Physical Chemistry 3: — Chemical Kinetics - Christian
... The scriptum gives a summary of the material covered in the lecture classes to allow students to repeat the material more economically. It covers basic material that all chemistry students should learn irrespective of their possible inclination towards inorganic, organic or physical chemistry, but g ...
... The scriptum gives a summary of the material covered in the lecture classes to allow students to repeat the material more economically. It covers basic material that all chemistry students should learn irrespective of their possible inclination towards inorganic, organic or physical chemistry, but g ...
Peter Ertl - American Chemical Society
... An analysis of the substituents described hitherto produced a large database of about 850 000 substituents. To use this database in practical applications, the substituents need to be classified in some way. For applications in drug design, a classification based on substituent physicochemical prope ...
... An analysis of the substituents described hitherto produced a large database of about 850 000 substituents. To use this database in practical applications, the substituents need to be classified in some way. For applications in drug design, a classification based on substituent physicochemical prope ...
FREE Sample Here - We can offer most test bank and
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
HEAd START TO A LEVEL CHEMISTRY WORKbOOK
... The aim of the booklet is to help students to practise their skills in the areas of formulae, equations and simple mole equations. This booklet gives examples for students to work through to help build their confidence. During the summer holidays, I would like you to use your GCSE knowledge and exam ...
... The aim of the booklet is to help students to practise their skills in the areas of formulae, equations and simple mole equations. This booklet gives examples for students to work through to help build their confidence. During the summer holidays, I would like you to use your GCSE knowledge and exam ...
Introduction to Chemistry
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
mole - hrsbstaff.ednet.ns.ca
... Chapter 10 - How does a chemist measure? How does the chemist count? 2H2(g) + O2 → 2H2O(g) This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist ...
... Chapter 10 - How does a chemist measure? How does the chemist count? 2H2(g) + O2 → 2H2O(g) This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist ...
4) What is the term for the procedure of collecting data and recording
... What is the term for the value which indicates the number of protons for an atom of a given element? A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the atomic model that describes electrons circling the nucleus in an orbit of specific ener ...
... What is the term for the value which indicates the number of protons for an atom of a given element? A) atomic notation B) atomic number C) atomic mass D) mass number E) none of the above What is the term for the atomic model that describes electrons circling the nucleus in an orbit of specific ener ...
6 Chemical Bonding – Orbital Theory
... terms of the orbital theory of atomic structure. Heitler and London believed that electron cloud of the valence orbital on one atom ‘overlaps’ the electron cloud of the other bonding atom to form a covalent linkage. On the contrary, the electrovalent bond formation involves a physical transfer of th ...
... terms of the orbital theory of atomic structure. Heitler and London believed that electron cloud of the valence orbital on one atom ‘overlaps’ the electron cloud of the other bonding atom to form a covalent linkage. On the contrary, the electrovalent bond formation involves a physical transfer of th ...
M.Sc. Part-I Chemistry - North Maharashtra University
... State functions, exact and inexact differentials, test of exactness, internal energy change in terms of internal pressure and ୴ܥ, change in internal energy at constant pressure, expansion coefficient, temperature dependence of enthalpy, isothermal compressibility. Joule-Thomson effect: J-T experi ...
... State functions, exact and inexact differentials, test of exactness, internal energy change in terms of internal pressure and ୴ܥ, change in internal energy at constant pressure, expansion coefficient, temperature dependence of enthalpy, isothermal compressibility. Joule-Thomson effect: J-T experi ...
Unit 3 Answer Key
... 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen atoms. 4. You would not be able to see one person, but a mole of people is so many that they would be visible, as a group, from space. In fact, a mole of pe ...
... 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen atoms. 4. You would not be able to see one person, but a mole of people is so many that they would be visible, as a group, from space. In fact, a mole of pe ...
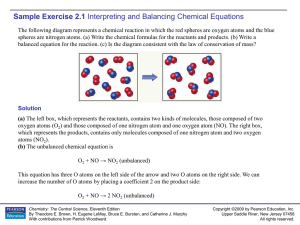
Sample Exercise 2.1
... Analyze We are given both the amount of a substance (0.350 mol) and its chemical formula (C 6H12O6). The unknown is the number of H atoms in the sample. Plan Avogadro’s number provides the conversion factor between the number of moles of C 6H12O6 and the number of molecules of C6H12O6. Once we know ...
... Analyze We are given both the amount of a substance (0.350 mol) and its chemical formula (C 6H12O6). The unknown is the number of H atoms in the sample. Plan Avogadro’s number provides the conversion factor between the number of moles of C 6H12O6 and the number of molecules of C6H12O6. Once we know ...
chemistry - Ethiopian Ministry of Education
... Chemistry is the science that deals with matter and the changes that it undergoes. It is a study of the composition, structure, and properties of matter and of the changes that occur in matter. Perhaps the only permanent thing in the world is change. Iron rusts, snow melts, paints peel off and firew ...
... Chemistry is the science that deals with matter and the changes that it undergoes. It is a study of the composition, structure, and properties of matter and of the changes that occur in matter. Perhaps the only permanent thing in the world is change. Iron rusts, snow melts, paints peel off and firew ...
Under Choice Based Credit System Proposed syllabus and Scheme of Examination
... Chemical Bonding and Molecular Structure Ionic Bonding: General characteristics of ionic bonding. Energy considerations in ionic bonding, lattice energy and solvation energy and their importance in the context of stability and solubility of ionic compounds. Statement of Born-Landé equation for calcu ...
... Chemical Bonding and Molecular Structure Ionic Bonding: General characteristics of ionic bonding. Energy considerations in ionic bonding, lattice energy and solvation energy and their importance in the context of stability and solubility of ionic compounds. Statement of Born-Landé equation for calcu ...
Minimum electrophilicity principle in Lewis acid–base complexes of
... 2. Results and discussion To reinvestigate the acidity strength of some boron trihalides (BX3; X = F, Cl and Br) from theoretical point of view, two sets of Lewis bases (weak and strong), which can form stable compounds with these acids, are considered here. It is expected that more stable complexes ...
... 2. Results and discussion To reinvestigate the acidity strength of some boron trihalides (BX3; X = F, Cl and Br) from theoretical point of view, two sets of Lewis bases (weak and strong), which can form stable compounds with these acids, are considered here. It is expected that more stable complexes ...
When wood, paper, and wax are burned, they ap
... In this chapter we will use what we have learned about chemical structure and formulas in studying the mass relationships of atoms and molecules. These relationships in turn will help us to explain the composition of compounds and the ways in which the composition changes. The mass of an atom is rel ...
... In this chapter we will use what we have learned about chemical structure and formulas in studying the mass relationships of atoms and molecules. These relationships in turn will help us to explain the composition of compounds and the ways in which the composition changes. The mass of an atom is rel ...
Conceptual Physics
... HS-PS3-2. Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects). HS-PS3-3. Design, build, and refine a device ...
... HS-PS3-2. Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects). HS-PS3-3. Design, build, and refine a device ...
Type - Enrico Fermi High
... What is the most likely type of IMF’s present in a substance if the substance has a high vapor pressure? dispersion forces As you go down the nitrogen group of the Periodic Table (N, P, As, Sb), the vapor pressures go down and the boiling points increase. Account for this based on IMF’s. As go down, ...
... What is the most likely type of IMF’s present in a substance if the substance has a high vapor pressure? dispersion forces As you go down the nitrogen group of the Periodic Table (N, P, As, Sb), the vapor pressures go down and the boiling points increase. Account for this based on IMF’s. As go down, ...
orange review book_2014_key
... To the Teacher: The Answer Key for the Brief Review in Chemistry provides answers to all of the questions in the book, including the sample Regents Examinations provided in the back of the book. To determine concepts that might require more intense review, students can take the Diagnostic Tests prov ...
... To the Teacher: The Answer Key for the Brief Review in Chemistry provides answers to all of the questions in the book, including the sample Regents Examinations provided in the back of the book. To determine concepts that might require more intense review, students can take the Diagnostic Tests prov ...
BASIC CONCEPTS OF CHEMISTRY
... only the initial and final states of the interacting bodies without considering the way in which the process takes place, and the development of the process in time. This approach is called the thermodynamics. For convenience, the objects of study should be isolated. Such a collection of bodies extr ...
... only the initial and final states of the interacting bodies without considering the way in which the process takes place, and the development of the process in time. This approach is called the thermodynamics. For convenience, the objects of study should be isolated. Such a collection of bodies extr ...
Water deuteration and ortho-to-para nuclear spin ratio of H2 in
... cloud, we use the one-dimensional shock model developed by B04 and H10. Here we briefly outline the model, while more details can be found in the original papers. The model describes the evolution of post-shock materials in a plane-parallel configuration. In order to obtain density and gas temperatu ...
... cloud, we use the one-dimensional shock model developed by B04 and H10. Here we briefly outline the model, while more details can be found in the original papers. The model describes the evolution of post-shock materials in a plane-parallel configuration. In order to obtain density and gas temperatu ...
Chemical Quantities(mole).
... Ideal gas Law: PV = nRT P = pressure (kPa) V = volume (Liters) n = 1 mole R = constant value (8.31 kPa ∙ L) mol ∙ K T = temperature (Kelvin) ...
... Ideal gas Law: PV = nRT P = pressure (kPa) V = volume (Liters) n = 1 mole R = constant value (8.31 kPa ∙ L) mol ∙ K T = temperature (Kelvin) ...
Ciprofloxacin Hcl (Cas No 86393-32-0)
... strengths. Ciprofloxacin Oral Suspension is a white to slightly yellowish suspension with strawberry flavor which may contain yellow-orange droplets. It is composed of ciprofloxacin microcapsules and diluent which are mixed prior to dispensing (See Instructions for Use/Handling). The components of t ...
... strengths. Ciprofloxacin Oral Suspension is a white to slightly yellowish suspension with strawberry flavor which may contain yellow-orange droplets. It is composed of ciprofloxacin microcapsules and diluent which are mixed prior to dispensing (See Instructions for Use/Handling). The components of t ...
From Ultracold Atoms to Condensed Matter Physics
... results: indeed one typically works in the dilute limit, at low temperatures, such that the s-wave scattering length as is the only parameter needed to describe the interactions. This means that the results obtained with one set of atoms will not depend on the details of the short-range atomic physi ...
... results: indeed one typically works in the dilute limit, at low temperatures, such that the s-wave scattering length as is the only parameter needed to describe the interactions. This means that the results obtained with one set of atoms will not depend on the details of the short-range atomic physi ...
Computational investigations of the electronic structure of molecular
... In this PhD thesis the electronic structure of a range of actinide compounds has been investigated using density functional theory. The reason for using DFT instead of other methods is mainly due to the size of the compounds which makes multireference calculations prohibitively expensive, but also t ...
... In this PhD thesis the electronic structure of a range of actinide compounds has been investigated using density functional theory. The reason for using DFT instead of other methods is mainly due to the size of the compounds which makes multireference calculations prohibitively expensive, but also t ...