CHEMISTRY I Final..#1..rev 4KEY
... Objective 3.03: Calculate quantitative relationships in chemical relationships (stoichiometry) ...
... Objective 3.03: Calculate quantitative relationships in chemical relationships (stoichiometry) ...
precipitation rxn_level_packet
... Directions for the following 4 reactions: a. In one well of a well-plate, add three drops of each substance. b. Write down your observations for the reactants above. c. In parenthesis provided above, indicate if the product is soluble with an “aq” or forms a precipitate (solid) with an “s.” 1. Write ...
... Directions for the following 4 reactions: a. In one well of a well-plate, add three drops of each substance. b. Write down your observations for the reactants above. c. In parenthesis provided above, indicate if the product is soluble with an “aq” or forms a precipitate (solid) with an “s.” 1. Write ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... The empirical formula of a compound is the simplest whole number ratio of atoms of each element in a molecule. Formula units for ionic compounds are equal to their empirical formulas. To determine the EF, given mass % or masses of each element in a compound: 1. Convert masses to moles. 2. Find the m ...
... The empirical formula of a compound is the simplest whole number ratio of atoms of each element in a molecule. Formula units for ionic compounds are equal to their empirical formulas. To determine the EF, given mass % or masses of each element in a compound: 1. Convert masses to moles. 2. Find the m ...
chemistry 11 exam review
... 16. What amount of oxygen, in moles, is available for a combustion reaction in a volume of 12.5 L at STP? 17. Potassium metal with a total mass of 15.0 g is dropped into a beaker of water. What volume of hydrogen gas will be produced if the temperature is 15ºC and the pressure is 100 kPa? Chemical e ...
... 16. What amount of oxygen, in moles, is available for a combustion reaction in a volume of 12.5 L at STP? 17. Potassium metal with a total mass of 15.0 g is dropped into a beaker of water. What volume of hydrogen gas will be produced if the temperature is 15ºC and the pressure is 100 kPa? Chemical e ...
use-2012_review_sheettest_form_c_reactions
... d) 3 Mg + 2 FeBr3 Fe2Mg3 + 3 Br2 e) N + H NH3 f) NaBr + CaF2 --> NaCa + FBr g) ...
... d) 3 Mg + 2 FeBr3 Fe2Mg3 + 3 Br2 e) N + H NH3 f) NaBr + CaF2 --> NaCa + FBr g) ...
sch3u unit 1 test: matter
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...
I, I, I, 4- Measurement Unit Conversions- Kilo
... atoms the largest ionization energy. As you go down a family/group (column), the atomic radius gets larger because you add in another energy level for the electrons for each space you go down and the ionization energy becomes less. (Think about a football player running with the ball. If he holds it ...
... atoms the largest ionization energy. As you go down a family/group (column), the atomic radius gets larger because you add in another energy level for the electrons for each space you go down and the ionization energy becomes less. (Think about a football player running with the ball. If he holds it ...
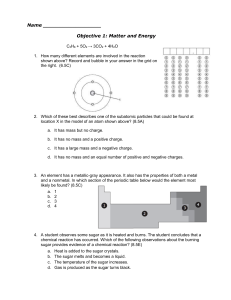
Name Objective 1: Matter and Energy C3H8 + 5O2 → 3CO2 + 4H2O
... a. C3H8 and C2H6 b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorgan ...
... a. C3H8 and C2H6 b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorgan ...
Chemistry Unit Test Review
... 4 carbon atoms and 10 hydrogen atoms 8 carbon atoms and 20 hydrogen atoms 8 carbon atoms and 10 hydrogen atoms 4 carbon atoms and 20 hydrogen atoms ...
... 4 carbon atoms and 10 hydrogen atoms 8 carbon atoms and 20 hydrogen atoms 8 carbon atoms and 10 hydrogen atoms 4 carbon atoms and 20 hydrogen atoms ...
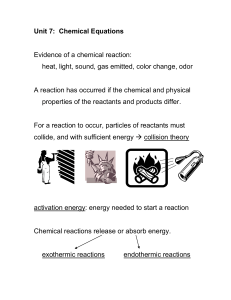
Chemical Equations
... The products are on the right side and show what was produced by the reaction. The arrow is like an = sign, but shows the direction of the reaction. The coefficients show the ratio in which the substances ...
... The products are on the right side and show what was produced by the reaction. The arrow is like an = sign, but shows the direction of the reaction. The coefficients show the ratio in which the substances ...
chemeqohnotes18f2005
... Examples: enzymes catalyze biochemical reactions catalytic converters convert CO into CO2 ...
... Examples: enzymes catalyze biochemical reactions catalytic converters convert CO into CO2 ...
Chemistry 432: Final Exam Review Sheet
... 3. Calculation molarity (M), molality (m), freezing point depression (Tf), and boiling point elevation (Tb). 4. Gas law calculations using combined gas law, Dalton's law, ideal gas law, Graham’s Law. 5. Stoichiometric calculations involving mass, volume, and particle relationships between substanc ...
... 3. Calculation molarity (M), molality (m), freezing point depression (Tf), and boiling point elevation (Tb). 4. Gas law calculations using combined gas law, Dalton's law, ideal gas law, Graham’s Law. 5. Stoichiometric calculations involving mass, volume, and particle relationships between substanc ...
Review Notes - Biochemistry
... gained it will be _NEGATIVE_charged and when an electron is lost it will be _POSITIVE_ charged. ...
... gained it will be _NEGATIVE_charged and when an electron is lost it will be _POSITIVE_ charged. ...
exam review - hrsbstaff.ednet.ns.ca
... b) Determine the volume of commercial grade nitric acid (15.8 mol/L HNO 3) that is needed to prepare 100.0 mL of 3.00 mol/L HNO3. c) Rubbing alcohol is commonly used as an antiseptic for small cuts. It is sold as a 23% (v/v) solution of isopropyl alcohol in water. What volume of isopropyl alcohol is ...
... b) Determine the volume of commercial grade nitric acid (15.8 mol/L HNO 3) that is needed to prepare 100.0 mL of 3.00 mol/L HNO3. c) Rubbing alcohol is commonly used as an antiseptic for small cuts. It is sold as a 23% (v/v) solution of isopropyl alcohol in water. What volume of isopropyl alcohol is ...
SOL Essential Knowledge
... 2. A mole is a way of counting any type of particle (atoms, molecules, formula units). 3. Stoichiometry involves quantitative relationships. 4. Stoichiometric relationships are based on mole quantities in a balanced equation. B. Know and use: 1. Avogadro’s number (6.02 x 1023 particles = 1 mole) 2. ...
... 2. A mole is a way of counting any type of particle (atoms, molecules, formula units). 3. Stoichiometry involves quantitative relationships. 4. Stoichiometric relationships are based on mole quantities in a balanced equation. B. Know and use: 1. Avogadro’s number (6.02 x 1023 particles = 1 mole) 2. ...
Chemistry - Beachwood City Schools
... 1. What is a chemical bond? Why do atoms form chemical bonds? How are covalent bonds and ionic bonds different? How are they the same? 2. How is the valence of an atom related to the number of bonds it usually forms? 3. What types of substances contain covalent bonds? 4. List the atoms in each of th ...
... 1. What is a chemical bond? Why do atoms form chemical bonds? How are covalent bonds and ionic bonds different? How are they the same? 2. How is the valence of an atom related to the number of bonds it usually forms? 3. What types of substances contain covalent bonds? 4. List the atoms in each of th ...
Chemistry EOC Review 2015 Name Per ___ This review is part of
... the number of each type of atom must be the same on each side of the equation. It also means that you must have the same type of atoms on each side of the reaction. The coefficients in the reaction tell you the constant ratio of the reactants and products. Use the REP chart to help balance equations ...
... the number of each type of atom must be the same on each side of the equation. It also means that you must have the same type of atoms on each side of the reaction. The coefficients in the reaction tell you the constant ratio of the reactants and products. Use the REP chart to help balance equations ...
Ch 2 notes
... 3. From combustion data: A 0.2500g sample of a hydrocarbon containing only carbon, hydrogen, and oxygen was combusted and produced 0.3664 g carbon dioxide and 0.1500 g water. What is the empirical formula of this hydrocarbon? ...
... 3. From combustion data: A 0.2500g sample of a hydrocarbon containing only carbon, hydrogen, and oxygen was combusted and produced 0.3664 g carbon dioxide and 0.1500 g water. What is the empirical formula of this hydrocarbon? ...
CHM_101_ASSIGNMENT_COPY_1_2
... Calculate the pressure equilibrium constant Kp for the reaction at this temperature. What is the partial pressure of chlorine in the vessel? 5. Write the expressions for the concentration equilibrium constant Kc and pressure equilibrium constant Kp for the following reactions: a) ...
... Calculate the pressure equilibrium constant Kp for the reaction at this temperature. What is the partial pressure of chlorine in the vessel? 5. Write the expressions for the concentration equilibrium constant Kc and pressure equilibrium constant Kp for the following reactions: a) ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.