purine
... Purine Catabolism and Salvage • All purine degradation leads to uric acid • Ingested nucleic acids are degraded to nucleotides by pancreatic nucleases, and intestinal phosphodiesterases in the intestine • Group-specific nucleotidases and non-specific phosphatases degrade nucleotides into nucleoside ...
... Purine Catabolism and Salvage • All purine degradation leads to uric acid • Ingested nucleic acids are degraded to nucleotides by pancreatic nucleases, and intestinal phosphodiesterases in the intestine • Group-specific nucleotidases and non-specific phosphatases degrade nucleotides into nucleoside ...
Lecture 12 Enzymes: Inhibition
... • group-specific chemical modifying reagents that would react with certain types of functional groups on many different enzymes • substrate analogs with a reactive group on them (so more specific for one enzyme) • "suicide" substrates (mechanism-based inhibitors): not reactive until the specific che ...
... • group-specific chemical modifying reagents that would react with certain types of functional groups on many different enzymes • substrate analogs with a reactive group on them (so more specific for one enzyme) • "suicide" substrates (mechanism-based inhibitors): not reactive until the specific che ...
finalcarbohydrat met..
... c) Pyruvate: which can be used in synthesis of amino acid alanine. 4. Aerobic glycolysis provides the mitochondria with pyruvate, which gives acetyl CoA Krebs' cycle. ...
... c) Pyruvate: which can be used in synthesis of amino acid alanine. 4. Aerobic glycolysis provides the mitochondria with pyruvate, which gives acetyl CoA Krebs' cycle. ...
Answers - U of L Class Index
... Glucogenic amino acids can be used to produce intermediates for glucogenesis, which is glucose ...
... Glucogenic amino acids can be used to produce intermediates for glucogenesis, which is glucose ...
Water - UCLA Chemistry and Biochemistry
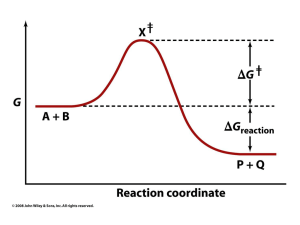
... • Preferential binding of transition state: binding interactions between the enzyme and TS are maximized; they are greater than those in the enzyme-substrate or enzyme-product complexes • General acid and general base catalysis: functional groups of the enzyme donate &/or accept protons • Covalent c ...
... • Preferential binding of transition state: binding interactions between the enzyme and TS are maximized; they are greater than those in the enzyme-substrate or enzyme-product complexes • General acid and general base catalysis: functional groups of the enzyme donate &/or accept protons • Covalent c ...
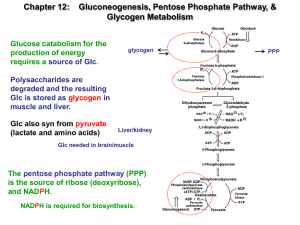
Carbohydrates Metabolism OVERVIEW Carbohydrates (saccharides
... reaction of glycolysis.Because there is only a limited amount of NAD+ in the cell, the NADH formed by this reaction must be reoxidized to NAD+ for glycolysis to continue. Two major mechanisms for oxidizing NADH are 1) the NADH-linked conversion of pyruvate to lactate and 2) oxidation of NADH via the ...
... reaction of glycolysis.Because there is only a limited amount of NAD+ in the cell, the NADH formed by this reaction must be reoxidized to NAD+ for glycolysis to continue. Two major mechanisms for oxidizing NADH are 1) the NADH-linked conversion of pyruvate to lactate and 2) oxidation of NADH via the ...
Phosphoketolase pathway dominates in
... the dry weights of the samples, and, hence, the signal-to-noise levels vary between the spectra. Duplicate samples from different batch cultures were analyzed in this study, and the spectra showed the same results. Enzyme assays. Cell extracts were prepared from cultures harvested in the late expone ...
... the dry weights of the samples, and, hence, the signal-to-noise levels vary between the spectra. Duplicate samples from different batch cultures were analyzed in this study, and the spectra showed the same results. Enzyme assays. Cell extracts were prepared from cultures harvested in the late expone ...
Discovery of substrate cycles in large scale metabolic networks
... either produce or consume an extracellular metabolite from the network, resulting in a graph comprising 1,418 reaction nodes. The rationale for removing reaction nodes connecting extracellular and intracellular metabolites was to focus on EFMs internal to the network. We then removed cofactors (for ...
... either produce or consume an extracellular metabolite from the network, resulting in a graph comprising 1,418 reaction nodes. The rationale for removing reaction nodes connecting extracellular and intracellular metabolites was to focus on EFMs internal to the network. We then removed cofactors (for ...
Slide 1
... UDP-Glc synthases in protists, animals, and fungi. ADP-Glc synthase in plants. Primer of 4 to 8 Glc on a Tyr (-OH) of glycogenin. 1st Glc from UDP-Glc via Glc transferase. Remaining Glc’s tranferred by glycogenin. Amylo-(1,4 1,6)-transglycolase catalyzes the branch point. (Alpha 1-6 link) ...
... UDP-Glc synthases in protists, animals, and fungi. ADP-Glc synthase in plants. Primer of 4 to 8 Glc on a Tyr (-OH) of glycogenin. 1st Glc from UDP-Glc via Glc transferase. Remaining Glc’s tranferred by glycogenin. Amylo-(1,4 1,6)-transglycolase catalyzes the branch point. (Alpha 1-6 link) ...
As a PDF file
... Lactic acid bacteria (LAB) are a heterogeneous group of gram-positive bacteria that produce lactic acid as their main end-product during sugar fermentation. Because the LAB are able to rapidly lower pH through acid formation and additionally produce many flavor compounds, they are commonly used in t ...
... Lactic acid bacteria (LAB) are a heterogeneous group of gram-positive bacteria that produce lactic acid as their main end-product during sugar fermentation. Because the LAB are able to rapidly lower pH through acid formation and additionally produce many flavor compounds, they are commonly used in t ...
Introduction - MRC Laboratory of Molecular Biology
... Circadian rhythms are cell-autonomous phenomena found throughout biology and have been shown to regulate many aspects of health and disease1. Despite this almost ubiquitous observation of cellular timekeeping, the genes generally proposed to be responsible show little or no homology between kingdoms ...
... Circadian rhythms are cell-autonomous phenomena found throughout biology and have been shown to regulate many aspects of health and disease1. Despite this almost ubiquitous observation of cellular timekeeping, the genes generally proposed to be responsible show little or no homology between kingdoms ...
Amino Acids, Proteins, and Enzymes
... • part of the coenzymes flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN). • needed for good vision and healthy skin. O H3C ...
... • part of the coenzymes flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN). • needed for good vision and healthy skin. O H3C ...
Enzyme Mechanisms - Research Centers
... We’ve already outlined the fact that most water-soluble coenzymes are derived from vitamins—typically B vitamins Typically the dietary form can be converted by a fairly short metabolic pathway into the coenzyme form, e.g. ...
... We’ve already outlined the fact that most water-soluble coenzymes are derived from vitamins—typically B vitamins Typically the dietary form can be converted by a fairly short metabolic pathway into the coenzyme form, e.g. ...
Theoretical Approaches to the Evolutionary Optimization of Glycolysis
... These three variables are not strictly independent, since the value of each usually affects the chemical possibilities that allow diversity of the others. Here, the chemical analysis of this problem is presented, showing that a number of different chemical solutions can exist. Then, the best solutio ...
... These three variables are not strictly independent, since the value of each usually affects the chemical possibilities that allow diversity of the others. Here, the chemical analysis of this problem is presented, showing that a number of different chemical solutions can exist. Then, the best solutio ...
BOOK NOTES ch9_sec3
... Aerobic Respiration • Organisms such as humans can use oxygen to produce ATP efficiently through aerobic respiration. • The first stage of aerobic respiration is the Krebs cycle, a series of reactions that produce electron carriers. • The electron carriers enter an electron transport chain, which po ...
... Aerobic Respiration • Organisms such as humans can use oxygen to produce ATP efficiently through aerobic respiration. • The first stage of aerobic respiration is the Krebs cycle, a series of reactions that produce electron carriers. • The electron carriers enter an electron transport chain, which po ...
Biology: Concepts and Connections, 6e (Campbell)
... 21) During cellular respiration, electrons move through a series of electron carrier molecules. Which of the following statements about this process is true? A) The electrons move from carriers that have more affinity for them to carriers that have less affinity for them. B) Molecular oxygen is even ...
... 21) During cellular respiration, electrons move through a series of electron carrier molecules. Which of the following statements about this process is true? A) The electrons move from carriers that have more affinity for them to carriers that have less affinity for them. B) Molecular oxygen is even ...
Biology: Concepts and Connections, 6e
... 21) During cellular respiration, electrons move through a series of electron carrier molecules. Which of the following statements about this process is true? A) The electrons move from carriers that have more affinity for them to carriers that have less affinity for them. B) Molecular oxygen is even ...
... 21) During cellular respiration, electrons move through a series of electron carrier molecules. Which of the following statements about this process is true? A) The electrons move from carriers that have more affinity for them to carriers that have less affinity for them. B) Molecular oxygen is even ...
NADP+/NADPH Assay Kit (Colorimetric)
... FOR RESEARCH USE ONLY Not for use in diagnostic procedures ...
... FOR RESEARCH USE ONLY Not for use in diagnostic procedures ...
Requires Rubisco
... However, plants living in hot climates need to conserve water, which requires them to use low CO2 concentration (water is used in rubisco reaction!) The disadvantage of C4 plants is extra ATP used and a more complex pathway http://methanogens .pdx.edu/boone/courses/BI336/BI336Lectures/200201/BI336Le ...
... However, plants living in hot climates need to conserve water, which requires them to use low CO2 concentration (water is used in rubisco reaction!) The disadvantage of C4 plants is extra ATP used and a more complex pathway http://methanogens .pdx.edu/boone/courses/BI336/BI336Lectures/200201/BI336Le ...
(a) (b)
... pyruvate dehydrogenase (E1), dihydrolipoyl transacetylase (E2), and dihydrolipoyl dehydrogenase (E3)—each present in multiple copies. In the bovine enzyme complex, 60 identical copies of E2 form a dodecahedron core with a diameter of about 25 nm. E2 has three domains: the amino-terminal lipoyl domai ...
... pyruvate dehydrogenase (E1), dihydrolipoyl transacetylase (E2), and dihydrolipoyl dehydrogenase (E3)—each present in multiple copies. In the bovine enzyme complex, 60 identical copies of E2 form a dodecahedron core with a diameter of about 25 nm. E2 has three domains: the amino-terminal lipoyl domai ...
Peer-reviewed Article PDF
... the Citric Acid Cycle [1]. Citrate synthase is localized within eukaryotic cells in the mitochondrial matrix, and it is commonly used as a quantitative enzyme marker for the presence of intact mitochondria. Oxaloacetate - the first substrate which binds to the citrate synthase induces the enzyme to ...
... the Citric Acid Cycle [1]. Citrate synthase is localized within eukaryotic cells in the mitochondrial matrix, and it is commonly used as a quantitative enzyme marker for the presence of intact mitochondria. Oxaloacetate - the first substrate which binds to the citrate synthase induces the enzyme to ...
What are enzymes?
... solutions to their industrial processes, and in many cases enzymes can help them do it. Chemicals used in industrial processes are one of the most severe threats to nature and man today. By using enzymes instead of chemicals, the problem is solved. Enzymes present no threat to the environment whatso ...
... solutions to their industrial processes, and in many cases enzymes can help them do it. Chemicals used in industrial processes are one of the most severe threats to nature and man today. By using enzymes instead of chemicals, the problem is solved. Enzymes present no threat to the environment whatso ...
Enzyme Mechanisms
... covalently bonded to substrate carbonyl carbon, moving - charge to the carbonyl O. Oxyanion is on the substrate's oxygen Oxyanion stabilized by additional interaction in addition to the protonated his 57: main-chain NH group from gly 193 H-bonds to oxygen atom (or ion) from the substrate, further st ...
... covalently bonded to substrate carbonyl carbon, moving - charge to the carbonyl O. Oxyanion is on the substrate's oxygen Oxyanion stabilized by additional interaction in addition to the protonated his 57: main-chain NH group from gly 193 H-bonds to oxygen atom (or ion) from the substrate, further st ...
Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotide (NAD) is a coenzyme found in all living cells. The compound is a dinucleotide, because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide. Nicotinamide adenine dinucleotide exists in two forms, an oxidized and reduced form abbreviated as NAD+ and NADH respectively.In metabolism, nicotinamide adenine dinucleotide is involved in redox reactions, carrying electrons from one reaction to another. The coenzyme is, therefore, found in two forms in cells: NAD+ is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD. However, it is also used in other cellular processes, the most notable one being a substrate of enzymes that add or remove chemical groups from proteins, in posttranslational modifications. Because of the importance of these functions, the enzymes involved in NAD metabolism are targets for drug discovery.In organisms, NAD can be synthesized from simple building-blocks (de novo) from the amino acids tryptophan or aspartic acid. In an alternative fashion, more complex components of the coenzymes are taken up from food as the vitamin called niacin. Similar compounds are released by reactions that break down the structure of NAD. These preformed components then pass through a salvage pathway that recycles them back into the active form. Some NAD is also converted into nicotinamide adenine dinucleotide phosphate (NADP); the chemistry of this related coenzyme is similar to that of NAD, but it has different roles in metabolism.Although NAD+ is written with a superscript plus sign because of the formal charge on a particular nitrogen atom, at physiological pH for the most part it is actually a singly charged anion (charge of minus 1), while NADH is a doubly charged anion.