study material(2014-15) class xii-chemistry
... Students‘ common errors, un-attempted questions and their remediation. Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials pre ...
... Students‘ common errors, un-attempted questions and their remediation. Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials pre ...
CLUE - virtual laboratories
... many times do we hear about “natural remedies, without drugs or chemicals,” despite the fact that everything is composed of chemicals and the most toxic chemicals known are natural products.2 A growing body of research results on student understanding of chemistry indicates, pretty emphatically, tha ...
... many times do we hear about “natural remedies, without drugs or chemicals,” despite the fact that everything is composed of chemicals and the most toxic chemicals known are natural products.2 A growing body of research results on student understanding of chemistry indicates, pretty emphatically, tha ...
Chapter 19 Homework Problems Answers
... We can arrive at this net reaction in an equivalent way, namely, by vaporizing all of the necessary elements to give gaseous atoms, and then allowing the gaseous atoms to form all of the appropriate bonds. The overall enthalpy of formation by this route is numerically equal to that for the above rea ...
... We can arrive at this net reaction in an equivalent way, namely, by vaporizing all of the necessary elements to give gaseous atoms, and then allowing the gaseous atoms to form all of the appropriate bonds. The overall enthalpy of formation by this route is numerically equal to that for the above rea ...
CHAPTER 4 - Myschoolpages.com
... Nonelectrolytes are not dissociated into ions in solution Extent of dissolution does not dictate strong or weak electrolyte solution (i.e., HC2H3O2 is very soluble but is a weak electrolyte while Ba(OH)2 is only slightly soluble is a strong electrolyte) ...
... Nonelectrolytes are not dissociated into ions in solution Extent of dissolution does not dictate strong or weak electrolyte solution (i.e., HC2H3O2 is very soluble but is a weak electrolyte while Ba(OH)2 is only slightly soluble is a strong electrolyte) ...
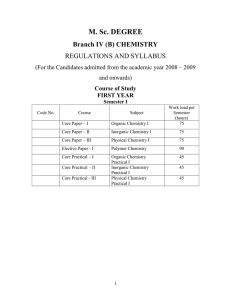
M.Sc. Chemistry - Periyar University
... PHYSICAL CHEMISTRY – I (75 Hours) Unit – I Classical Thermodynamics – I (15 Hours) Maxwell’s relations and thermodynamic equations of state – applications in the evaluation of Cp – Cv for solids and for vanderwaals gases, Cp – Cv in terms of coefficient of expansion and coefficient of compressibilit ...
... PHYSICAL CHEMISTRY – I (75 Hours) Unit – I Classical Thermodynamics – I (15 Hours) Maxwell’s relations and thermodynamic equations of state – applications in the evaluation of Cp – Cv for solids and for vanderwaals gases, Cp – Cv in terms of coefficient of expansion and coefficient of compressibilit ...
introduction - TestBankTop
... Strategy: For subtraction and addition, the number of significant figures to the right of the decimal point in that part of the calculation is determined by the lowest number of digits to the right of the decimal point in any of the original numbers. For the division part of the calculation, the num ...
... Strategy: For subtraction and addition, the number of significant figures to the right of the decimal point in that part of the calculation is determined by the lowest number of digits to the right of the decimal point in any of the original numbers. For the division part of the calculation, the num ...
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical
... The third equation has water as a product, and we need to “consume” the water formed in equation two, so let’s reverse equation 3—changing the sign—AND multiply it by 6 6 H2O (g) → 6 H2 (g) + 3 O2 (g) ΔH = (+241.8)(6) kJ Adding these 3 equations gives 2 N2 (g) + 2 O2 (g) → 4 ΝΟ (g ) ...
... The third equation has water as a product, and we need to “consume” the water formed in equation two, so let’s reverse equation 3—changing the sign—AND multiply it by 6 6 H2O (g) → 6 H2 (g) + 3 O2 (g) ΔH = (+241.8)(6) kJ Adding these 3 equations gives 2 N2 (g) + 2 O2 (g) → 4 ΝΟ (g ) ...
STUDY MATERIAL 2016-17 CHEMISTRY CLASS XII
... a=4R/√3 (where R = Radius of sphere) 26. Imperfection is the irregularity in the arrangement of constituent particles. 27. Point defect or Atomic defect: It is the deviation from ideal arrangement of constituent atoms. 28. Vacancy defect lowers the density of crystal lattice. 29. Interstitial defect ...
... a=4R/√3 (where R = Radius of sphere) 26. Imperfection is the irregularity in the arrangement of constituent particles. 27. Point defect or Atomic defect: It is the deviation from ideal arrangement of constituent atoms. 28. Vacancy defect lowers the density of crystal lattice. 29. Interstitial defect ...
SCH3U0FinalExamReview - Savita Pall and Chemistry
... 1. What is the meaning of effective nuclear charge? How does the effective nuclear charge felt by the outer electrons vary going down a group? How does it change as we go from left to right across a period? 2. Choose the larger atom in each pair: (a) Na or Si; (b) P or Sb. 3. Choose the larger atom ...
... 1. What is the meaning of effective nuclear charge? How does the effective nuclear charge felt by the outer electrons vary going down a group? How does it change as we go from left to right across a period? 2. Choose the larger atom in each pair: (a) Na or Si; (b) P or Sb. 3. Choose the larger atom ...
GCE Chemistry Specification (From 2015 - WALES ONLY
... principles of the mass spectrometer and its use in determining relative atomic mass and relative abundance of isotopes ...
... principles of the mass spectrometer and its use in determining relative atomic mass and relative abundance of isotopes ...
File
... of most sand, rocks, and soils. Again, it is not too surprising that silicon is the second most abundant element, as it is involved in the composition of much of the earth. The four most abundant elements in the human body are oxygen, carbon, hydrogen, and nitrogen. Not surprisingly, these elements ...
... of most sand, rocks, and soils. Again, it is not too surprising that silicon is the second most abundant element, as it is involved in the composition of much of the earth. The four most abundant elements in the human body are oxygen, carbon, hydrogen, and nitrogen. Not surprisingly, these elements ...
INTRODUCTION - Test Bank wizard
... Strategy: For subtraction and addition, the number of significant figures to the right of the decimal point in that part of the calculation is determined by the lowest number of digits to the right of the decimal point in any of the original numbers. For the division part of the calculation, the num ...
... Strategy: For subtraction and addition, the number of significant figures to the right of the decimal point in that part of the calculation is determined by the lowest number of digits to the right of the decimal point in any of the original numbers. For the division part of the calculation, the num ...
Ratios and fraction arithmetic
... Continued ratios are used to compare more than two numbers together. They are also usually written with colons (:). For example, the ratio of 4 to 8 to 12 is written 4 : 8 : 12. Example: Thomas needs 2 spiral notebooks, 4 pencils, 1 red correcting pen and 2 sharpies for each of his classes every day ...
... Continued ratios are used to compare more than two numbers together. They are also usually written with colons (:). For example, the ratio of 4 to 8 to 12 is written 4 : 8 : 12. Example: Thomas needs 2 spiral notebooks, 4 pencils, 1 red correcting pen and 2 sharpies for each of his classes every day ...
2008 Equilibrium -- without math (PowerPoint 13 MB)
... between forward and reverse reactions. In most cases, this balance is quite delicate. Changes in experimental conditions (concentration, pressure, volume and temperature) may disturb the balance and shift the equilibrium position so that more or less of the desired product is formed. When we say tha ...
... between forward and reverse reactions. In most cases, this balance is quite delicate. Changes in experimental conditions (concentration, pressure, volume and temperature) may disturb the balance and shift the equilibrium position so that more or less of the desired product is formed. When we say tha ...
1 1411_chapter 6 exercises with answers CHEM 1411, chapter 6
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. ...
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. ...
Entropy and Free Energy
... Instead, for processes other than isothermal expansion or compression of an ideal gas (for which we can use Equation 14.4), we routinely determine entropy changes using tabulated values. Using calorimetry [9 Section 10.4], it is possible to determine the absolute value of the entropy of a substance, ...
... Instead, for processes other than isothermal expansion or compression of an ideal gas (for which we can use Equation 14.4), we routinely determine entropy changes using tabulated values. Using calorimetry [9 Section 10.4], it is possible to determine the absolute value of the entropy of a substance, ...
Solving General Chemistry Problems 5e
... only arithmetic and simple algebra. Nevertheless, if you don't understand it, you can expect troubles before long. So, before you can really get into chemistry, you need to master the mathematical operations in the first six chapters. 3. Don't think of your calculator as a security blanket that will ...
... only arithmetic and simple algebra. Nevertheless, if you don't understand it, you can expect troubles before long. So, before you can really get into chemistry, you need to master the mathematical operations in the first six chapters. 3. Don't think of your calculator as a security blanket that will ...
Conformational studies of aliphatic secondary ozonides
... The only experimental attempt to define the geometrical structure of propene ozonide was made by L. Kuczkowski et al in 1974 [8], by collection of a microwave absorption spectra of gaseous propene SOZ at 195 K. In this study only one conformer of the molecule had been observed, possessing the O-O hal ...
... The only experimental attempt to define the geometrical structure of propene ozonide was made by L. Kuczkowski et al in 1974 [8], by collection of a microwave absorption spectra of gaseous propene SOZ at 195 K. In this study only one conformer of the molecule had been observed, possessing the O-O hal ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.