called a “shell”. All ...
... WAVE MECHANICS * The energy depends only on the principal quantum number, as in the Bohr model: En = -2.179 X 10-18J /n2 * The orbitals are named by giving the n value followed by a letter symbol for l: l= 0,1, 2, 3, 4, 5, ... s p d f g h ... * All orbitals with the same n are called a “shell”. All ...
H CH 4 Homework
... 6. Distinguish between the ground state and the excited state. Ground state is the initial state of the electron. Excited state is after it absorbs energy 7. According to Bohr, how is a line spectrum produced. Electron falls from the excited state and emits a photon 8. What is the major difference b ...
... 6. Distinguish between the ground state and the excited state. Ground state is the initial state of the electron. Excited state is after it absorbs energy 7. According to Bohr, how is a line spectrum produced. Electron falls from the excited state and emits a photon 8. What is the major difference b ...
Chemistry Name______________________________________
... While there are many different orbits they can only occupy one at a time. They will gain energy to jump to higher orbit and lose energy to fall to lower lowest energy for atom (all electrons in orbits closest to nucleus) couldnot explain other atom’s spectra couldnot account for chemical behavior of ...
... While there are many different orbits they can only occupy one at a time. They will gain energy to jump to higher orbit and lose energy to fall to lower lowest energy for atom (all electrons in orbits closest to nucleus) couldnot explain other atom’s spectra couldnot account for chemical behavior of ...
Chapter 1 Atoms Properties of Matter Intensive vs. Extensive
... Chapter 1 Atoms Properties of Matter o Intensive vs. Extensive, physical vs. chemical Chemical Change Physical Change Mixtures and Pure Substances Elements and Compounds o Group or Family o Period or Row o Metals o Nonmetals o Metalloids Chapter 2 Scientific Method SI Units of Measur ...
... Chapter 1 Atoms Properties of Matter o Intensive vs. Extensive, physical vs. chemical Chemical Change Physical Change Mixtures and Pure Substances Elements and Compounds o Group or Family o Period or Row o Metals o Nonmetals o Metalloids Chapter 2 Scientific Method SI Units of Measur ...
Physics PHYS 352 Mechanics II Problem Set #4
... vertical plane and rotates about a vertical diameter with constant angular velocity . ...
... vertical plane and rotates about a vertical diameter with constant angular velocity . ...
Chapter 9 The Atom - Bakersfield College
... discovery of the photoelectric effect could not be explained by the electromagnetic theory of light. Albert Einstein developed the quantum theory of light in ...
... discovery of the photoelectric effect could not be explained by the electromagnetic theory of light. Albert Einstein developed the quantum theory of light in ...
Waves QM and SCh eq
... within a material medium, such as air, water, and rock. 2. Electromagnetic waves. Examples include visible and ultraviolet light, radio and television waves, microwaves, x rays, and radar waves. These waves require no material medium to exist. Light waves from stars, for example, travel through the ...
... within a material medium, such as air, water, and rock. 2. Electromagnetic waves. Examples include visible and ultraviolet light, radio and television waves, microwaves, x rays, and radar waves. These waves require no material medium to exist. Light waves from stars, for example, travel through the ...
Tunneling Effect and Its Applications Quantum
... nucleus because of the high energy requirement to escape the very strong potential. In quantum mechanics, however, there is a probability the particle can tunnel through the potential and escape. Then the half-life of the particle becomes finite and the energy of the emission is broadened. ...
... nucleus because of the high energy requirement to escape the very strong potential. In quantum mechanics, however, there is a probability the particle can tunnel through the potential and escape. Then the half-life of the particle becomes finite and the energy of the emission is broadened. ...