Chemistry: the study of composition, structure, and properties of
... Chemistry: the study of composition, structure, and properties of matter* and the changes it undergoes. *Matter: composed of mass and takes up space. ...
... Chemistry: the study of composition, structure, and properties of matter* and the changes it undergoes. *Matter: composed of mass and takes up space. ...
Things to Know to Pass the Chemistry Regents
... 9. Atoms are neutral because # of protons(+) = # of electrons(-) 10. Isotopes are atoms with same # of protons and electrons, but different number of neutrons *therefore also have different mass numbers 11. Metal atoms lose e- to form (+) ions smaller than atom 12. Nonmetals gain e- to form (-) ions ...
... 9. Atoms are neutral because # of protons(+) = # of electrons(-) 10. Isotopes are atoms with same # of protons and electrons, but different number of neutrons *therefore also have different mass numbers 11. Metal atoms lose e- to form (+) ions smaller than atom 12. Nonmetals gain e- to form (-) ions ...
Notes matter energy
... Solution: Write the percentages as fractions, then form the algebraic sum from the isotope compositions and fractions: 6*0.075 + 7*0.925 = 0.45 + 6.475 = 6.925 = 6.93. (The products have 2SD and 3SD because 6 and 7 are counted (exact). The sum has 3SD because the answer is known to the hundredths pl ...
... Solution: Write the percentages as fractions, then form the algebraic sum from the isotope compositions and fractions: 6*0.075 + 7*0.925 = 0.45 + 6.475 = 6.925 = 6.93. (The products have 2SD and 3SD because 6 and 7 are counted (exact). The sum has 3SD because the answer is known to the hundredths pl ...
PHY 1020 SI PRACTICE FINAL EXAM
... a) an electron jumps from a higher energy level to a lower one b) a proton jumps from a higher energy level to a lower one c) an electron jumps from a lower energy level to a higher one d) a proton jumps from a lower energy level to a higher one (16) Of the three fundamental particles that make up a ...
... a) an electron jumps from a higher energy level to a lower one b) a proton jumps from a higher energy level to a lower one c) an electron jumps from a lower energy level to a higher one d) a proton jumps from a lower energy level to a higher one (16) Of the three fundamental particles that make up a ...
File
... Do Now Nuclear Chemistry 17. Which equation represents a transmutation reaction? A) B) C) D) 18. A change in the nucleus of an atom that converts the atom from one element to another element is called A) combustion C) polymerization ...
... Do Now Nuclear Chemistry 17. Which equation represents a transmutation reaction? A) B) C) D) 18. A change in the nucleus of an atom that converts the atom from one element to another element is called A) combustion C) polymerization ...
-30- Section 9: f"
... -31ψ = nasty looking function which includes E (energy) and n, l and ml , constants analogous to nx, ny and nz of square well. Boundary conditions restrict these numbers to values I will put on board. "Subshells" are sometimes lettered: s state means l = 0, p state means l = 1, d state means l = 2, ...
... -31ψ = nasty looking function which includes E (energy) and n, l and ml , constants analogous to nx, ny and nz of square well. Boundary conditions restrict these numbers to values I will put on board. "Subshells" are sometimes lettered: s state means l = 0, p state means l = 1, d state means l = 2, ...
Materials Science for Chemical Engineers
... one electron is places in all orbitals of equal energy before two electrons are placed in any one of these orbitals. Rule 3. Pauli Exclusion principle a maximum of two electrons can occupy an orbital. No two electrons can have the same four quantum numbers. ...
... one electron is places in all orbitals of equal energy before two electrons are placed in any one of these orbitals. Rule 3. Pauli Exclusion principle a maximum of two electrons can occupy an orbital. No two electrons can have the same four quantum numbers. ...
Unit 2 Review: Chemistry - Mr. Hoover's Science Classes
... types of matter. Atomic theory helps us to understand why there are different kinds of atoms. It explains how atoms combine to form over 100 known elements and all other forms of matter, including compounds and ...
... types of matter. Atomic theory helps us to understand why there are different kinds of atoms. It explains how atoms combine to form over 100 known elements and all other forms of matter, including compounds and ...
The buoyant force on an object totally submerged in a fluid depends
... accept or that we will reject. We are willing to receive all truth, from whatever source it may come; for truth will stand, truth will endure." -- Joseph F. Smith ...
... accept or that we will reject. We are willing to receive all truth, from whatever source it may come; for truth will stand, truth will endure." -- Joseph F. Smith ...
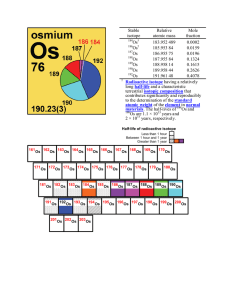
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... positron – the antimatter counterpart of the electron, with a mass identical to that of the electron and an equal but opposite (positive) charge. proton – an elementary particle having a rest mass of about 1.673 × 10–27 kg, slightly less than that of a neutron, and a positive electric charge equal a ...
... positron – the antimatter counterpart of the electron, with a mass identical to that of the electron and an equal but opposite (positive) charge. proton – an elementary particle having a rest mass of about 1.673 × 10–27 kg, slightly less than that of a neutron, and a positive electric charge equal a ...
Review 3rd Qtr KEY
... Given calculations with the calculator answer, write the answers with the appropriate # of significant figures. ...
... Given calculations with the calculator answer, write the answers with the appropriate # of significant figures. ...
Chemistry I Exams and Keys 2014 Season
... Choose the answer that best completes the statements or questions below and fill in the appropriate response on the form. If you change an answer, be sure to completely erase your first choice. You may use the given periodic table and formula sheet as well as a calculator. On the formula sheets is a ...
... Choose the answer that best completes the statements or questions below and fill in the appropriate response on the form. If you change an answer, be sure to completely erase your first choice. You may use the given periodic table and formula sheet as well as a calculator. On the formula sheets is a ...
Atomic Structure and Periodicity
... 14. Which group possesses the lowest first ionization energy in their respective period? For 15 – 17: a. Bohr model b. deBroglie’s wave hypothesis c. Heisenberg’s uncertainty principle d. Quantum theory e. Atomic theory 15. Which principle provides that all matter may be considered a wave? 16. What ...
... 14. Which group possesses the lowest first ionization energy in their respective period? For 15 – 17: a. Bohr model b. deBroglie’s wave hypothesis c. Heisenberg’s uncertainty principle d. Quantum theory e. Atomic theory 15. Which principle provides that all matter may be considered a wave? 16. What ...
Planck`s Constant and the Photon
... The sketch below shows a Hydrogen atom as per the Space Vortex Theory (SVT). Here the shaded core is the assembly of electrons and positrons, thus building a neutron. A space vortex enclosing the neutron, shown by the circular streamlines provides charge and makes it a proton. The adjoining electron ...
... The sketch below shows a Hydrogen atom as per the Space Vortex Theory (SVT). Here the shaded core is the assembly of electrons and positrons, thus building a neutron. A space vortex enclosing the neutron, shown by the circular streamlines provides charge and makes it a proton. The adjoining electron ...
Correlation of Aqueous Redox Potentials with Gaseous Ionization
... were discovered for every group of elements of the periodic table.1 This linearity was then used to estimate the hitherto unknown ionization potentials of actinide elements from their aqueous polarographic half-wave potentials.2 However, in these correlations, the ionization potentials (which are ge ...
... were discovered for every group of elements of the periodic table.1 This linearity was then used to estimate the hitherto unknown ionization potentials of actinide elements from their aqueous polarographic half-wave potentials.2 However, in these correlations, the ionization potentials (which are ge ...
eassy - BSE8J2009
... particles (mostly electrons) accelerate along the magnetic field lines into the upper atmosphere, where they collide with gas atoms, causing the atoms to give off light. But why does that happen? To find the answer, we must look further away, to the Sun. The spectacular, "great" auroras in "what do ...
... particles (mostly electrons) accelerate along the magnetic field lines into the upper atmosphere, where they collide with gas atoms, causing the atoms to give off light. But why does that happen? To find the answer, we must look further away, to the Sun. The spectacular, "great" auroras in "what do ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.