Visible Light Photoredox Catalysis with Transition
... sporadically employed as photocatalysts in the area of organic synthesis. The limited exploration of this area is perhaps surprising, as single-electron, radical processes have long been employed in C−C bond construction and often provide access to reactivity that is complementary to that of closed- ...
... sporadically employed as photocatalysts in the area of organic synthesis. The limited exploration of this area is perhaps surprising, as single-electron, radical processes have long been employed in C−C bond construction and often provide access to reactivity that is complementary to that of closed- ...
Chapter - Imperial Valley College
... Representing Compounds Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the ...
... Representing Compounds Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the ...
Chapter 2 1.Certain gases in the 293K and 9.97 × 104Pa when the
... Answer: In general, the electron affinity and energy with the decrease of atomic radius increases because the radius of hours, the nuclear charge of the electron cited large, therefore, electron affinity and energy left to right in the same cycle have an increasing trend, while the same The tribe wa ...
... Answer: In general, the electron affinity and energy with the decrease of atomic radius increases because the radius of hours, the nuclear charge of the electron cited large, therefore, electron affinity and energy left to right in the same cycle have an increasing trend, while the same The tribe wa ...
Chapter
... Representing Compounds Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the ...
... Representing Compounds Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the ...
IB Chemistry Online SAQ_Ans
... c From the equation, amount of H2SO4 = amount of NaOH ÷ 2 = 0.0125 mol in 20.0 cm3, so ‘scaling up’ to 1000 cm3 to obtain the concentration of diluted sulfuric acid 1000 × 0.0125mol dm −3 ...
... c From the equation, amount of H2SO4 = amount of NaOH ÷ 2 = 0.0125 mol in 20.0 cm3, so ‘scaling up’ to 1000 cm3 to obtain the concentration of diluted sulfuric acid 1000 × 0.0125mol dm −3 ...
contact - DTU Kemi
... DTU Chemistry works hard to ensure the best possible platform to generate and develop ideas to the benefit of a sustainable development. In order to strengthen the research profile and our visibility further, the Department will be organized in two major sections in 2015. One section will cover Phys ...
... DTU Chemistry works hard to ensure the best possible platform to generate and develop ideas to the benefit of a sustainable development. In order to strengthen the research profile and our visibility further, the Department will be organized in two major sections in 2015. One section will cover Phys ...
Photo-oxidation of pinonaldehyde at low NOx
... those of the precursor molecules, and methanol is also substantially less reactive with OH. Consequently the methanol had a minimal effect on the radical balance. There is no evidence for interference from isobaric ions for either the precursors or the OH tracer; they are the dominant gas-phase orga ...
... those of the precursor molecules, and methanol is also substantially less reactive with OH. Consequently the methanol had a minimal effect on the radical balance. There is no evidence for interference from isobaric ions for either the precursors or the OH tracer; they are the dominant gas-phase orga ...
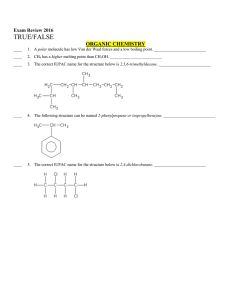
Multiple Choice Exam Review June 2016
... ____ 17. Carbon tetrafluoride, CF4, contains four polar bonds and is a polar molecule. _________________________ ____ 18. A molecule whose central atom is surrounded by two lone pairs and four atoms will have a square planar shape. _________________________ ____ 19. CH3Cl is a polar molecule. ______ ...
... ____ 17. Carbon tetrafluoride, CF4, contains four polar bonds and is a polar molecule. _________________________ ____ 18. A molecule whose central atom is surrounded by two lone pairs and four atoms will have a square planar shape. _________________________ ____ 19. CH3Cl is a polar molecule. ______ ...
Chapter 4: Quantities of Reactants and Products
... Check your answers: A properly balanced equation has the same numbers of atoms of each type (4 C, 12 H, and 14 O) in the products and reactants. The law of conservation of mass says that masses of the reactants must add up to the masses of the products, 284.16 g. This looks right. 12. Define the pro ...
... Check your answers: A properly balanced equation has the same numbers of atoms of each type (4 C, 12 H, and 14 O) in the products and reactants. The law of conservation of mass says that masses of the reactants must add up to the masses of the products, 284.16 g. This looks right. 12. Define the pro ...
44. Find рН of formic acid solution with mass percent ω=5
... 17. What happens to erythrocyte which is placed in 3% glucose solution? Prove with calculations. 18. Depression in freezing point of blood serum is 0,56 K. Find the molar concentration of salts in blood if all salts are binary electrolytes and completely dissociated. 19. Compare osmotic pressures of ...
... 17. What happens to erythrocyte which is placed in 3% glucose solution? Prove with calculations. 18. Depression in freezing point of blood serum is 0,56 K. Find the molar concentration of salts in blood if all salts are binary electrolytes and completely dissociated. 19. Compare osmotic pressures of ...
1412_lecture_ch16 Fall_2014
... Complex-Ion Formation The formation constant, Kf, is the equilibrium constant for the formation of a complex ion from the aqueous metal ion and the ligands. The large value means that the complex ion is quite stable. When a large amount of NH3 is added to a solution of Ag+, you expect most of the A ...
... Complex-Ion Formation The formation constant, Kf, is the equilibrium constant for the formation of a complex ion from the aqueous metal ion and the ligands. The large value means that the complex ion is quite stable. When a large amount of NH3 is added to a solution of Ag+, you expect most of the A ...
2016-2018 Syllabus - Cambridge International Examinations
... The following grade descriptions indicate the level of attainment characteristic of the middle of the given grade band. They give a general indication of the required standard at each specified grade. The descriptions should be interpreted in relation to the content outlined in the syllabus; they ar ...
... The following grade descriptions indicate the level of attainment characteristic of the middle of the given grade band. They give a general indication of the required standard at each specified grade. The descriptions should be interpreted in relation to the content outlined in the syllabus; they ar ...
Chapter 4 - AP Chemistry with dr hart
... • It is helpful to pay attention to exactly what species are present in a reaction mixture (i.e., solid, liquid, gas, aqueous solution). • If we are to understand reactivity, we must be aware of just what is changing during the course of a reaction. Aqueous Reactions © 2009, Prentice-Hall, Inc. ...
... • It is helpful to pay attention to exactly what species are present in a reaction mixture (i.e., solid, liquid, gas, aqueous solution). • If we are to understand reactivity, we must be aware of just what is changing during the course of a reaction. Aqueous Reactions © 2009, Prentice-Hall, Inc. ...
2014 Syllabus - Cambridge International Examinations
... Paper 3 – Advanced Practical Skills 1/2 In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers w ...
... Paper 3 – Advanced Practical Skills 1/2 In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers w ...
Solving Problems: A Chemistry Handbook
... a. taste of an apple d. length of a rod b. mass of a brick e. texture of a leaf c. speed of a car f. weight of an elephant A hypothesis is a possible explanation for what has been observed. Based on the observations of ozone thinning and CFC buildup in the atmosphere, the chemists Mario Molina and F ...
... a. taste of an apple d. length of a rod b. mass of a brick e. texture of a leaf c. speed of a car f. weight of an elephant A hypothesis is a possible explanation for what has been observed. Based on the observations of ozone thinning and CFC buildup in the atmosphere, the chemists Mario Molina and F ...
the chemical and physical properties of condensed
... Examples of the influence of external factors upon the transitions of phosphates are found in the transitions of sodium tetrametaphosphate tetrahydrate and sodium triphosphate hexahydrate. The tetrametaphosphate will be discussed first. In this transition Na4P4O124H2O suffers a crystallographic chan ...
... Examples of the influence of external factors upon the transitions of phosphates are found in the transitions of sodium tetrametaphosphate tetrahydrate and sodium triphosphate hexahydrate. The tetrametaphosphate will be discussed first. In this transition Na4P4O124H2O suffers a crystallographic chan ...
content - Thesis Scientist
... CBSE (Delhi) SET–II Questions Uncommon to Set-I 1. What is the total number of atoms per unit cell in a face-centred cubic (fcc) structure? 2. What is primary cell? Give an example. 6. Write the IUPAC name of the following compound: CH3 COCH2 COCH3 9. State Raoult’s law for solutions of volatile liq ...
... CBSE (Delhi) SET–II Questions Uncommon to Set-I 1. What is the total number of atoms per unit cell in a face-centred cubic (fcc) structure? 2. What is primary cell? Give an example. 6. Write the IUPAC name of the following compound: CH3 COCH2 COCH3 9. State Raoult’s law for solutions of volatile liq ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.