Exploration Activity: Enzymes
... Graph (a) shows that this enzyme will work best at what type of temperatures? Graph (b) shows that this enzyme will work best at what type of temperatures? Graph (c) shows that this enzyme will work best at what type of pH? Graph (d) shows that this enzyme will work best at what type of pH? ...
... Graph (a) shows that this enzyme will work best at what type of temperatures? Graph (b) shows that this enzyme will work best at what type of temperatures? Graph (c) shows that this enzyme will work best at what type of pH? Graph (d) shows that this enzyme will work best at what type of pH? ...
ETA Position Paper Biotechnology, Enzymes and Allergies
... all living cells produce and need enzymes to live and grow. However, enzymes themselves are not living organisms. Enzymes are catalysts: substances which speed up chemical reactions by being present in very small amounts and without being changed in the reaction. What is an allergy? The purpose of t ...
... all living cells produce and need enzymes to live and grow. However, enzymes themselves are not living organisms. Enzymes are catalysts: substances which speed up chemical reactions by being present in very small amounts and without being changed in the reaction. What is an allergy? The purpose of t ...
Catalytic mechanism of the inverting N
... Experimental data on the mechanism of GnT-I indicate an ordered sequential mechanism (Nishikawa et al., 1988) in which the enzyme first binds to both the metal cofactor and UDP-GlcNAc and subsequently to the Man5GlcNAc2Asn-X oligosaccharide acceptor. The oligosaccharide product, GlcNAcMan5GlcNAc2-As ...
... Experimental data on the mechanism of GnT-I indicate an ordered sequential mechanism (Nishikawa et al., 1988) in which the enzyme first binds to both the metal cofactor and UDP-GlcNAc and subsequently to the Man5GlcNAc2Asn-X oligosaccharide acceptor. The oligosaccharide product, GlcNAcMan5GlcNAc2-As ...
Enzyme-Catalyzed Reactions Lab
... Part III: Does acid destroy an enzyme’s ability to speed up a chemical reaction? Hypothesis: If acid is added to catalase, then fewer bubbles of O2 will be produced. IV (manipulated)= ___________________________ DV (measured) = ______________________________ Materials: dropper, liver, 2 test tubes, ...
... Part III: Does acid destroy an enzyme’s ability to speed up a chemical reaction? Hypothesis: If acid is added to catalase, then fewer bubbles of O2 will be produced. IV (manipulated)= ___________________________ DV (measured) = ______________________________ Materials: dropper, liver, 2 test tubes, ...
Adding Enzymes To Dairy Diets
... during pelleting) or digestion (e.g., contact with proteases), enzyme activity will diminish and the enzyme will not be as effective at digesting feed. Stability of enzyme activity in adverse environmental conditions differs widely among enzymes. Commercial enzyme products marketed as feed suppleme ...
... during pelleting) or digestion (e.g., contact with proteases), enzyme activity will diminish and the enzyme will not be as effective at digesting feed. Stability of enzyme activity in adverse environmental conditions differs widely among enzymes. Commercial enzyme products marketed as feed suppleme ...
Chapter 12. Strategies for metabolic control and their application to
... are in a high affinity or a low affinity state, with the balance lying to the low affinity side. Binding of a substrate molecule tips the equilibrium to the high affinity side. Allosteric modifiers alter the equilibrium between the low affinity (T or tense) and high affinity states (R or relaxed), ...
... are in a high affinity or a low affinity state, with the balance lying to the low affinity side. Binding of a substrate molecule tips the equilibrium to the high affinity side. Allosteric modifiers alter the equilibrium between the low affinity (T or tense) and high affinity states (R or relaxed), ...
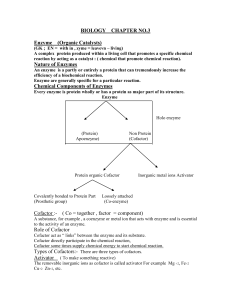
Nature of Enzymes
... Heat increase Kinetic energy of reactions, this increases the chances of their effective collision with each other and with enzyme. Above optimum, further supply of heat energy increases the vibrations of atom making enzyme molecules. Such strong vibrations destroy globular structure of enzymes. Enz ...
... Heat increase Kinetic energy of reactions, this increases the chances of their effective collision with each other and with enzyme. Above optimum, further supply of heat energy increases the vibrations of atom making enzyme molecules. Such strong vibrations destroy globular structure of enzymes. Enz ...
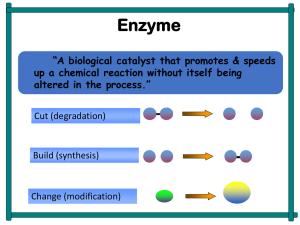
Enzymes: The Biological Catalysts of Life
... information, the transport of compounds across the membranes, motility of organisms and conversion of chemical energy. Enzyme catalysis is essential for making biochemical reactions proceed at appropriate speed in physiological conditions. They speed up the reactions in the cells so that they may oc ...
... information, the transport of compounds across the membranes, motility of organisms and conversion of chemical energy. Enzyme catalysis is essential for making biochemical reactions proceed at appropriate speed in physiological conditions. They speed up the reactions in the cells so that they may oc ...
Enzyme immobilization
... • When it is added to a detergent, it results into the following effects: • Colour brightening-When garments made of cotton or cotton blends have been washed several times, they tend to get a 'fluffy' look and the colours become duller. • This effect is due to the formation of microfibrils that beco ...
... • When it is added to a detergent, it results into the following effects: • Colour brightening-When garments made of cotton or cotton blends have been washed several times, they tend to get a 'fluffy' look and the colours become duller. • This effect is due to the formation of microfibrils that beco ...
Lecture 11 Enzymes: Kinetics
... If KM = KES, which enzyme above binds its substrate the most tightly? Which enzyme has the most rapid catalytic turnover when the enzyme is saturated with substrate? Which enzymes have the highest catalytic efficiency? Are they near the limit of diffusion control? NOTE: kcat/KM for an enzyme can hav ...
... If KM = KES, which enzyme above binds its substrate the most tightly? Which enzyme has the most rapid catalytic turnover when the enzyme is saturated with substrate? Which enzymes have the highest catalytic efficiency? Are they near the limit of diffusion control? NOTE: kcat/KM for an enzyme can hav ...
Enzymes
... • Describe how changing temperature and pH will change the rate of reaction of an enzyme-catalysed reaction (C) • Explain the specificity of enzymes in terms of the ‘lock and key’ mechanism (B) • Explain how enzyme activity is affected by pH and temperature (A) • Link the effect of temperature and p ...
... • Describe how changing temperature and pH will change the rate of reaction of an enzyme-catalysed reaction (C) • Explain the specificity of enzymes in terms of the ‘lock and key’ mechanism (B) • Explain how enzyme activity is affected by pH and temperature (A) • Link the effect of temperature and p ...
Enzymes 1. All cells in multicellular organisms contain thousands of
... Enzymes help catalyze, or speed up, these chemical reactions by lowering the amount of activation energy needed for the reactions to proceed. Reactions that do not use an enzyme require more energy. 12. According to the chemical equation, water and oxygen are produced when hydrogen peroxide is broke ...
... Enzymes help catalyze, or speed up, these chemical reactions by lowering the amount of activation energy needed for the reactions to proceed. Reactions that do not use an enzyme require more energy. 12. According to the chemical equation, water and oxygen are produced when hydrogen peroxide is broke ...
Enzyme Mechanisms Serine proteases - Rose
... The serine protease mechanism does, however, involve stabilization of the transition state. This has been observed in mutant studies in which all of the catalytic triad residues were mutated. Although the reaction mechanism might be interpreted to predict that the loss of the active site serine wou ...
... The serine protease mechanism does, however, involve stabilization of the transition state. This has been observed in mutant studies in which all of the catalytic triad residues were mutated. Although the reaction mechanism might be interpreted to predict that the loss of the active site serine wou ...
Enzyme Lab - marric.us
... .....Note that the enzyme is not changed in the reaction and can be recycled to break down additional substrate molecules. ..Each enzyme is specific for a particular reaction because its amino acid sequence is unique and causes it to have a unique three-dimensional structure. ..The active site is th ...
... .....Note that the enzyme is not changed in the reaction and can be recycled to break down additional substrate molecules. ..Each enzyme is specific for a particular reaction because its amino acid sequence is unique and causes it to have a unique three-dimensional structure. ..The active site is th ...
Enzymes - fblocks
... Km (Michaelis Constant) of an enzyme is numerically equal to the substrate concentration at which the velocity of reaction is equal to 1/2 Vmax Km is the substrate concentration at which 1/2 maximal velocity is reached If Km is small, the substrate concentration required for the reaction to reach 1/ ...
... Km (Michaelis Constant) of an enzyme is numerically equal to the substrate concentration at which the velocity of reaction is equal to 1/2 Vmax Km is the substrate concentration at which 1/2 maximal velocity is reached If Km is small, the substrate concentration required for the reaction to reach 1/ ...
Name
... and 1,000 amino acids in a very specific and unique order. The chain of amino acids then folds into a unique shape. That shape allows the enzyme to carry out specific chemical reactions -- an enzyme acts as a very efficient catalyst for a specific chemical reaction. For example, the sugar maltose is ...
... and 1,000 amino acids in a very specific and unique order. The chain of amino acids then folds into a unique shape. That shape allows the enzyme to carry out specific chemical reactions -- an enzyme acts as a very efficient catalyst for a specific chemical reaction. For example, the sugar maltose is ...
Enzyme Inhibition
... This enzyme an active site for fructose-6-phosphate molecules to bind with another phosphate group It has an allosteric site for ATP molecules, the inhibitor When the cell consumes a lot of ATP the level of ATP in the cell falls No ATP binds to the allosteric site of ...
... This enzyme an active site for fructose-6-phosphate molecules to bind with another phosphate group It has an allosteric site for ATP molecules, the inhibitor When the cell consumes a lot of ATP the level of ATP in the cell falls No ATP binds to the allosteric site of ...
Lecture 2: Enzymes
... In general, a protein is a chain of amino acids (aa) covalently linked (when the chain is short ~5-10 aa, the protein is often called oligopeptide/polypeptide or simply peptide) ...
... In general, a protein is a chain of amino acids (aa) covalently linked (when the chain is short ~5-10 aa, the protein is often called oligopeptide/polypeptide or simply peptide) ...
Experiment 7 (Lab Period 8) Quantitative Determination of
... particulate matter captured during phagocytosis. Still others are active in the cytoplasm and serve to recycle phosphorus in metabolism or to remove phosphate groups from proteins whose activities are regulated by the addition and removal of phosphate groups. Many phosphatases are not very specific ...
... particulate matter captured during phagocytosis. Still others are active in the cytoplasm and serve to recycle phosphorus in metabolism or to remove phosphate groups from proteins whose activities are regulated by the addition and removal of phosphate groups. Many phosphatases are not very specific ...
Biochemistry Note Packet
... 5) Look at the structural formulas below. These three sugars all have the same chemical formula (C6H12O6). a. Are the structural formulas the same? __________________ b. What do we call compounds that have the same chemical formula, but differ in their structural formulas? _______________________ ...
... 5) Look at the structural formulas below. These three sugars all have the same chemical formula (C6H12O6). a. Are the structural formulas the same? __________________ b. What do we call compounds that have the same chemical formula, but differ in their structural formulas? _______________________ ...
Concept review: Enzyme kinetics
... change the apparent Km. • A noncompetitive inhibitor binds at a different site on the free enzyme and changes the shape of the active site such that the reaction proceeds at a reduced rate. No matter how much substrate is present, the inhibitor will still be able to bind to the enzyme. Accordingly, ...
... change the apparent Km. • A noncompetitive inhibitor binds at a different site on the free enzyme and changes the shape of the active site such that the reaction proceeds at a reduced rate. No matter how much substrate is present, the inhibitor will still be able to bind to the enzyme. Accordingly, ...
Isomerase

Isomerases are a general class of enzymes which convert a molecule from one isomer to another. Isomerases can either facilitate intramolecular rearrangements in which bonds are broken and formed or they can catalyze conformational changes. The general form of such a reaction is as follows:A–B → B–AThere is only one substrate yielding one product. This product has the same molecular formula as the substrate but differs in bond connectivity or spatial arrangements. Isomerases catalyze reactions across many biological processes, such as in glycolysis and carbohydrate metabolism.