Chapter 14
... entropy of every pure substance approaches the same value as T 0. K. Third law of thermodynamics: The absolute entropy (S) of a perfect crystal of any pure substance at absolute zero is 0.0 J/mol.K. Because there are standard ways of find the change in entropy for a pure substance as we change th ...
... entropy of every pure substance approaches the same value as T 0. K. Third law of thermodynamics: The absolute entropy (S) of a perfect crystal of any pure substance at absolute zero is 0.0 J/mol.K. Because there are standard ways of find the change in entropy for a pure substance as we change th ...
Massachusetts Tests for Educator Licensure (MTEL ) www.mtel
... This practice test may be used as one indicator of potential strengths and weaknesses in your knowledge of the content on the official test. However, because of potential differences in format and difficulty between the practice test and an official MTEL Chemistry (12) test, it is not possible to pr ...
... This practice test may be used as one indicator of potential strengths and weaknesses in your knowledge of the content on the official test. However, because of potential differences in format and difficulty between the practice test and an official MTEL Chemistry (12) test, it is not possible to pr ...
Oxidation Growth Of SiO2 On Silicon
... combine with silicon to form silicon dioxide. The chemical reactions that take place are either ...
... combine with silicon to form silicon dioxide. The chemical reactions that take place are either ...
Document
... Is this a constant pressure process? What is the system? What are the surroundings? ΔH > 0 endothermic ...
... Is this a constant pressure process? What is the system? What are the surroundings? ΔH > 0 endothermic ...
Alternative Coverage of moles, molarity, and Chemical Calculations
... Like atomic masses, molecular masses are relative masses. A molecule of oxygen, O2, has a mass of 32 u, twice that of a molecule of methane, 16 u. A molecule of ozone has a mass of 48 u, three times that of a molecule of methane. Using the same reasoning we used for atomic substances, we conclude th ...
... Like atomic masses, molecular masses are relative masses. A molecule of oxygen, O2, has a mass of 32 u, twice that of a molecule of methane, 16 u. A molecule of ozone has a mass of 48 u, three times that of a molecule of methane. Using the same reasoning we used for atomic substances, we conclude th ...
Chapter 3
... element the same on both sides of the equation. Do not change the subscripts. 3. Start by balancing those elements that appear in only one reactant and one product. 4. Balance those elements that appear in two or more reactants or products. 4. Remove all fractions (generally by multiplying everythin ...
... element the same on both sides of the equation. Do not change the subscripts. 3. Start by balancing those elements that appear in only one reactant and one product. 4. Balance those elements that appear in two or more reactants or products. 4. Remove all fractions (generally by multiplying everythin ...
Atomic Masses
... Atomic Mass • The balanced Chemical equation for the reaction of solid carbon and gaseous oxygen to form gaseous carbon dioxide is as follows: • Now suppose you have a small pile of carbon, and you want to know how many oxygen molecules are required to C(s) + O2 (g) CO2 (g) convert all this carb ...
... Atomic Mass • The balanced Chemical equation for the reaction of solid carbon and gaseous oxygen to form gaseous carbon dioxide is as follows: • Now suppose you have a small pile of carbon, and you want to know how many oxygen molecules are required to C(s) + O2 (g) CO2 (g) convert all this carb ...
Stench Chemicals Fact Sheet
... bleach trap to a potassium hydroxide trap before venting to the fume hood. The oxidation reaction is exothermic, thus if a large amount of stench chemical is being oxidized then cooling of the traps will be necessary to keep their temperature from rising. The cooling can be achieved by placing the t ...
... bleach trap to a potassium hydroxide trap before venting to the fume hood. The oxidation reaction is exothermic, thus if a large amount of stench chemical is being oxidized then cooling of the traps will be necessary to keep their temperature from rising. The cooling can be achieved by placing the t ...
Chemistry
... 2000) will generally be adopted although the traditional names sulfate, sulfite, nitrate, nitrite, sulfurous and nitrous acids will be used in question papers. Sulfur (and all compounds of sulfur) will be spelt with f (not with ph) in question papers, however candidates can use either spelling in th ...
... 2000) will generally be adopted although the traditional names sulfate, sulfite, nitrate, nitrite, sulfurous and nitrous acids will be used in question papers. Sulfur (and all compounds of sulfur) will be spelt with f (not with ph) in question papers, however candidates can use either spelling in th ...
6 Grade Math
... Students complete Topic A by solidifying the theorem that if two ratios are equivalent, then they have the same value. Example Problem and Solution Drew Brees was at a practice session for the Pro-Bowl. He threw 40 passes but only completed 25. Describe and write more than one ratio related to this ...
... Students complete Topic A by solidifying the theorem that if two ratios are equivalent, then they have the same value. Example Problem and Solution Drew Brees was at a practice session for the Pro-Bowl. He threw 40 passes but only completed 25. Describe and write more than one ratio related to this ...
Titration #1 Determination of [NaOH]
... To start the reaction, add some distilled water to the beaker containing the CuCl2 and the Al foil, as directed by the teacher. Don’t add too much water. You may stir the solution with a glass rod. ...
... To start the reaction, add some distilled water to the beaker containing the CuCl2 and the Al foil, as directed by the teacher. Don’t add too much water. You may stir the solution with a glass rod. ...
Unit 13: Electrochemistry (Link to Prentice Hall Text: Chapters 22
... Why would you ever want to force a nonspontaneous reaction? (a) To obtain pure metals Many metals are only found as compounds in nature. Electrolysis can lead to a deposit of the pure metal on the cathode. (b) To recharge a battery A car battery powers the car through a spontaneous reaction, but wha ...
... Why would you ever want to force a nonspontaneous reaction? (a) To obtain pure metals Many metals are only found as compounds in nature. Electrolysis can lead to a deposit of the pure metal on the cathode. (b) To recharge a battery A car battery powers the car through a spontaneous reaction, but wha ...
Ch 16 Power Point
... and the processes of heat transfer 11C use thermochemical equations to calculate energy changes that occur in chemical reactions and classify reactions as exothermic or ...
... and the processes of heat transfer 11C use thermochemical equations to calculate energy changes that occur in chemical reactions and classify reactions as exothermic or ...
Lecture 1 Atomic Structure
... Discharge tube experiments provided strong evidence for the existence of subatomic particles. A discharge tube is a glass tube having two electrodes sealed in at each end. It is connected to a high voltage battery to provide required voltage and to a vacuum pump to evacuate air or gas from the tube. ...
... Discharge tube experiments provided strong evidence for the existence of subatomic particles. A discharge tube is a glass tube having two electrodes sealed in at each end. It is connected to a high voltage battery to provide required voltage and to a vacuum pump to evacuate air or gas from the tube. ...
PREPARMACY PHYSICAL CHEMISTRY THERMOCHEMISTRY
... number of moles of reactants as represented by the balanced chemical equation change completely into the products. For example, the heat change for the reaction of one mole of carbon monoxide with 0.5 mole of oxygen to form one mole of carbon dioxide is -284.5 KJ. This means that 284.5 KJ of heat is ...
... number of moles of reactants as represented by the balanced chemical equation change completely into the products. For example, the heat change for the reaction of one mole of carbon monoxide with 0.5 mole of oxygen to form one mole of carbon dioxide is -284.5 KJ. This means that 284.5 KJ of heat is ...
Learning Outcomes Leaving Certificate Chemistry
... (examples should include simple biological substances, such as glucose and urea) calculate percentage composition by mass define structural formula deduce, describe and explain structural formulas (simple examples) 3.5 Chemical Equations (11 class periods) By the end of this section pupils should be ...
... (examples should include simple biological substances, such as glucose and urea) calculate percentage composition by mass define structural formula deduce, describe and explain structural formulas (simple examples) 3.5 Chemical Equations (11 class periods) By the end of this section pupils should be ...
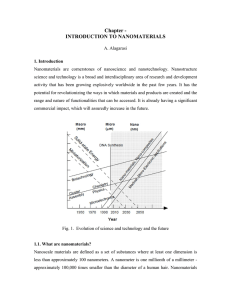
Chapter - INTRODUCTION TO NANOMATERIALS
... powder mass, the time of milling and the milling atmosphere. Nanoparticles are produced by the shear action during grinding. Milling in cryogenic liquids can greatly increase the brittleness of the powders influencing the fracture process. As with any process that produces fine particles, an adequat ...
... powder mass, the time of milling and the milling atmosphere. Nanoparticles are produced by the shear action during grinding. Milling in cryogenic liquids can greatly increase the brittleness of the powders influencing the fracture process. As with any process that produces fine particles, an adequat ...
Slides
... •Depth First Search up to “height” deep •‘(‘ going down, ‘)’ going back up [email protected] ...
... •Depth First Search up to “height” deep •‘(‘ going down, ‘)’ going back up [email protected] ...
Answers - Pearson-Global
... break; weaker forces between layers in graphite allow layers to slide); graphite less dense than diamond (comparatively large distances between the layers in graphite mean that less atoms can be fitted into a given volume); graphite conducts electricity, diamond doesn’t (each carbon in graphite form ...
... break; weaker forces between layers in graphite allow layers to slide); graphite less dense than diamond (comparatively large distances between the layers in graphite mean that less atoms can be fitted into a given volume); graphite conducts electricity, diamond doesn’t (each carbon in graphite form ...
Physical Chemistry 3: — Chemical Kinetics
... The scriptum gives a summary of the material covered in the scheduled lectures to allow students to repeat the material more economically. It covers basic material that all chemistry students should learn irrespective of their possible inclination towards inorganic, organic or physical chemistry, bu ...
... The scriptum gives a summary of the material covered in the scheduled lectures to allow students to repeat the material more economically. It covers basic material that all chemistry students should learn irrespective of their possible inclination towards inorganic, organic or physical chemistry, bu ...
X PS EM - deo kadapa
... Ans:- Photosynthesis is an endothermic reaction as it takes energy from sun. 3. Why should a magnesium ribbon cleaned before burning in air? Ans:- Magnesium ribbon should be cleaned before burning in air, because it forms magnesium oxide by reacting with oxygen present in air. This oxide layer shoul ...
... Ans:- Photosynthesis is an endothermic reaction as it takes energy from sun. 3. Why should a magnesium ribbon cleaned before burning in air? Ans:- Magnesium ribbon should be cleaned before burning in air, because it forms magnesium oxide by reacting with oxygen present in air. This oxide layer shoul ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.










![Titration #1 Determination of [NaOH]](http://s1.studyres.com/store/data/006790962_1-275eb40711c8f3ecc850bd432b1a775b-300x300.png)