deltahpps
... If you formed the products from their elements you should need the same amounts of every substance as if you formed the reactants from their elements. ...
... If you formed the products from their elements you should need the same amounts of every substance as if you formed the reactants from their elements. ...
2013 - SQA
... 19. Limonene is one of the terpene molecules responsible for the flavour of lemons. ...
... 19. Limonene is one of the terpene molecules responsible for the flavour of lemons. ...
Rhenium- and molybdenum-catalyzed dehydration reactions
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
... ratio close to one and are highly functionalized with hydroxyl groups. Therefore a completely different type of chemistry is required to acquire building blocks from lignocellulosic biomass suitable for the chemical industry: while in the case of fossil feedstocks functionality must be added, functi ...
Complete Solution Manual
... For a spontaneous reaction at standard conditions, E ocell must be positive (E ocell = E ored + E oox > 0). For any two half-reactions, there is only one way to manipulate them to come up with a positive E ocell (a spontaneous reaction). Because the half-reactions do not depend on how many times the ...
... For a spontaneous reaction at standard conditions, E ocell must be positive (E ocell = E ored + E oox > 0). For any two half-reactions, there is only one way to manipulate them to come up with a positive E ocell (a spontaneous reaction). Because the half-reactions do not depend on how many times the ...
23. Oxidation and Reduction
... 3. The algebraic sum of the oxidation numbers of the atoms in the formula of a compound is zero. 4. In compounds, the oxidation number of hydrogen is +1. (There is one exception. In compounds known as hydrides, it can be -1. Sodium hydride is NaH.) 5. In compounds, the oxidation number of oxygen is ...
... 3. The algebraic sum of the oxidation numbers of the atoms in the formula of a compound is zero. 4. In compounds, the oxidation number of hydrogen is +1. (There is one exception. In compounds known as hydrides, it can be -1. Sodium hydride is NaH.) 5. In compounds, the oxidation number of oxygen is ...
Thermochemistry - hrsbstaff.ednet.ns.ca
... that harness energy-producing processes. Humans continually devise new technologies that use chemical reactions to produce materials with useful properties. Since the invention of fire, humans have also worked to devise technologies that harness energy. These technologies depend on the fact that eve ...
... that harness energy-producing processes. Humans continually devise new technologies that use chemical reactions to produce materials with useful properties. Since the invention of fire, humans have also worked to devise technologies that harness energy. These technologies depend on the fact that eve ...
Complete Solution Manual
... For a spontaneous reaction at standard conditions, E ocell must be positive (E ocell = E ored + E oox > 0). For any two half-reactions, there is only one way to manipulate them to come up with a positive E ocell (a spontaneous reaction). Because the half-reactions do not depend on how many times the ...
... For a spontaneous reaction at standard conditions, E ocell must be positive (E ocell = E ored + E oox > 0). For any two half-reactions, there is only one way to manipulate them to come up with a positive E ocell (a spontaneous reaction). Because the half-reactions do not depend on how many times the ...
5. Coenzyme HAD+ is derived
... 2 Practical Skills: -apply safety rules at work in chemical laboratories, chemical use utensils, basic tools and methods of physical and chemical measurements needed in medical practice; - calculate the molar, molal concentration, molar concentration equivalent mass fraction of the solute, the titer ...
... 2 Practical Skills: -apply safety rules at work in chemical laboratories, chemical use utensils, basic tools and methods of physical and chemical measurements needed in medical practice; - calculate the molar, molal concentration, molar concentration equivalent mass fraction of the solute, the titer ...
General and Inorganic Chemistry
... 3. III Basic laboratory procedures and methods ............................................................................... 20 1. III.1 Basic guidelines for working with hazardous materials .............................................. 20 1.1. III.1.1 Laboratory safety .......................... ...
... 3. III Basic laboratory procedures and methods ............................................................................... 20 1. III.1 Basic guidelines for working with hazardous materials .............................................. 20 1.1. III.1.1 Laboratory safety .......................... ...
LABORATORY MANUAL GENERAL CHEMISTRY 120 Dr. Steven Fawl
... calculation. Calculations must be neat and easy to follow; units must be used every time they apply. You may add paper to your report, if you need additional room for your calculations. (Hint: do your calculations on scratch paper first, and then copy to report.) It is very important that you do no ...
... calculation. Calculations must be neat and easy to follow; units must be used every time they apply. You may add paper to your report, if you need additional room for your calculations. (Hint: do your calculations on scratch paper first, and then copy to report.) It is very important that you do no ...
orange review book_2014_key
... 18. Which substance can be broken down by a chemical change? (1) antimony (3) hexane (2) carbon (4) sulfur 19. One similarity between all mixtures and compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute ...
... 18. Which substance can be broken down by a chemical change? (1) antimony (3) hexane (2) carbon (4) sulfur 19. One similarity between all mixtures and compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute ...
organic problems - St. Olaf College
... A) they are all constitutional isomers. B) they are always hydrocarbons. C) each differs from its nearest neighbors by 14 amu. D) they may also be classified as tautomers. 28 How many structurally distinct (different) sets of hydrogens are present in (CH3)3CCH2OCH3? A) 2 B) 3 C) 4 D) 8 29 The pKas o ...
... A) they are all constitutional isomers. B) they are always hydrocarbons. C) each differs from its nearest neighbors by 14 amu. D) they may also be classified as tautomers. 28 How many structurally distinct (different) sets of hydrogens are present in (CH3)3CCH2OCH3? A) 2 B) 3 C) 4 D) 8 29 The pKas o ...
Tro Chemistry a Molecular Approach, 3E
... (feel your engine after a drive if you doubt this). The total energy given off by the combustion reaction exactly equals the sum of the amount of energy that goes to propel the car and the amount dissipated as heat—energy is conserved. In other words, when it comes to energy, you can’t win; you cann ...
... (feel your engine after a drive if you doubt this). The total energy given off by the combustion reaction exactly equals the sum of the amount of energy that goes to propel the car and the amount dissipated as heat—energy is conserved. In other words, when it comes to energy, you can’t win; you cann ...
2nd Semester Practice Chemistry Final 2009
... b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the gas state? a. vaporization c. surface tension b. evaporation d. aeration 45. What can happe ...
... b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the gas state? a. vaporization c. surface tension b. evaporation d. aeration 45. What can happe ...
File - UTeach Dallas Project
... use direct and inverse proportion. use positive, negative and whole number indices. draw charts and graphs from given data. interpret charts and graphs. select suitable scales and axes for graphs. make approximate evaluations of numerical expressions. recognise and use the relationship between lengt ...
... use direct and inverse proportion. use positive, negative and whole number indices. draw charts and graphs from given data. interpret charts and graphs. select suitable scales and axes for graphs. make approximate evaluations of numerical expressions. recognise and use the relationship between lengt ...
Unit 9 Stoichiometry Notes
... Stoichiometry is a big word for a process that chemist’s use to calculate amounts in reactions. It makes use of the coefficient ratio set up by balanced reaction equations to make connections between the reactants and products in reactions. Stoichiometry calculates the quantities of reactants an ...
... Stoichiometry is a big word for a process that chemist’s use to calculate amounts in reactions. It makes use of the coefficient ratio set up by balanced reaction equations to make connections between the reactants and products in reactions. Stoichiometry calculates the quantities of reactants an ...
CHANNELING OF SUBSTRATES AND INTERMEDIATES IN
... a conduit for indole. Subsequent higher-resolution structural analyses revealed two sites along the tunnel that are partially blocked. At one site, Phe-280 in the β-subunit inserts directly into the channel (35). Strikingly, exchange of potassium or cesium ions for sodium ions results in a movement ...
... a conduit for indole. Subsequent higher-resolution structural analyses revealed two sites along the tunnel that are partially blocked. At one site, Phe-280 in the β-subunit inserts directly into the channel (35). Strikingly, exchange of potassium or cesium ions for sodium ions results in a movement ...
National German competition
... To become a member of the German IChO-team you have to be successful in four rounds. The problems to be solved in the 1st round are sent to all highschools. To solve the problems the students may use all resources available, e.g. textbooks etc. All the students who solve about 70% will receive the p ...
... To become a member of the German IChO-team you have to be successful in four rounds. The problems to be solved in the 1st round are sent to all highschools. To solve the problems the students may use all resources available, e.g. textbooks etc. All the students who solve about 70% will receive the p ...
Document
... What is the heat capacity of a container if 100. g of water (s = 4.18 J/g ˚C) at 100. ˚C are added to 100. g of water at 25 ˚C in the container and the final temperature is 61˚C? qlost by hot water = m × t × s =(100. g)(61 °C – 100.˚C)(4.18 J/g ˚C) = –16,302 J qgained by cold water = (100. g)(61 °C ...
... What is the heat capacity of a container if 100. g of water (s = 4.18 J/g ˚C) at 100. ˚C are added to 100. g of water at 25 ˚C in the container and the final temperature is 61˚C? qlost by hot water = m × t × s =(100. g)(61 °C – 100.˚C)(4.18 J/g ˚C) = –16,302 J qgained by cold water = (100. g)(61 °C ...
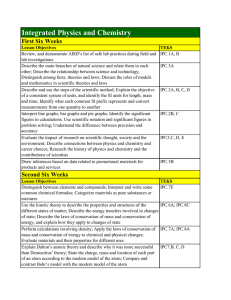
Integrated Physics and Chemistry
... periodic table; Locate semiconductors, halogens, and noble gases in the periodic table; Relate an element’s chemical properties to the electron arrangement of its atoms Explain the relationship between a mole of a substance and Avogadro’s constant; Find the molar mass of an element by using the peri ...
... periodic table; Locate semiconductors, halogens, and noble gases in the periodic table; Relate an element’s chemical properties to the electron arrangement of its atoms Explain the relationship between a mole of a substance and Avogadro’s constant; Find the molar mass of an element by using the peri ...
Energy Changes in Chemical Reactions
... however; the reaction also releases energy in the form of heat and light. So our description of this reaction was incomplete. A complete description of a chemical reaction includes not only the identity, amount, and chemical form of the reactants and products but also the quantity of energy produced ...
... however; the reaction also releases energy in the form of heat and light. So our description of this reaction was incomplete. A complete description of a chemical reaction includes not only the identity, amount, and chemical form of the reactants and products but also the quantity of energy produced ...
Energy Changes in Chemical Reactions
... however; the reaction also releases energy in the form of heat and light. So our description of this reaction was incomplete. A complete description of a chemical reaction includes not only the identity, amount, and chemical form of the reactants and products but also the quantity of energy produced ...
... however; the reaction also releases energy in the form of heat and light. So our description of this reaction was incomplete. A complete description of a chemical reaction includes not only the identity, amount, and chemical form of the reactants and products but also the quantity of energy produced ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.