Reversible Competitive Inhibitor
... can serve as drug targets. Drugs that interfere in an enzyme's function are called inhibitors. Inhibitors are classified as being reversible or irreversible. Among the reversible inhibitors are three different types: competitive, noncompetitive, and uncompetitive. All inhibitors, when bound to an en ...
... can serve as drug targets. Drugs that interfere in an enzyme's function are called inhibitors. Inhibitors are classified as being reversible or irreversible. Among the reversible inhibitors are three different types: competitive, noncompetitive, and uncompetitive. All inhibitors, when bound to an en ...
enzyme names end in “ase”
... LD1 was first discovered in the heart and has four (4) identical protein sub-units. Since they are identical and from the heart, each sub-unit is called an “H” sub-unit. There are four (4) H subunits in LD1. LD5 was identified in muscle and it, too, had four (4) identical sub-units, called “M” for m ...
... LD1 was first discovered in the heart and has four (4) identical protein sub-units. Since they are identical and from the heart, each sub-unit is called an “H” sub-unit. There are four (4) H subunits in LD1. LD5 was identified in muscle and it, too, had four (4) identical sub-units, called “M” for m ...
K m - kois.sk
... - non-competitive inhibitor is not influenced by the concentration of the substrate. It inhibits by binding irreversibly to the enzyme but not at the active site, e.g. cyanide combines with the Iron in the enzymes cytochrome oxidase, heavy metals Ag or Hg combine with –SH groups. These can be remove ...
... - non-competitive inhibitor is not influenced by the concentration of the substrate. It inhibits by binding irreversibly to the enzyme but not at the active site, e.g. cyanide combines with the Iron in the enzymes cytochrome oxidase, heavy metals Ag or Hg combine with –SH groups. These can be remove ...
enzyme kinetics
... with the same enzyme concentration). Plot each data set as product concentration vs. time. Fit the data to a straight line and determine the slope. The slope is the rate of the reaction, V, for a particular value of [S]. Note: In the “Add Trendline” dialog box (accessed from the Chart menu), first s ...
... with the same enzyme concentration). Plot each data set as product concentration vs. time. Fit the data to a straight line and determine the slope. The slope is the rate of the reaction, V, for a particular value of [S]. Note: In the “Add Trendline” dialog box (accessed from the Chart menu), first s ...
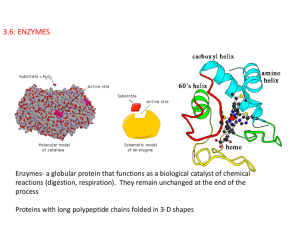
3.6: ENZYMES
... contain many +/- regions, some around the active site. An excess of H+ ions in an acidic solution can lead to bonding between the H+ ions and the negative charges in the active site. (same with OH- in basic solutions where the OH- ions bond to the positive sites). This will inhibit the matching proc ...
... contain many +/- regions, some around the active site. An excess of H+ ions in an acidic solution can lead to bonding between the H+ ions and the negative charges in the active site. (same with OH- in basic solutions where the OH- ions bond to the positive sites). This will inhibit the matching proc ...
Simple kinetics of enzyme action
... specificity of a fixed amount of the enzyme for its substrate (more precisely defined in terms of kcat/K m). The substrate concentration, which gives a rate of half the maximum reaction velocity, is equal to the K m. ...
... specificity of a fixed amount of the enzyme for its substrate (more precisely defined in terms of kcat/K m). The substrate concentration, which gives a rate of half the maximum reaction velocity, is equal to the K m. ...
Enzymes II – How Enzymes Work
... chances of them colliding such that the carboxyl group of one amino acid comes into contact with the amino group of the other amino acid is very slim. If the proper enzyme is present, however, the amino acids may bind to the active site of the enzyme such that their amino and carboxyl groups are hel ...
... chances of them colliding such that the carboxyl group of one amino acid comes into contact with the amino group of the other amino acid is very slim. If the proper enzyme is present, however, the amino acids may bind to the active site of the enzyme such that their amino and carboxyl groups are hel ...
enzyme kinetics
... enzyme. At very low substrate concentrations, where [S] << Km, most of the enzyme is free. Thus we can think of [E] = [Eo], so that the Michaelis–Menten equation becomes V = kcat/Km [E][S]. Thus, the ratio kcat/Km is equivalent to the rate constant for the reaction between free enzyme and free subst ...
... enzyme. At very low substrate concentrations, where [S] << Km, most of the enzyme is free. Thus we can think of [E] = [Eo], so that the Michaelis–Menten equation becomes V = kcat/Km [E][S]. Thus, the ratio kcat/Km is equivalent to the rate constant for the reaction between free enzyme and free subst ...
metabolism - Chavis Biology
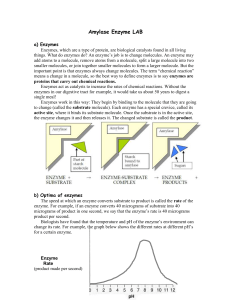
... increase in the reaction rate. Reaction rate will eventually level off as all available substrates are used up. Draw this: ...
... increase in the reaction rate. Reaction rate will eventually level off as all available substrates are used up. Draw this: ...
Enzymology Part 2
... PABA is required for the synthesis of folic acid Sulphanamida is a drug that competes with PABA for the enzymes in the folic acid synthesis pathway. ...
... PABA is required for the synthesis of folic acid Sulphanamida is a drug that competes with PABA for the enzymes in the folic acid synthesis pathway. ...
... In this Appendix there are 3 art@ies. These articles provide a basic understanding of the mechanism of how HCA works. The first article is by John Lowenstein of Brandeis University who was one of the key figures in the early research conductad on HCA. The article entitled “Experiments with Hydroxyci ...
1. What are enzymes? Be able to describe the chemical nature of
... Optimum conditions vary slightly for each enzyme but are generally near normal body temperature and the pH of the body fluid in which the enzyme functions. ...
... Optimum conditions vary slightly for each enzyme but are generally near normal body temperature and the pH of the body fluid in which the enzyme functions. ...
Enzyme Kinetics
... Incubation of reaction mix including Supersomes TM (human recombinant enzymes), test compound (multiple concentrations, usually 8 to 10), and NADPH for a single time points, e.g. 60 minutes. ...
... Incubation of reaction mix including Supersomes TM (human recombinant enzymes), test compound (multiple concentrations, usually 8 to 10), and NADPH for a single time points, e.g. 60 minutes. ...
Enzyme Kinetics
... elicits a conformational change. As a result, the apparent Vmax decreases and the apparent Km may increase or decrease. – When the Km is not affected by the inhibitor, it is said as non competitive. ...
... elicits a conformational change. As a result, the apparent Vmax decreases and the apparent Km may increase or decrease. – When the Km is not affected by the inhibitor, it is said as non competitive. ...
Chem452_Quiz_2
... b. The distal histidine, while not one of the ligands for the Fe2+ ion, nonetheless plays some important roles with respect to oxygen binding by hemoglobin. Describe two of these. c. Using the axes provided below, illustrate how the binding of oxygen to myoglobin differs from that for hemoglobin. Dr ...
... b. The distal histidine, while not one of the ligands for the Fe2+ ion, nonetheless plays some important roles with respect to oxygen binding by hemoglobin. Describe two of these. c. Using the axes provided below, illustrate how the binding of oxygen to myoglobin differs from that for hemoglobin. Dr ...
Physiology Lecture Outline: Enzymes
... as a result. They are like reactants in a spontaneous chemical reaction (but are called substrates when enzymes are involved) and they are transformed into products. All enzymes are proteins with specific 3-D shapes and they contain small regions where substrates bind to the enzyme called the active ...
... as a result. They are like reactants in a spontaneous chemical reaction (but are called substrates when enzymes are involved) and they are transformed into products. All enzymes are proteins with specific 3-D shapes and they contain small regions where substrates bind to the enzyme called the active ...
White Cell Enzymes
... Won’t detect Mucolipidoses, and other lysosomal storage disorders, that may present with similar clinical presentation Not reliable in babies <3months (high excretion due to rapid growth & tissue turnover) or dilute urine samples (creatinine <1.0mmol/L – additive errors in calculating ratios. How re ...
... Won’t detect Mucolipidoses, and other lysosomal storage disorders, that may present with similar clinical presentation Not reliable in babies <3months (high excretion due to rapid growth & tissue turnover) or dilute urine samples (creatinine <1.0mmol/L – additive errors in calculating ratios. How re ...
Pre-‐lab AP Lab: Enzyme Catalysis Name
... such as that of human blood (0.9%) or cytoplasm, is the optimum for many enzymes. 2. pH. pH is a logarithmic scale that measures the acidity, or H+ concentration, in a solution. The scale runs from 0 to 14 with 0 being the highest in acidity and 14 the lowest. When the pH is in the range of 0-7, a s ...
... such as that of human blood (0.9%) or cytoplasm, is the optimum for many enzymes. 2. pH. pH is a logarithmic scale that measures the acidity, or H+ concentration, in a solution. The scale runs from 0 to 14 with 0 being the highest in acidity and 14 the lowest. When the pH is in the range of 0-7, a s ...
File
... Read: biochemical reactions usually require more than one enzyme. This is a series of enzymes functioning in a sequence to produce a final product. ...
... Read: biochemical reactions usually require more than one enzyme. This is a series of enzymes functioning in a sequence to produce a final product. ...
Document
... Why does the enzyme rate decrease when its temperature is below its temperature optimum? You might think at first that the enzyme is denatured, but that is not correct. The reason for the rate decrease is that molecules move slower at colder temperatures. You can think of the enzyme molecule and its ...
... Why does the enzyme rate decrease when its temperature is below its temperature optimum? You might think at first that the enzyme is denatured, but that is not correct. The reason for the rate decrease is that molecules move slower at colder temperatures. You can think of the enzyme molecule and its ...
Enzymes: the little molecules that could
... bonded, the active site of the enzyme is positioned near the reaction site of the substrate, which it alters to reduce the energy barrier. In this example, the substrate is a protein. are chains of amino acids linked by peptide bonds. ...
... bonded, the active site of the enzyme is positioned near the reaction site of the substrate, which it alters to reduce the energy barrier. In this example, the substrate is a protein. are chains of amino acids linked by peptide bonds. ...
3+7 – HL Enzymes Page 1 1. Structure of Enzymes Like all proteins
... and thus reducing the activation energy. When a substrate collides with an enzyme and enters the active site, the substrate is stressed in a way that allows its bonds to break easier. Enzymes can process reactions such as this very rapidly. Some enzymes can have quite broad specificity, e.g. some pr ...
... and thus reducing the activation energy. When a substrate collides with an enzyme and enters the active site, the substrate is stressed in a way that allows its bonds to break easier. Enzymes can process reactions such as this very rapidly. Some enzymes can have quite broad specificity, e.g. some pr ...
CHAPTER 4: Enzyme Structure
... site. It binds to another part of the enzyme molecule, changing the shape of the whole enzyme, including the active site, so that it can no longer bind substrate molecules. Non-competitive inhibitors therefore simply reduce the amount of active enzyme. This is the same as decreasing the enzyme conce ...
... site. It binds to another part of the enzyme molecule, changing the shape of the whole enzyme, including the active site, so that it can no longer bind substrate molecules. Non-competitive inhibitors therefore simply reduce the amount of active enzyme. This is the same as decreasing the enzyme conce ...