
Learning at the symbolic level
... Keith S. Taber, University of Cambridge, U. K. ([email protected]) Abstract: The symbolic language of chemistry is extensive, and is used ubiquitously in teaching and learning the subject at secondary level and beyond. This chapter considers how this ‘language’, which acts as such a powerful facilitat ...
... Keith S. Taber, University of Cambridge, U. K. ([email protected]) Abstract: The symbolic language of chemistry is extensive, and is used ubiquitously in teaching and learning the subject at secondary level and beyond. This chapter considers how this ‘language’, which acts as such a powerful facilitat ...
Solution - HCC Learning Web
... Plan We write the chemical formulas of the reactants and products and then determine which product is insoluble. We then write and balance the molecular equation. Next, we write each soluble strong electrolyte as separated ions to obtain the complete ionic equation. Finally, we eliminate the spectat ...
... Plan We write the chemical formulas of the reactants and products and then determine which product is insoluble. We then write and balance the molecular equation. Next, we write each soluble strong electrolyte as separated ions to obtain the complete ionic equation. Finally, we eliminate the spectat ...
Stoichiometry - Social Circle City Schools
... The mole (abbreviated mol) is the unit chemists use when counting numbers of atoms or molecules in a sample. The number of particles (atoms, molecules, or other objects) in one mole is equal to the number of atoms in exactly 12 g of carbon-12. This number of particles is called Avogadro’s number (NA ...
... The mole (abbreviated mol) is the unit chemists use when counting numbers of atoms or molecules in a sample. The number of particles (atoms, molecules, or other objects) in one mole is equal to the number of atoms in exactly 12 g of carbon-12. This number of particles is called Avogadro’s number (NA ...
Chapter 4 Classifying Reactions: Chemicals in Balance
... In writing an equation the reactants are the starting materials and they are on the left side. The products are the new substances that form and they are on the right side. A plus sign, “+”, separate more than one reactant or product from another and an →, read as “to form”, separates the reactant(s ...
... In writing an equation the reactants are the starting materials and they are on the left side. The products are the new substances that form and they are on the right side. A plus sign, “+”, separate more than one reactant or product from another and an →, read as “to form”, separates the reactant(s ...
Common Curriculum Map Discipline: Science Course: Chemistry
... -How do you practice safety in the laboratory? -What units are used in science for measurement? -What are metric prefixes and how are they used? -What causes uncertainty in measurements? -What is the difference between accuracy and precision? -What information is needed to calculate the density of a ...
... -How do you practice safety in the laboratory? -What units are used in science for measurement? -What are metric prefixes and how are they used? -What causes uncertainty in measurements? -What is the difference between accuracy and precision? -What information is needed to calculate the density of a ...
Document
... The thermochemical equation is the chemical equation for a reaction (including phase labels) in which the equation is given a molar interpretation, and the enthalpy of reaction for these molar amounts is written directly after the equation. For the reaction of sodium metal with water, the thermochem ...
... The thermochemical equation is the chemical equation for a reaction (including phase labels) in which the equation is given a molar interpretation, and the enthalpy of reaction for these molar amounts is written directly after the equation. For the reaction of sodium metal with water, the thermochem ...
Use the following answers for questions 1
... AP MULTIPLE CHOICE PROBLEMS 52. 3 Ag(s) + 4 HNO3 <===> 3 AgNO3 + NO(g) + 2 H2O The reaction of silver metal and dilute nitric acid proceeds according to the equation above. If 0.10 mole of powdered silver is added to 10. milliliters of 6.0-molar nitric acid, the number of moles of NO gas that can be ...
... AP MULTIPLE CHOICE PROBLEMS 52. 3 Ag(s) + 4 HNO3 <===> 3 AgNO3 + NO(g) + 2 H2O The reaction of silver metal and dilute nitric acid proceeds according to the equation above. If 0.10 mole of powdered silver is added to 10. milliliters of 6.0-molar nitric acid, the number of moles of NO gas that can be ...
Chapter 11, Kinetics
... Reactant Concentration and Time Relationship for First Order Reactions 16. The first order rate constant for the decomposition of a certain hormone in water at 25oC is 3.42 x 10-4 day-1. a. If a 0.0200 M solution of the hormone is stored at 25oC for two months, what will its concentration be at the ...
... Reactant Concentration and Time Relationship for First Order Reactions 16. The first order rate constant for the decomposition of a certain hormone in water at 25oC is 3.42 x 10-4 day-1. a. If a 0.0200 M solution of the hormone is stored at 25oC for two months, what will its concentration be at the ...
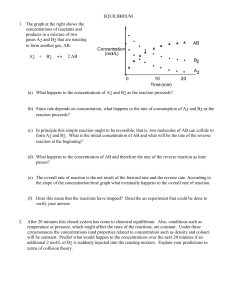
EQUILIBRIUM - SCH4U1-CCVI
... concentrations may be used to calculate the value of the equilibrium constant for this reaction. Then predictions may be made about the equilibrium concentrations in other solutions. To prepare a solution with a known concentration of the coloured complex, a dilute solution of thiocyanate ion is rea ...
... concentrations may be used to calculate the value of the equilibrium constant for this reaction. Then predictions may be made about the equilibrium concentrations in other solutions. To prepare a solution with a known concentration of the coloured complex, a dilute solution of thiocyanate ion is rea ...
A) Sn4+ → Sn2+ + 2e
... activity of the metal temperature of the electrolyte quantity of electrical charge passed through the cell E) molarity of the ions in solution 81. If fused silver chloride, AgCl, is electrolyzed, the Ag+ ions are A) B) C) D) E) ...
... activity of the metal temperature of the electrolyte quantity of electrical charge passed through the cell E) molarity of the ions in solution 81. If fused silver chloride, AgCl, is electrolyzed, the Ag+ ions are A) B) C) D) E) ...
First Semester Final Review
... occurs because on the mountaintop the a. equilibrium water vapor pressure is higher due to the higher atmospheric pressure. b. equilibrium water vapor pressure is lower due to the higher atmospheric pressure. c. equilibrium water vapor pressure equals the atmospheric pressure at a lower temperature ...
... occurs because on the mountaintop the a. equilibrium water vapor pressure is higher due to the higher atmospheric pressure. b. equilibrium water vapor pressure is lower due to the higher atmospheric pressure. c. equilibrium water vapor pressure equals the atmospheric pressure at a lower temperature ...
g - Haiku
... Analyze Mixing solutions of HCl and NaOH results in an acid–base reaction: HCl(aq) + NaOH(aq) → H2O(l) + NaCl(aq) We need to calculate the heat produced per mole of HCl, given the temperature increase of the solution, the number of moles of HCl and NaOH involved, and the density and specific heat of ...
... Analyze Mixing solutions of HCl and NaOH results in an acid–base reaction: HCl(aq) + NaOH(aq) → H2O(l) + NaCl(aq) We need to calculate the heat produced per mole of HCl, given the temperature increase of the solution, the number of moles of HCl and NaOH involved, and the density and specific heat of ...
Chapter 1 – Reaction Kinetics Answer Key
... (the quotient of moles/volume) does not change. For aqueous species, increased numbers of moles in the same volume of solution certainly does increase the concentration. The same is true for gases ...
... (the quotient of moles/volume) does not change. For aqueous species, increased numbers of moles in the same volume of solution certainly does increase the concentration. The same is true for gases ...
[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION
... 1)Chemical reaction— Chemical changes or chemical reactions are the changes in which one or more new substances are formed. 2)Chemical Equations – Representation of a chemical reaction in terms of symbols and formulae of the reactants and products is known as chemical equation. 3)Balanced Chemical e ...
... 1)Chemical reaction— Chemical changes or chemical reactions are the changes in which one or more new substances are formed. 2)Chemical Equations – Representation of a chemical reaction in terms of symbols and formulae of the reactants and products is known as chemical equation. 3)Balanced Chemical e ...
Chemistry - Onslow College
... Writing word equations and balanced chemical equations for inorganic reactions By the end of this topic students will be able to 1. use solubility rules to predict precipitation and identify the precipitate. 2. carry out precipitation reactions and report experimental observations 3. from experime ...
... Writing word equations and balanced chemical equations for inorganic reactions By the end of this topic students will be able to 1. use solubility rules to predict precipitation and identify the precipitate. 2. carry out precipitation reactions and report experimental observations 3. from experime ...
Nanoenergetic Materials for MEMS: A Review - LAAS-CNRS
... and technological ways to tailor EMs to the targeted applications either to produce gas, heat, or chemical compound. An important challenge that is not really addressed is the compatibility of the EMs with MEMS technologies. This implies two exigencies. First, the thin film of EMs must be grown or d ...
... and technological ways to tailor EMs to the targeted applications either to produce gas, heat, or chemical compound. An important challenge that is not really addressed is the compatibility of the EMs with MEMS technologies. This implies two exigencies. First, the thin film of EMs must be grown or d ...
1 3. Molecular mass transport 3.1 Introduction to mass transfer 3.2
... Certain molecules diffuse as molecules, while others which are designated as electrolytes ionize in solutions and diffuse as ions. For example, sodium chloride (NaCl), diffuses in water as ions Na + and Cl-. Though each ions has a different mobility, the electrical neutrality of the solution indicat ...
... Certain molecules diffuse as molecules, while others which are designated as electrolytes ionize in solutions and diffuse as ions. For example, sodium chloride (NaCl), diffuses in water as ions Na + and Cl-. Though each ions has a different mobility, the electrical neutrality of the solution indicat ...
ESO - ENCIGA
... order to be able to predict its behaviour and understand its history. Science is based on systematic experimentation and on observation of natural phenomena to discover facts about them and to formulate laws and principles based on these facts. The organized knowledge that is derived from scientific ...
... order to be able to predict its behaviour and understand its history. Science is based on systematic experimentation and on observation of natural phenomena to discover facts about them and to formulate laws and principles based on these facts. The organized knowledge that is derived from scientific ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.


















![[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION](http://s1.studyres.com/store/data/008930072_1-5a35e1ae8e3204ea88999f1418a93013-300x300.png)



