Section 16.1 A Model for Reaction Rates
... • Q5: Explain why the average rate of a reaction depends on the length of the time interval over which the rate is measured. • The rate of change of a reactant or product in a chemical reaction is not linear in time • Q6: Describe the relationship between activation energy and the rate of a reactio ...
... • Q5: Explain why the average rate of a reaction depends on the length of the time interval over which the rate is measured. • The rate of change of a reactant or product in a chemical reaction is not linear in time • Q6: Describe the relationship between activation energy and the rate of a reactio ...
Answer Key to Sample Questions
... positive because one molecule breaks to form two molecules b. What is the sign of H for this reaction? positive because a bond is broken, but none is formed. c. In which temperature range will this reaction be thermodynamically favored? It is entropy favored, enthalpy disfavored, so favored overall ...
... positive because one molecule breaks to form two molecules b. What is the sign of H for this reaction? positive because a bond is broken, but none is formed. c. In which temperature range will this reaction be thermodynamically favored? It is entropy favored, enthalpy disfavored, so favored overall ...
O 2
... sort of manageable scheme. There are 5 general types of chemical reactions that Make up this scheme ...
... sort of manageable scheme. There are 5 general types of chemical reactions that Make up this scheme ...
File
... 55. Which pair of solutions forms a buffer when equal volumes of each are mixed? A) 0.20 M HCl and 0.20 M NaCl C) 0.20 M HCl and 0.20 M NH3 B) 0.40 M HC2H3O2 and 0.20 M NaOH D) 0.40 M HCl and 0.20 M NH3 56. A student is attempting to standardize a NaOH solution with a 0.500 molar solution of oxalic ...
... 55. Which pair of solutions forms a buffer when equal volumes of each are mixed? A) 0.20 M HCl and 0.20 M NaCl C) 0.20 M HCl and 0.20 M NH3 B) 0.40 M HC2H3O2 and 0.20 M NaOH D) 0.40 M HCl and 0.20 M NH3 56. A student is attempting to standardize a NaOH solution with a 0.500 molar solution of oxalic ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... 35. Write and balance the chemical equations for the following reactions. Include the physical states (aq, s, l, g) of the reactants and products. a. When zinc metal and sulfur powder are heated, they form solid zinc sulfide. b. When sodium metal is placed in a beaker of water, hydrogen gas and sodi ...
... 35. Write and balance the chemical equations for the following reactions. Include the physical states (aq, s, l, g) of the reactants and products. a. When zinc metal and sulfur powder are heated, they form solid zinc sulfide. b. When sodium metal is placed in a beaker of water, hydrogen gas and sodi ...
Stoichiometry Notes
... b. Recall that the molar mass of a compound is the mass, in grams, of one mole of that compound. determine the molar mass of a compound, add together the molar mass of all of the elements in the compound. Practice problems: 3. What is the molar mass of nitrogen dioxide? ...
... b. Recall that the molar mass of a compound is the mass, in grams, of one mole of that compound. determine the molar mass of a compound, add together the molar mass of all of the elements in the compound. Practice problems: 3. What is the molar mass of nitrogen dioxide? ...
Click here for the Reaction NOTES Handout
... The key is first to write the compound’s chemical formula and then determine if it’s soluble. If it is soluble, then ionize it—if it isn’t, don’t ionize it; leave it as a “molecule”. Check the CRC handbook (Table of Ionic compounds) to see what color any insoluble compounds are (precipitates). Now t ...
... The key is first to write the compound’s chemical formula and then determine if it’s soluble. If it is soluble, then ionize it—if it isn’t, don’t ionize it; leave it as a “molecule”. Check the CRC handbook (Table of Ionic compounds) to see what color any insoluble compounds are (precipitates). Now t ...
Physical Science Chapter 7 Chemical Reactions Section 7.1
... A ______________________________________________________________________ is called an exothermic reaction. In exothermic reactions, the ________________________________________as the products form __________________________________________________ to break the bonds in the reactants. Combustion is _ ...
... A ______________________________________________________________________ is called an exothermic reaction. In exothermic reactions, the ________________________________________as the products form __________________________________________________ to break the bonds in the reactants. Combustion is _ ...
Chapter 2 - Speedway High School
... • An element is a substance that cannot be broken down to other substances by chemical reactions • A compound is a substance consisting of two or more elements in a fixed ratio ...
... • An element is a substance that cannot be broken down to other substances by chemical reactions • A compound is a substance consisting of two or more elements in a fixed ratio ...
82KB - NZQA
... releases a colourless gas, carbon dioxide, CO2, and forms another white solid calcium oxide, CaO. CaCO3(s) → CaO(s) + CO2(g) When white calcium hydroxide solid, Ca(OH)2, is strongly heated it releases a colourless gas, water, H2O, and also forms the white solid calcium oxide, CaO. Ca(OH)2(s) → CaO(s ...
... releases a colourless gas, carbon dioxide, CO2, and forms another white solid calcium oxide, CaO. CaCO3(s) → CaO(s) + CO2(g) When white calcium hydroxide solid, Ca(OH)2, is strongly heated it releases a colourless gas, water, H2O, and also forms the white solid calcium oxide, CaO. Ca(OH)2(s) → CaO(s ...
unit 7a homework packet - District 196 e
... 9. 132 grams of sodium reacts vigorously with excess water (hydrogen hydroxide) in a single displacement reactant. What volume of hydrogen gas is produced? (Hint: Write the balanced chemical equation for the reaction. Then use the mole train to solve the problem.) ...
... 9. 132 grams of sodium reacts vigorously with excess water (hydrogen hydroxide) in a single displacement reactant. What volume of hydrogen gas is produced? (Hint: Write the balanced chemical equation for the reaction. Then use the mole train to solve the problem.) ...
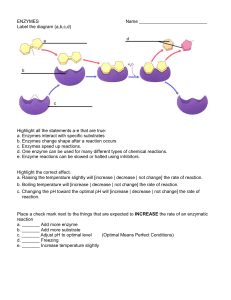
ENZYMES
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
Review for test, Chapter 9
... the changes in temperature of this sample. What is the melting point of the sample and the total time required to completely melt the sample after it has reached its melting point? ...
... the changes in temperature of this sample. What is the melting point of the sample and the total time required to completely melt the sample after it has reached its melting point? ...
South Pasadena · AP Chemistry
... 13. Sodium metal reacts vigorously with water to produce a solution of sodium hydroxide and hydrogen gas: 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g) What mass of hydrogen gas can be produced when 10 grams of sodium is added to 15 grams of water? 14. Nitrous oxide reacts with oxygen to produce nitrogen dio ...
... 13. Sodium metal reacts vigorously with water to produce a solution of sodium hydroxide and hydrogen gas: 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g) What mass of hydrogen gas can be produced when 10 grams of sodium is added to 15 grams of water? 14. Nitrous oxide reacts with oxygen to produce nitrogen dio ...
Journal of Physical and Chemical Reference Data
... number of ways and it is therefore literally impossible to catalog all the possible heats of reaction. To get around this problem we define for each substance a standard reaction and tabulate its associated heat of reaction. These reactions and their associated heats of reaction can then be used to ...
... number of ways and it is therefore literally impossible to catalog all the possible heats of reaction. To get around this problem we define for each substance a standard reaction and tabulate its associated heat of reaction. These reactions and their associated heats of reaction can then be used to ...
Endothermic And Exothermic Reactions
... A chemical reaction that absorbs energy from its surroundings. More energy is required to break the bonds in the reactants than is released by the formation of bonds in the products. In these reactions, heat is shown as one This is a typical graph of an of the reactants endothermic reaction with the ...
... A chemical reaction that absorbs energy from its surroundings. More energy is required to break the bonds in the reactants than is released by the formation of bonds in the products. In these reactions, heat is shown as one This is a typical graph of an of the reactants endothermic reaction with the ...
CHEMISTRY
... (3) Solution of Br2 in CCI4 (4) Conc. H2SO4 51. When 50 cm2 of a strong acid is added to 50cm3 of an alkali, the temperature rises by 50C. If 250 cm3 of each liquid are mixed, the temperature rise would be: ...
... (3) Solution of Br2 in CCI4 (4) Conc. H2SO4 51. When 50 cm2 of a strong acid is added to 50cm3 of an alkali, the temperature rises by 50C. If 250 cm3 of each liquid are mixed, the temperature rise would be: ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.