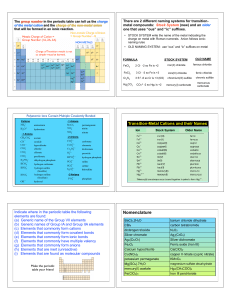
Calculations and the Chemical Equation
... 2. How many grams of sulfur are found in 0.150 mol of sulfur? [Use atomic weight: S, 32.06 amu] 3. How many moles of sulfur are found in 1.81 ´ 1024 atoms of sulfur? [Use atomic weight: S, 32.06 amu] 4. How many atoms are present in a 7.31 g sample of copper? [Use atomic weight: Cu, 63.55 amu] 5. Wh ...
... 2. How many grams of sulfur are found in 0.150 mol of sulfur? [Use atomic weight: S, 32.06 amu] 3. How many moles of sulfur are found in 1.81 ´ 1024 atoms of sulfur? [Use atomic weight: S, 32.06 amu] 4. How many atoms are present in a 7.31 g sample of copper? [Use atomic weight: Cu, 63.55 amu] 5. Wh ...
Chapter 3 Sem 2 2013-14
... 2 atoms of Al and 3 molecules of (SO4)2- = 1 formula unit Al2(SO4)3 2 moles of Al and 3 moles of (SO4)2- = 1 formula unit Al2(SO4)3 1 formula unit Al2(SO4)3 = 342.17 amu Al2(SO4)3 1 mole Al2(SO4)3 = 342.17 g Al2(SO4)3 1 mole Al2(SO4)3 = 6.022 x 1023 formula units Al2(SO4)3 1 mole Al2(SO4)3 = 2 mol A ...
... 2 atoms of Al and 3 molecules of (SO4)2- = 1 formula unit Al2(SO4)3 2 moles of Al and 3 moles of (SO4)2- = 1 formula unit Al2(SO4)3 1 formula unit Al2(SO4)3 = 342.17 amu Al2(SO4)3 1 mole Al2(SO4)3 = 342.17 g Al2(SO4)3 1 mole Al2(SO4)3 = 6.022 x 1023 formula units Al2(SO4)3 1 mole Al2(SO4)3 = 2 mol A ...
Chemistry Essentials For Dummies
... Greenville, South Carolina. After a stint in the United States Army, he decided to try his hand at teaching. In 1971, he joined the chemistry faculty of Stephen F. Austin State University in Nacogdoches, Texas where he still teaches chemistry. In 1985, he started back to school part time and in 1991 ...
... Greenville, South Carolina. After a stint in the United States Army, he decided to try his hand at teaching. In 1971, he joined the chemistry faculty of Stephen F. Austin State University in Nacogdoches, Texas where he still teaches chemistry. In 1985, he started back to school part time and in 1991 ...
Chapter 3 2014
... (a) what is the formula mass of Na2B4O7 (b) how many moles of borax is 20.0 g? (c) how many moles of boron are present in 20.0 g Na2B4O7? (d) how many grams of boron are present in 20.0 g Na2B4O7? (e) how many atoms of B are present in 20.0g? (f) how many atoms of O are present in 20.0g? (g) how man ...
... (a) what is the formula mass of Na2B4O7 (b) how many moles of borax is 20.0 g? (c) how many moles of boron are present in 20.0 g Na2B4O7? (d) how many grams of boron are present in 20.0 g Na2B4O7? (e) how many atoms of B are present in 20.0g? (f) how many atoms of O are present in 20.0g? (g) how man ...
A Model For the Calculation of Solvent ... Reaction Rates for Process Design Purposes
... mountain and down one of its steepest trails. I wish to thank Professor George Stephanopoulos for replicating the sensation of the ride down over the course of my research. The way was hard, full of bumps and falls but with his guidance and support it was also an exciting experience and full of lear ...
... mountain and down one of its steepest trails. I wish to thank Professor George Stephanopoulos for replicating the sensation of the ride down over the course of my research. The way was hard, full of bumps and falls but with his guidance and support it was also an exciting experience and full of lear ...
Stoichiometry
... • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
Regents Chemistry Review - New York Science Teacher
... • Which of the following numerical setups can be used to calculate the atomic mass of Si? (1) (27.98 + 28.98 + 29.97)/3 x 100 ...
... • Which of the following numerical setups can be used to calculate the atomic mass of Si? (1) (27.98 + 28.98 + 29.97)/3 x 100 ...
Here
... (b) In the Bohr model, a hydrogen atom consists of an electron in a circular orbit about a proton. (c) A conical flask used in chemistry labs to carry out reactions. (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero ...
... (b) In the Bohr model, a hydrogen atom consists of an electron in a circular orbit about a proton. (c) A conical flask used in chemistry labs to carry out reactions. (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero ...
No Slide Title
... sum of the atomic masses (in amu) in a molecule. For all practical purposes think of amu as grams ...
... sum of the atomic masses (in amu) in a molecule. For all practical purposes think of amu as grams ...
Chapter 14 (Kinetics) – Slides and Practice
... because the reactants run out or because the system has reached equilibrium ...
... because the reactants run out or because the system has reached equilibrium ...
Non-ideal Plastic Behavior
... food ingredients, and how they are effected by changes in environment, such as temperature, pressure and mechanical agitation. Basic research tends to be carried out on simple model systems with well-defined ...
... food ingredients, and how they are effected by changes in environment, such as temperature, pressure and mechanical agitation. Basic research tends to be carried out on simple model systems with well-defined ...
Chemistry: Percent Yield
... Unit 3/4 – Moles / Stoichiometry proportion. A chemical compound can be broken down by chemical means. A chemical compound can be represented by a specific chemical formula and assigned a name based on the IUPAC system. 35: 3.3f The percent composition by mass of each element in a compound can be c ...
... Unit 3/4 – Moles / Stoichiometry proportion. A chemical compound can be broken down by chemical means. A chemical compound can be represented by a specific chemical formula and assigned a name based on the IUPAC system. 35: 3.3f The percent composition by mass of each element in a compound can be c ...
M.Sc. Chemistry - Periyar University
... rate – Hard – Sphere collision theory of reaction rates – molecular beams – Reaction cross section – effectiveness of collisions – Probability factor. Transition state theory of reaction rates – Potential energy surface – Partition functions and activated complex – Eyring equation – Comparison of co ...
... rate – Hard – Sphere collision theory of reaction rates – molecular beams – Reaction cross section – effectiveness of collisions – Probability factor. Transition state theory of reaction rates – Potential energy surface – Partition functions and activated complex – Eyring equation – Comparison of co ...
Chemistry 101L
... will be making. Remember to include room for multiple trials and average values, if appropriate. If appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that th ...
... will be making. Remember to include room for multiple trials and average values, if appropriate. If appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that th ...
Physical Chemistry 3: — Chemical Kinetics
... Reactions in the solid phase: • Atoms and molecules localized on fixed lattice positions. • Reaction rate is determined by rate of diffusion (“hopping”) of the atoms and molecules via vacancies (unoccupied lattice positions, “Fehlstellen”) or interstitial sites (“Zwischengitterplätze”). • Hopping fro ...
... Reactions in the solid phase: • Atoms and molecules localized on fixed lattice positions. • Reaction rate is determined by rate of diffusion (“hopping”) of the atoms and molecules via vacancies (unoccupied lattice positions, “Fehlstellen”) or interstitial sites (“Zwischengitterplätze”). • Hopping fro ...
5 pancakes 2 eggs
... 3 blocks cream cheese + 5 eggs + 1 cup sugar = 1 cheese cake. How many eggs should you use with 9 blocks of cheese? 5 eggs 9 blocks cheese x 3 blocks cheese= 15 eggs How much sugar do we need for 5 cheese cakes? (5) ...
... 3 blocks cream cheese + 5 eggs + 1 cup sugar = 1 cheese cake. How many eggs should you use with 9 blocks of cheese? 5 eggs 9 blocks cheese x 3 blocks cheese= 15 eggs How much sugar do we need for 5 cheese cakes? (5) ...
BS Chemistry - Government College University Faisalabad
... Ideal and real gases, equations of state, critical phenomenon and critical constants. Molecules in motion collision diameter and mean free path. Physical properties of liquids surface tension, viscosity, refractive index etc. and their properties of liquids; surface tension, viscosity, refractive in ...
... Ideal and real gases, equations of state, critical phenomenon and critical constants. Molecules in motion collision diameter and mean free path. Physical properties of liquids surface tension, viscosity, refractive index etc. and their properties of liquids; surface tension, viscosity, refractive in ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... • The limiting reactant is the reactant present in the smallest stoichiometric amount. – In other words, it’s the reactant you’ll run out of first (in this case, the H2). ...
... • The limiting reactant is the reactant present in the smallest stoichiometric amount. – In other words, it’s the reactant you’ll run out of first (in this case, the H2). ...
Cyclam ``capa` POT.4` to ``capa` POT.3` denticity change
... example, blood pressure reduction effects53,54 and antiproliferative and trypanocidal55 activities. For example,54 administration of the less toxic trans-[Ru(NO)Cl(cyclam)]2+ to hypertensive male Wistar rats resulted in blood pressure reduction 26 times longer than sodium nitroprusside (SNP), a clas ...
... example, blood pressure reduction effects53,54 and antiproliferative and trypanocidal55 activities. For example,54 administration of the less toxic trans-[Ru(NO)Cl(cyclam)]2+ to hypertensive male Wistar rats resulted in blood pressure reduction 26 times longer than sodium nitroprusside (SNP), a clas ...
expected output
... SYLLABUS:The concept of development and underdevelopment; socio-economic indicators of growth and development; group dynamics, organising people and activities, e.g. Harambee, e.t.c. division of labour; fundamentals of project management technology and society; role and responsibility project manage ...
... SYLLABUS:The concept of development and underdevelopment; socio-economic indicators of growth and development; group dynamics, organising people and activities, e.g. Harambee, e.t.c. division of labour; fundamentals of project management technology and society; role and responsibility project manage ...
expected output
... SYLLABUS:The concept of development and underdevelopment; socio-economic indicators of growth and development; group dynamics, organising people and activities, e.g. Harambee, e.t.c. division of labour; fundamentals of project management technology and society; role and responsibility project manage ...
... SYLLABUS:The concept of development and underdevelopment; socio-economic indicators of growth and development; group dynamics, organising people and activities, e.g. Harambee, e.t.c. division of labour; fundamentals of project management technology and society; role and responsibility project manage ...
chemistry - My Study materials – Kumar
... Molecule is the smallest particle of a compound. Atoms exist in free states in the form of molecule. A molecule may be formed by the combination of two or more similar atoms of an element, such asoxygen molecule is formed by the combination of two oxygen atoms, molecule of hydrogen which is formed b ...
... Molecule is the smallest particle of a compound. Atoms exist in free states in the form of molecule. A molecule may be formed by the combination of two or more similar atoms of an element, such asoxygen molecule is formed by the combination of two oxygen atoms, molecule of hydrogen which is formed b ...