Prentice Hall Ch 02 Atoms Molecules Ions
... General Chemistry 4th edition, Hill, Petrucci, McCreary, Perry ...
... General Chemistry 4th edition, Hill, Petrucci, McCreary, Perry ...
East Meck Chemistry
... Because of Dalton’s atomic theory, most scientists in the 1800s believed that the atom was like a tiny solid ball that could not be broken up into parts. In 1897, a British physicist, J.J. Thomson, discovered that this solid-ball model was not accurate. Thomson’s experiments used a _________________ ...
... Because of Dalton’s atomic theory, most scientists in the 1800s believed that the atom was like a tiny solid ball that could not be broken up into parts. In 1897, a British physicist, J.J. Thomson, discovered that this solid-ball model was not accurate. Thomson’s experiments used a _________________ ...
L-11 Chemical thermodynamics
... You have learnt that chemical reactions are accompanied by energy changes. How do we determine these energy changes? You know that we cannot create or destroy energy. Energy only changes from one form to another. This is the observation made by many scientists over the years. This observation has ta ...
... You have learnt that chemical reactions are accompanied by energy changes. How do we determine these energy changes? You know that we cannot create or destroy energy. Energy only changes from one form to another. This is the observation made by many scientists over the years. This observation has ta ...
STUDY MATERIAL 2015-16 CHEMISTRY CLASS XI
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
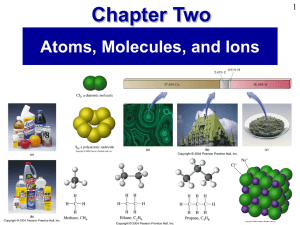
Molecules, Compounds, and Chemical Equations
... The properties of compounds are generally very different from the properties of the elements that compose them. When two elements combine to form a compound, an entirely new substance results. Common table salt, for example, is a compound composed of sodium and chlorine. Sodium is a highly reactive, ...
... The properties of compounds are generally very different from the properties of the elements that compose them. When two elements combine to form a compound, an entirely new substance results. Common table salt, for example, is a compound composed of sodium and chlorine. Sodium is a highly reactive, ...
Stoichiometry: Calculations with Chemical Formulas and
... Molar Mass • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... Molar Mass • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
Chemistry - talcher autonomous college
... collision frequency; collision diameter; mean free path and viscosity of gases, including their temperature and pressure dependence, relation between mean free path and coefficient of viscosity, calculation of σ from η; variation of viscosity with temperature and pressure. Maxwell distribution and i ...
... collision frequency; collision diameter; mean free path and viscosity of gases, including their temperature and pressure dependence, relation between mean free path and coefficient of viscosity, calculation of σ from η; variation of viscosity with temperature and pressure. Maxwell distribution and i ...
P. LeClair
... 1. A spherical volume of radius a is filled with charge of uniform density ρ. We want to know the potential energy U of this sphere of charge, that is, the work done in assembling it. Calculate it by building up the sphere up layer by layer, making use of the fact that the field outside a spherical ...
... 1. A spherical volume of radius a is filled with charge of uniform density ρ. We want to know the potential energy U of this sphere of charge, that is, the work done in assembling it. Calculate it by building up the sphere up layer by layer, making use of the fact that the field outside a spherical ...
Mechanochemistry: the varied applications of mechanical bond
... milling.3–7 All of these reactions are chemically driven and preparatively used. Exhaustive reviews are available for >1000 solid-state molecular reactions that are waste-free with 100% yield8 and for stereoselective ones.9 The mechanistic aspects have also been reviewed.1,10 Unfortunately, various ...
... milling.3–7 All of these reactions are chemically driven and preparatively used. Exhaustive reviews are available for >1000 solid-state molecular reactions that are waste-free with 100% yield8 and for stereoselective ones.9 The mechanistic aspects have also been reviewed.1,10 Unfortunately, various ...
Chemistry 120
... liquids such as acetone, hexane, benzene or ether or water. Water is the most important solvent. The oceans cover ~ ¾ of the surface of the planet and every cell is mainly composed of water. Solutions in water are termed aqueous solutions and species are written as E(aq). ...
... liquids such as acetone, hexane, benzene or ether or water. Water is the most important solvent. The oceans cover ~ ¾ of the surface of the planet and every cell is mainly composed of water. Solutions in water are termed aqueous solutions and species are written as E(aq). ...
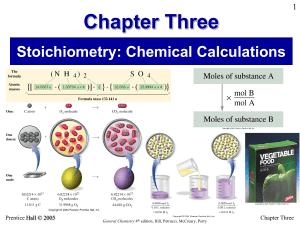
Stoichiometry Objectives
... Sulfuric acid is formed when sulfur dioxide reacts with oxygen and water. Write the balanced chemical equation for the reaction. If 12.5 mol SO2 reacts, how many moles H2SO4 can be produced? How many mole O2 is needed? ...
... Sulfuric acid is formed when sulfur dioxide reacts with oxygen and water. Write the balanced chemical equation for the reaction. If 12.5 mol SO2 reacts, how many moles H2SO4 can be produced? How many mole O2 is needed? ...
Chapter Three - CNG Chemistry
... In these examples, note the common practice of expressing molar mass and Avogadro’s number with at least one significant figure more than the number of significant figures in the least precisely known quantity. Doing this ensures that the precision of the calculated results is limited only by the le ...
... In these examples, note the common practice of expressing molar mass and Avogadro’s number with at least one significant figure more than the number of significant figures in the least precisely known quantity. Doing this ensures that the precision of the calculated results is limited only by the le ...
Chemical bonding and structure
... as protons and electrons. This is because the number of protons (+) is equal to the number of electrons (−), and so their charges cancel each other out. The positively charged protons, located within the nucleus of the atom, are not transferred during chemical reactions. Electrons, however, position ...
... as protons and electrons. This is because the number of protons (+) is equal to the number of electrons (−), and so their charges cancel each other out. The positively charged protons, located within the nucleus of the atom, are not transferred during chemical reactions. Electrons, however, position ...
Holt Modern Chemistry Workbook
... whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain two or more atoms of the same element, as in oxygen. Or, a molecule may consist of two or more atoms of different eleme ...
... whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain two or more atoms of the same element, as in oxygen. Or, a molecule may consist of two or more atoms of different eleme ...
Name: ______KEY__________________ Date: ______ CHM 130
... _E__ 1. (5 pts) Which of the following describes properties of gases? a. They are compressible. b. They expand to fill a container. c. They take the shape of a container. d. The particles in a gas are far apart. e. All of the above are true. ...
... _E__ 1. (5 pts) Which of the following describes properties of gases? a. They are compressible. b. They expand to fill a container. c. They take the shape of a container. d. The particles in a gas are far apart. e. All of the above are true. ...
File
... Only whole-number ratios make physical sense because molecules contain whole atoms. The 3.03 in this case could result from a small experimental error in the molecular weight. We therefore multiply each subscript in the empirical formula by 3 to give the molecular formula: C9H12. Check We can have c ...
... Only whole-number ratios make physical sense because molecules contain whole atoms. The 3.03 in this case could result from a small experimental error in the molecular weight. We therefore multiply each subscript in the empirical formula by 3 to give the molecular formula: C9H12. Check We can have c ...
Electron configuration
... • This reaction tells us that by mixing 1 mole of methane with 2 moles of oxygen we will get 1 mole of carbon dioxide and 2 moles of water. • If we want to get 10 moles of water, how many moles of methane and oxygen is needed? How many grams of CO2 is produced? ...
... • This reaction tells us that by mixing 1 mole of methane with 2 moles of oxygen we will get 1 mole of carbon dioxide and 2 moles of water. • If we want to get 10 moles of water, how many moles of methane and oxygen is needed? How many grams of CO2 is produced? ...
Theories of the constitution of gases in the early nineteenth century
... At the beginning of the nineteenth century those chemists who speculated on the microscopic nature of gases used a model based on small nuclei, different for each chemical element, to which only "imponderable" elements: heat, light, electricity or motion had to added to provide all the physical prop ...
... At the beginning of the nineteenth century those chemists who speculated on the microscopic nature of gases used a model based on small nuclei, different for each chemical element, to which only "imponderable" elements: heat, light, electricity or motion had to added to provide all the physical prop ...
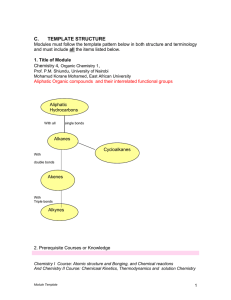
Organic Molecules
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
2.6 M - Thierry Karsenti
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
03_Worked_Examples
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. The Na and O atoms are balanced, but the number of H atoms is not. To increase the number of H atoms on the left, let’s try placing the ...
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. The Na and O atoms are balanced, but the number of H atoms is not. To increase the number of H atoms on the left, let’s try placing the ...
Practice Exercise 1
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. The Na and O atoms are balanced, but the number of H atoms is not. To increase the number of H atoms on the left, let’s try placing the ...
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. The Na and O atoms are balanced, but the number of H atoms is not. To increase the number of H atoms on the left, let’s try placing the ...
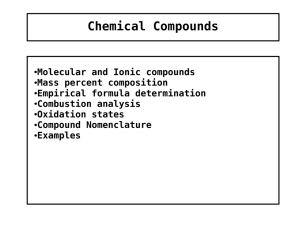
Chemical Compounds
... 4. The oxidation state of hydrogen is generally +1 except when it is bonded to metals such as sodium (NaH) in which case it's oxidation number is -1. 5. Fluorine has an oxidation number of -1 in its compounds … always. Group 1 elements have an oxidation number of +1 in their compounds … always. Grou ...
... 4. The oxidation state of hydrogen is generally +1 except when it is bonded to metals such as sodium (NaH) in which case it's oxidation number is -1. 5. Fluorine has an oxidation number of -1 in its compounds … always. Group 1 elements have an oxidation number of +1 in their compounds … always. Grou ...