College Chemistry
... Note that symbols for multiplied units may be separated by a dot or a space, e.g., kg•s or kg s. Symbols for divided units may be written with a solidus or an exponent, e.g., m/s or m•s-1 or m s-1. Temperature is an independent dimension which cannot be defined in terms of mass, length, and time. Th ...
... Note that symbols for multiplied units may be separated by a dot or a space, e.g., kg•s or kg s. Symbols for divided units may be written with a solidus or an exponent, e.g., m/s or m•s-1 or m s-1. Temperature is an independent dimension which cannot be defined in terms of mass, length, and time. Th ...
Document
... The number of protons and electrons determine the element (in uncharged atoms the number of protons equals the number of electrons) Atoms of an element with differing numbers of neutrons are called isotopes ...
... The number of protons and electrons determine the element (in uncharged atoms the number of protons equals the number of electrons) Atoms of an element with differing numbers of neutrons are called isotopes ...
physical setting chemistry
... 5 Which event must always occur for a chemical reaction to take place? (1) formation of a precipitate (2) formation of a gas (3) effective collisions between reacting particles (4) addition of a catalyst to the reaction system P.S./Chem.–Jan. ’03 ...
... 5 Which event must always occur for a chemical reaction to take place? (1) formation of a precipitate (2) formation of a gas (3) effective collisions between reacting particles (4) addition of a catalyst to the reaction system P.S./Chem.–Jan. ’03 ...
Isotope-Exchange Evidence that Glucose 6
... were determined from progress curves. All reactions were carried out in 50-mM imidazole nitrate buffer, pH 6.5, containing I-mM dithiothreitol, I-mM excess Mg2+ over the total adenine nucleotide concentration and 100-mM KCI. An excess of 1-mM MgZ is sufficient to ensure that ATP and ADP exist in a l ...
... were determined from progress curves. All reactions were carried out in 50-mM imidazole nitrate buffer, pH 6.5, containing I-mM dithiothreitol, I-mM excess Mg2+ over the total adenine nucleotide concentration and 100-mM KCI. An excess of 1-mM MgZ is sufficient to ensure that ATP and ADP exist in a l ...
Determination of Equilibrium Constants for Reactions between Nitric
... two compared absorbents when simulated flue gas was treated with about 5% oxygen. Moreover, the main byproducts were nitrite and nitrate, which are sources of fertilizers. Thus, the hexamminecobalt(II) solution can be considered as an ideal absorbent for wet flue gas treatment (FGT) technology. A seri ...
... two compared absorbents when simulated flue gas was treated with about 5% oxygen. Moreover, the main byproducts were nitrite and nitrate, which are sources of fertilizers. Thus, the hexamminecobalt(II) solution can be considered as an ideal absorbent for wet flue gas treatment (FGT) technology. A seri ...
BARIUM NITRATE
... 9.32eV, Be2+ 18.21 eV; oxidation state +2 Occurrence and Uses Beryllium is widely distributed in the earth’s crust at trace concentration, 2.8 mg/kg. The element was first discovered by Vauquelin in 1797. Wohler and Bussy in 1828 independently isolated beryllium in the metallic form from its oxide. ...
... 9.32eV, Be2+ 18.21 eV; oxidation state +2 Occurrence and Uses Beryllium is widely distributed in the earth’s crust at trace concentration, 2.8 mg/kg. The element was first discovered by Vauquelin in 1797. Wohler and Bussy in 1828 independently isolated beryllium in the metallic form from its oxide. ...
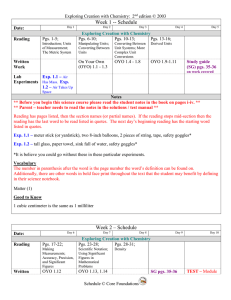
Week 1 -- Schedule
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
CHML 1045 General Chemistry I: Laboratory Manual
... Consequently, as the semester moves forward the labs often become more and more sophisticated. But, you will be building upon the work you did previously, and hence it shouldn’t be that much harder; only what you prep for will change. Getting into a habit of good preparation will make this easier. B ...
... Consequently, as the semester moves forward the labs often become more and more sophisticated. But, you will be building upon the work you did previously, and hence it shouldn’t be that much harder; only what you prep for will change. Getting into a habit of good preparation will make this easier. B ...
Chapter 1: Matter and Measurements
... (b) “A molecule is made up of nonmetal atoms” is always true, by definition. (c) “An ionic compound has at least one metal atom” is usually true. Most ionic compounds do contain a metal, but there are ionic compounds in which the cation (positive ion) is not a metal, such as in ammonium chloride, NH ...
... (b) “A molecule is made up of nonmetal atoms” is always true, by definition. (c) “An ionic compound has at least one metal atom” is usually true. Most ionic compounds do contain a metal, but there are ionic compounds in which the cation (positive ion) is not a metal, such as in ammonium chloride, NH ...
Chapter 1
... 1.3 Associating an element’s name with its symbol requires memorization of both names and symbols. The examples in this problem all begin with the letter “A” but their names do not necessarily begin with “A” too: (a) Arsenic; (b) argon; (c) aluminum; (d) americium; (e) silver; (f) gold; (g) astatine ...
... 1.3 Associating an element’s name with its symbol requires memorization of both names and symbols. The examples in this problem all begin with the letter “A” but their names do not necessarily begin with “A” too: (a) Arsenic; (b) argon; (c) aluminum; (d) americium; (e) silver; (f) gold; (g) astatine ...
Acid Base Equilibrium Diploma Questions
... replacing the vinegar with OH-(aq) reacting the vinegar with carbonic acid shifting the equilibrium to the right because [OH-(aq)] would decrease shifting the equilibrium to the left, producing more carbonic acid Use the following information to answer the next _ questions. In Canada, the annual pro ...
... replacing the vinegar with OH-(aq) reacting the vinegar with carbonic acid shifting the equilibrium to the right because [OH-(aq)] would decrease shifting the equilibrium to the left, producing more carbonic acid Use the following information to answer the next _ questions. In Canada, the annual pro ...
Homework 5-7 answers
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
Chapter 4 - Chemistry
... Solution: CH3COOH is a weak acid. It will be shown as a molecule in the ionic equation. KOH is a strong base. It completely ionizes to K and OH ions. Since CH3COOH is an acid, it donates an H to the base, OH, producing water. The other product is the salt, CH3COOK, which is soluble and remains i ...
... Solution: CH3COOH is a weak acid. It will be shown as a molecule in the ionic equation. KOH is a strong base. It completely ionizes to K and OH ions. Since CH3COOH is an acid, it donates an H to the base, OH, producing water. The other product is the salt, CH3COOK, which is soluble and remains i ...
Chapter 5: Gases - HCC Learning Web
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
Civilian Radioactive Waste Management System Management & Operating Contractor Thomas W. Wolery,
... “data0” file (which can be created or modified by a text editor) and a parallel unformatted form known as a “data1” file. The latter is sometimes referred to as a “binary” file; however, it is technically merely unformatted. The two forms of a data file share a three-letter “datafilekey,” which serv ...
... “data0” file (which can be created or modified by a text editor) and a parallel unformatted form known as a “data1” file. The latter is sometimes referred to as a “binary” file; however, it is technically merely unformatted. The two forms of a data file share a three-letter “datafilekey,” which serv ...
Chapter 3 - Chemistry
... Strategy: We are asked to solve for the number of N, C, O, and H atoms in 1.68 104 g of urea. We cannot convert directly from grams urea to atoms. What unit do we need to obtain first before we can convert to atoms? How should Avogadro's number be used here? How many atoms of N, C, O, or H are in ...
... Strategy: We are asked to solve for the number of N, C, O, and H atoms in 1.68 104 g of urea. We cannot convert directly from grams urea to atoms. What unit do we need to obtain first before we can convert to atoms? How should Avogadro's number be used here? How many atoms of N, C, O, or H are in ...
CHAPTER 3 MASS RELATIONSHIPS IN CHEMICAL REACTIONS
... Strategy: We can look up the molar mass of arsenic (As) on the periodic table (74.92 g/mol). We want to find the mass of a single atom of arsenic (unit of g/atom). Therefore, we need to convert from the unit mole in the denominator to the unit atom in the denominator. What conversion factor is neede ...
... Strategy: We can look up the molar mass of arsenic (As) on the periodic table (74.92 g/mol). We want to find the mass of a single atom of arsenic (unit of g/atom). Therefore, we need to convert from the unit mole in the denominator to the unit atom in the denominator. What conversion factor is neede ...
Experiments in General Chemistry: Featuring MeasureNet
... that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and solids. The density of a substance can be used to identify a liquid or solid because density is an intensive property. Intensive properties are p ...
... that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and solids. The density of a substance can be used to identify a liquid or solid because density is an intensive property. Intensive properties are p ...
Homework 5-8 answers
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
... A) the energy stored within the structural units of chemical substances. B) the energy associated with the random motion of atoms and molecules. C) solar energy, i.e. energy that comes from the sun. D) energy available by virtue of an object's position. Ans: C Category: Easy Section: 6.1 2. Thermal ...
Moles Workbook
... protons and neutrons. The electrons circulate round the nucleus in specific orbits or shells. We can picture the hydrogen atom - the simplest of all atoms with one electron, and one proton in the nucleus - by considering a pea placed in the centre of a football pitch, to represent the nucleus with i ...
... protons and neutrons. The electrons circulate round the nucleus in specific orbits or shells. We can picture the hydrogen atom - the simplest of all atoms with one electron, and one proton in the nucleus - by considering a pea placed in the centre of a football pitch, to represent the nucleus with i ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.