Glossary
... Dynamics − the branch of mechanics dealing with the motion of physical systems. Equilibrium − stable, at minimum energy with no apparent motion. Equipartition theorem − consequence of the kinetic molecular theory that molecules have average kinetic energy proportional to the number of different type ...
... Dynamics − the branch of mechanics dealing with the motion of physical systems. Equilibrium − stable, at minimum energy with no apparent motion. Equipartition theorem − consequence of the kinetic molecular theory that molecules have average kinetic energy proportional to the number of different type ...
A-level Chemistry
... Teaching and learning methods used Teaching and learning methods used include lectures, group work, extensive practical work, independent learning and external workshops through professional membership of external organisations How your work will be assessed Routine formative and summative assessmen ...
... Teaching and learning methods used Teaching and learning methods used include lectures, group work, extensive practical work, independent learning and external workshops through professional membership of external organisations How your work will be assessed Routine formative and summative assessmen ...
SUMMER WORK AP Chemistry
... experiment requires 15.0 g of cyclohexane, whose density at 25 °C is 0.7781 g/mL. What volume of cyclohexane should be used? (c) A spherical ball of lead has a diameter of 5.0 cm. What is the mass of the sphere if lead has a density of 11.34 g.cm3? (The volume of a sphere is (4/3)πr3where r is the r ...
... experiment requires 15.0 g of cyclohexane, whose density at 25 °C is 0.7781 g/mL. What volume of cyclohexane should be used? (c) A spherical ball of lead has a diameter of 5.0 cm. What is the mass of the sphere if lead has a density of 11.34 g.cm3? (The volume of a sphere is (4/3)πr3where r is the r ...
harvesting-aware energy management for time
... CPSs that utilize Wireless Sensor Networking (WSN) technologies will require the use of energy harvesting methods to extend their lifetimes. For this application class, there are currently few algorithmic techniques that combine performance sensitive processing and communication with efficient manag ...
... CPSs that utilize Wireless Sensor Networking (WSN) technologies will require the use of energy harvesting methods to extend their lifetimes. For this application class, there are currently few algorithmic techniques that combine performance sensitive processing and communication with efficient manag ...
2 - Yale University
... Jack Dunitz: At the time when I was reading that book I was wondering whether chemistry was really as interesting as I had hoped it was going to be. And I think I was almost ready to give it up and do something else. I didn't care very much for this chemistry which was full of facts and recipes and ...
... Jack Dunitz: At the time when I was reading that book I was wondering whether chemistry was really as interesting as I had hoped it was going to be. And I think I was almost ready to give it up and do something else. I didn't care very much for this chemistry which was full of facts and recipes and ...
Quantum Chemistry for Excited States of Large Molecules: from
... In my talk, the current status of existing methods for the computation of electronic excited states of large and medium-sized molecules will be discussed. In particular the potential of time-dependent density functional theory (TDDFT) and its failure for charge-transfer excited states will be highli ...
... In my talk, the current status of existing methods for the computation of electronic excited states of large and medium-sized molecules will be discussed. In particular the potential of time-dependent density functional theory (TDDFT) and its failure for charge-transfer excited states will be highli ...
Learning Standards vocab chemical basis and molecules of life 09
... sources of food and nutrition, fossil fuels). Describe at least three chemical reactions of particular importance to humans (e.g., burning of fossil fuels, photosynthesis, rusting of metals). Use a chemical equation to illustrate how the atoms in molecules are arranged before and after a reactio ...
... sources of food and nutrition, fossil fuels). Describe at least three chemical reactions of particular importance to humans (e.g., burning of fossil fuels, photosynthesis, rusting of metals). Use a chemical equation to illustrate how the atoms in molecules are arranged before and after a reactio ...
Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory
... 2 valence electrons max: H, He 8 valence electrons max: 2nd row elements <8 valence electrons exceptions: Be=4, B=6 (formal charge) >8 valence electrons: 3rd row elements and below Consider formal charges in evaluating structures When to draw resonance structures, and how many (reso ...
... 2 valence electrons max: H, He 8 valence electrons max: 2nd row elements <8 valence electrons exceptions: Be=4, B=6 (formal charge) >8 valence electrons: 3rd row elements and below Consider formal charges in evaluating structures When to draw resonance structures, and how many (reso ...
Ch 8 AP Practice
... (a) Write the equation for the ionization of atomic fluorine that requires 1,681.0 kJ mol-1. (b) Account for the fact that the first ionization energy of atomic fluorine is greater than that of atomic oxygen. (You must discuss both atoms in your response.) (c) Predict whether the first ionization en ...
... (a) Write the equation for the ionization of atomic fluorine that requires 1,681.0 kJ mol-1. (b) Account for the fact that the first ionization energy of atomic fluorine is greater than that of atomic oxygen. (You must discuss both atoms in your response.) (c) Predict whether the first ionization en ...
Chapter 1: Chemistry and You
... 2015-2016 Chemistry Midterm Review This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests a ...
... 2015-2016 Chemistry Midterm Review This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests a ...
chapter 6 sec 2 resonance structure
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
Diablo Valley College Course Outline for CHEM
... A. Apply the basic concepts of chemistry, both as ends in themselves and as bases for further study in chemistry. B. Analyze and solve both quantitative and qualitative problems as well as be able to explain the results verbally or in a written fashion. C. Apply the inductive-deductive process by wh ...
... A. Apply the basic concepts of chemistry, both as ends in themselves and as bases for further study in chemistry. B. Analyze and solve both quantitative and qualitative problems as well as be able to explain the results verbally or in a written fashion. C. Apply the inductive-deductive process by wh ...
Honors Midterm Review – 2015-16
... _________ responsible for the uncertainty principle which states that it is impossible to know (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus ...
... _________ responsible for the uncertainty principle which states that it is impossible to know (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus ...
Books
... 5. V. I. Arnold - Ordinary Differential Equations, MIT Press, Cambridge, MA (1973). 6. A. D. Bruno - Local Methods in Nonlinear Differential Equations, Springer-Verlag, Heidelberg (1989). 7. D. R. Smith - Singular Perturbation Theory, Cambridge University Press, New York ...
... 5. V. I. Arnold - Ordinary Differential Equations, MIT Press, Cambridge, MA (1973). 6. A. D. Bruno - Local Methods in Nonlinear Differential Equations, Springer-Verlag, Heidelberg (1989). 7. D. R. Smith - Singular Perturbation Theory, Cambridge University Press, New York ...
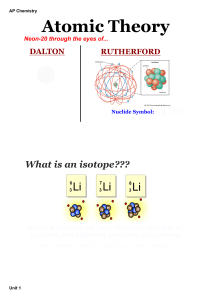
Atomic Line Emission Spectra and Niels Bohr
... Bohr said classical view is wrong. Need a new theory — now called QUANTUM or WAVE MECHANICS. e- can only exist in certain discrete orbits e- is restricted to QUANTIZED energy state (quanta = bundles of energy) ...
... Bohr said classical view is wrong. Need a new theory — now called QUANTUM or WAVE MECHANICS. e- can only exist in certain discrete orbits e- is restricted to QUANTIZED energy state (quanta = bundles of energy) ...
Role of mathematics in chemistry
... quantification of empirical data (empirical models) (b) statistical chemical data analysis, quantitative analytical chemistry, emerging into cheminformatics and chemical data analysis (c) molecular similarity described in terms of target phenotypes leading to quantitative structure– activity correla ...
... quantification of empirical data (empirical models) (b) statistical chemical data analysis, quantitative analytical chemistry, emerging into cheminformatics and chemical data analysis (c) molecular similarity described in terms of target phenotypes leading to quantitative structure– activity correla ...