Enzymes - Healing Energies at London West
... enzyme lactase breaks down the lactose sugar in milk. The enzyme maltase breaks down the malt sugar maltose. Protease enzymes are found in the juices of the pancreas, the stomach and the intestinal tract and help with the breakdown and the digestion of proteins. Lipase enzymes are found in the j ...
... enzyme lactase breaks down the lactose sugar in milk. The enzyme maltase breaks down the malt sugar maltose. Protease enzymes are found in the juices of the pancreas, the stomach and the intestinal tract and help with the breakdown and the digestion of proteins. Lipase enzymes are found in the j ...
Name: Date: ______ Block: ______ ENZYMES A CATALYST is a
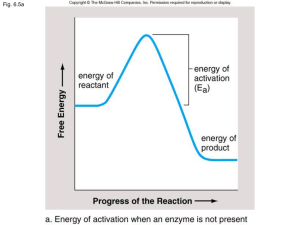
... enzyme causes some of the bonds in the substrate to weaken – lessening the activation energy needed to break the bonds and start the reaction. As with the lock and key method, the enzyme returns to its original shape after releasing the products and can be used again. ...
... enzyme causes some of the bonds in the substrate to weaken – lessening the activation energy needed to break the bonds and start the reaction. As with the lock and key method, the enzyme returns to its original shape after releasing the products and can be used again. ...
AP Laboratory Investigation Lab 13 – Enzyme Activity 2H2O2
... 1. Put a piece of liver into the bottom of a clean test tube and cover it with a small amount of water. Place this test tube in a boiling water bath for 5 minutes. Remove the test tube ...
... 1. Put a piece of liver into the bottom of a clean test tube and cover it with a small amount of water. Place this test tube in a boiling water bath for 5 minutes. Remove the test tube ...
Pdf - Text of NPTEL IIT Video Lectures
... Also talking to reversible enzyme inhibition now let us discusses irreversible enzyme inhibition. Inhibitor is very tightly bound to enzyme either covalently or non-covalently in such a way that this inhibition is irreversible. Let us take the very classical example penicillin the first anti biotech ...
... Also talking to reversible enzyme inhibition now let us discusses irreversible enzyme inhibition. Inhibitor is very tightly bound to enzyme either covalently or non-covalently in such a way that this inhibition is irreversible. Let us take the very classical example penicillin the first anti biotech ...
Chapter-4 Photosynthesis in Higher Plants
... This pathway also named as Hatch and Slack Pathway, is again a cyclic process. The primary CO2 acceptor is a 3-carbon molecule phosphoenol pyruvate (PEP) and is present in the mesophyll cells. The enzyme responsible for this fixation is PEP carboxylase or PEPcase. Mesophyll cells lack RuBisCO enzyme ...
... This pathway also named as Hatch and Slack Pathway, is again a cyclic process. The primary CO2 acceptor is a 3-carbon molecule phosphoenol pyruvate (PEP) and is present in the mesophyll cells. The enzyme responsible for this fixation is PEP carboxylase or PEPcase. Mesophyll cells lack RuBisCO enzyme ...
Enzymes and Substrates – Penny Lab 2015
... In class we discussed what would happen to the reaction rate if we increased the substrate or enzyme amounts. We also saw a graph of those results. In this lab, we will use pennies to make a working model that shows why the substrate and enzyme concentrations affect the reaction rate. Listen for cha ...
... In class we discussed what would happen to the reaction rate if we increased the substrate or enzyme amounts. We also saw a graph of those results. In this lab, we will use pennies to make a working model that shows why the substrate and enzyme concentrations affect the reaction rate. Listen for cha ...
How to write an effective conclusion
... It was predicted that as the temperature increased from 0°C, the rate of enzyme reaction will increased up until the optimum temperature for this enzyme. This is the temperature at which the enzyme will be working with maximum efficiency. After this point the rate of reaction will begin to decrease ...
... It was predicted that as the temperature increased from 0°C, the rate of enzyme reaction will increased up until the optimum temperature for this enzyme. This is the temperature at which the enzyme will be working with maximum efficiency. After this point the rate of reaction will begin to decrease ...
AP Biology
... At your lab bench you will find a round vial with a rubber stopper that fits in the top. This will be referred to as the reaction chamber. You will also find a eudiometer (a gas collection tube), a ring stand and clamp, and a plastic pan which will be used as a water bath. All of the equipment needs ...
... At your lab bench you will find a round vial with a rubber stopper that fits in the top. This will be referred to as the reaction chamber. You will also find a eudiometer (a gas collection tube), a ring stand and clamp, and a plastic pan which will be used as a water bath. All of the equipment needs ...
Enzymes Notes #2- Enzyme Substrate Complexes and Factors
... Enzymes are specific for one particular reaction or group of related reactions. Many reactions cannot occur without the correct enzyme present. They are often named by adding “ASE" to the name of the substrate. Example: Dehydrogenases are enzymes that remove hydrogen. ...
... Enzymes are specific for one particular reaction or group of related reactions. Many reactions cannot occur without the correct enzyme present. They are often named by adding “ASE" to the name of the substrate. Example: Dehydrogenases are enzymes that remove hydrogen. ...
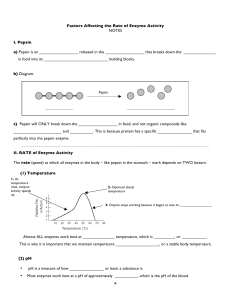
8 Factors Affecting the Rate of Enzyme Activity NOTES I. Pepsin a
... Which statement best expresses the amount of product that will be formed at each temperature if the experiment is repeated at a pH of 4? (1) The amount of product formed will be equal to that produced at pH 6. (2) The amount of product formed will be greater than that produced at pH 6. (3) The amoun ...
... Which statement best expresses the amount of product that will be formed at each temperature if the experiment is repeated at a pH of 4? (1) The amount of product formed will be equal to that produced at pH 6. (2) The amount of product formed will be greater than that produced at pH 6. (3) The amoun ...
Enzymes: The Spit Lab
... 1. Cell Biology: The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism's cells. 1b. Students know enzymes are proteins that catalyze biochemical reactions with altering the reaction equilibrium and the activitie ...
... 1. Cell Biology: The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism's cells. 1b. Students know enzymes are proteins that catalyze biochemical reactions with altering the reaction equilibrium and the activitie ...
A metal enzyme that can cleave benzene rings
... The enzyme uses activated benzoic acid benzoyl-CoA with an aromatic ring as substrate. It is an important metabolic intermediate in bacteria that degrade monocyclic aromatic compounds. Boll's enzyme occurs in two conformations, closed involving zinc, and open involving benzoylCoA. Boll discovered th ...
... The enzyme uses activated benzoic acid benzoyl-CoA with an aromatic ring as substrate. It is an important metabolic intermediate in bacteria that degrade monocyclic aromatic compounds. Boll's enzyme occurs in two conformations, closed involving zinc, and open involving benzoylCoA. Boll discovered th ...
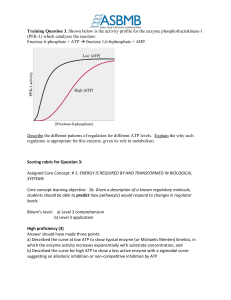
Training Question 3: Rubric
... Core concept learning objective: 1b. Given a description of a known regulatory molecule, students should be able to predict how pathway(s) would respond to changes in regulator levels. Bloom’s level: a) Level 2 comprehension b) Level 3 application High proficiency (3) Answer should have made three p ...
... Core concept learning objective: 1b. Given a description of a known regulatory molecule, students should be able to predict how pathway(s) would respond to changes in regulator levels. Bloom’s level: a) Level 2 comprehension b) Level 3 application High proficiency (3) Answer should have made three p ...
Enzyme
... • It is incorrect to say low pH denatures enzymes—as this is not true in the stomach • Each enzyme has its own optimum pH • Below or above the optimum, the enzyme will alter its’ shape, and therefore its’ ability to function • The further away from optimum, the lower the ability to function ...
... • It is incorrect to say low pH denatures enzymes—as this is not true in the stomach • Each enzyme has its own optimum pH • Below or above the optimum, the enzyme will alter its’ shape, and therefore its’ ability to function • The further away from optimum, the lower the ability to function ...
lecture notes-enzyme-web
... - the binding force between enzyme and carrier is so strong that no leakage of the enzymes occurs. ...
... - the binding force between enzyme and carrier is so strong that no leakage of the enzymes occurs. ...
which pennies are turned over by pennyase?
... Name: ___________________________________________ Date: ___________ Pd: _____ ...
... Name: ___________________________________________ Date: ___________ Pd: _____ ...
Datasheet - Sigma
... catalyze the rapid conversion of carbon dioxide to bicarbonate and protons, a reaction that occurs rather slowly in the absence of a catalyst. The active site of most carbonic anhydrases contains a zinc ion. They are, therefore, classified as metalloenzymes.1-2 Carbonic anhydrases are widely distrib ...
... catalyze the rapid conversion of carbon dioxide to bicarbonate and protons, a reaction that occurs rather slowly in the absence of a catalyst. The active site of most carbonic anhydrases contains a zinc ion. They are, therefore, classified as metalloenzymes.1-2 Carbonic anhydrases are widely distrib ...
Introduction - Evergreen Archives
... This tendency for disorder to increase gives a directionality to physical and chemical processes, explaining why some reactions proceed in one direction rather than another. (See Figure 6.2b.) ...
... This tendency for disorder to increase gives a directionality to physical and chemical processes, explaining why some reactions proceed in one direction rather than another. (See Figure 6.2b.) ...
Introduction - Cedar Crest College
... This tendency for disorder to increase gives a directionality to physical and chemical processes, explaining why some reactions proceed in one direction rather than another. (See Figure 6.2b.) ...
... This tendency for disorder to increase gives a directionality to physical and chemical processes, explaining why some reactions proceed in one direction rather than another. (See Figure 6.2b.) ...
Investigating Catalase - churchillcollegebiblio
... • How will you collect your raw data? -make sure you have table drawn with sensible headings and units • How will you present your data? -what kind of graph would be appropriate? ...
... • How will you collect your raw data? -make sure you have table drawn with sensible headings and units • How will you present your data? -what kind of graph would be appropriate? ...
Single Molecule Approach to Enzymology
... where m is the index number for the turnovers and ∆τ(m) = τ(m) - <τ>, with the bracket denoting the average along the trajectory. The physical meaning of C(m) is as follows: In the absence of dynamic disorder, C(0) = 1 and C(m) = 0 (m > 0). This holds for not only a one step Poisson process (k1 or k ...
... where m is the index number for the turnovers and ∆τ(m) = τ(m) - <τ>, with the bracket denoting the average along the trajectory. The physical meaning of C(m) is as follows: In the absence of dynamic disorder, C(0) = 1 and C(m) = 0 (m > 0). This holds for not only a one step Poisson process (k1 or k ...
Pinpointing dynamic coupling in enzymes for efficient drug design
... hybrids of DHFR were prepared by chemical ligation techniques [20] , one in which the mobile N-terminal segment contained heavy isotopes while the remainder of the protein was of natural isotopic abundance, and one in which only the C-terminal region was isotopically labeled. These experiments reve ...
... hybrids of DHFR were prepared by chemical ligation techniques [20] , one in which the mobile N-terminal segment contained heavy isotopes while the remainder of the protein was of natural isotopic abundance, and one in which only the C-terminal region was isotopically labeled. These experiments reve ...
World record enzymes
... One vital class of proteins is enzymes, which are catalysts, i.e. they speed up chemical reactions without being consumed in the process. Without them, many reactions essential for life would be far too slow for life to exist. Catalysts do not affect the equilibrium of reactions, only the rate at wh ...
... One vital class of proteins is enzymes, which are catalysts, i.e. they speed up chemical reactions without being consumed in the process. Without them, many reactions essential for life would be far too slow for life to exist. Catalysts do not affect the equilibrium of reactions, only the rate at wh ...
Disaccharides - Home - KSU Faculty Member websites
... pressure. It swells in water to produce a viscous, colloidal solution. It is used to increase the viscosity and to stabilize lotions, suspensions, pastes, and ointments. In ophthalmic preparations as protectant. It is also used bulk laxative in chronic constipation and in treatment of obesity ...
... pressure. It swells in water to produce a viscous, colloidal solution. It is used to increase the viscosity and to stabilize lotions, suspensions, pastes, and ointments. In ophthalmic preparations as protectant. It is also used bulk laxative in chronic constipation and in treatment of obesity ...
Isomerase

Isomerases are a general class of enzymes which convert a molecule from one isomer to another. Isomerases can either facilitate intramolecular rearrangements in which bonds are broken and formed or they can catalyze conformational changes. The general form of such a reaction is as follows:A–B → B–AThere is only one substrate yielding one product. This product has the same molecular formula as the substrate but differs in bond connectivity or spatial arrangements. Isomerases catalyze reactions across many biological processes, such as in glycolysis and carbohydrate metabolism.