Enzyme Shape
... a direct effect on how it catalyzes a reaction. Why do enzymes have different shapes? An enzyme’s shape is determined by the sequence of amino acids in its structure, and the bonds which form between the atoms of those molecules. Different types of enzymes have different shapes and functions because ...
... a direct effect on how it catalyzes a reaction. Why do enzymes have different shapes? An enzyme’s shape is determined by the sequence of amino acids in its structure, and the bonds which form between the atoms of those molecules. Different types of enzymes have different shapes and functions because ...
9. AH Cell Enzymes - charlestonbiology
... Metabolism is organised as a series of metabolic pathways, and control of these pathways is an important feature of cell biochemistry End-product inhibition is energetically efficient as it avoids the excessive (and wasteful) production of the intermediates of a pathway This is a form of NEGATIVE FE ...
... Metabolism is organised as a series of metabolic pathways, and control of these pathways is an important feature of cell biochemistry End-product inhibition is energetically efficient as it avoids the excessive (and wasteful) production of the intermediates of a pathway This is a form of NEGATIVE FE ...
Allosteric Regulation of an Enzyme
... Michealis-Menton Enzyme Kinetics refers to how fast (quantity/time) an enzyme catalyzes a particular chemical reaction! There are special rules that apply inside a cell! • Velocity: [P]/time! • Initial Reaction Velocity: speed at start! • Saturation: When an enzyme can’t handle ANY more substrate p ...
... Michealis-Menton Enzyme Kinetics refers to how fast (quantity/time) an enzyme catalyzes a particular chemical reaction! There are special rules that apply inside a cell! • Velocity: [P]/time! • Initial Reaction Velocity: speed at start! • Saturation: When an enzyme can’t handle ANY more substrate p ...
1. Vmax, the maximum velocity, of an enzyme-catalyzed
... b. noncompetitive inhibitor. c. uncompetitive inhibitor. d. irreversible inhibitor. 4. Diisopropyl fluorophosphate (DIFP) inactivates chymotrypsin by covalently modifying serine-195. This occurs because a. ...
... b. noncompetitive inhibitor. c. uncompetitive inhibitor. d. irreversible inhibitor. 4. Diisopropyl fluorophosphate (DIFP) inactivates chymotrypsin by covalently modifying serine-195. This occurs because a. ...
Enzymes
... These are the opposite of activators. Inhibitors either slow down or stop the activity of an enzyme. They often bond to the protein and the shape changes. Remember - When the shape changes, the enzyme will not work the same way. A nasty example of an inhibitor is snake venom or maybe nerve gas from ...
... These are the opposite of activators. Inhibitors either slow down or stop the activity of an enzyme. They often bond to the protein and the shape changes. Remember - When the shape changes, the enzyme will not work the same way. A nasty example of an inhibitor is snake venom or maybe nerve gas from ...
Enzymes worksheet
... You need a key that is just the right shape to fit in that lock. Otherwise you're stuck in the cold. Enzymes work in a similar way. Enzymes complete very specific jobs and do nothing else. They are very specific locks and the compounds they work with are the special keys. Here's the deal... There ar ...
... You need a key that is just the right shape to fit in that lock. Otherwise you're stuck in the cold. Enzymes work in a similar way. Enzymes complete very specific jobs and do nothing else. They are very specific locks and the compounds they work with are the special keys. Here's the deal... There ar ...
Enzymes
... You need a key that is just the right shape to fit in that lock. Otherwise you're stuck in the cold. Enzymes work in a similar way. Enzymes complete very specific jobs and do nothing else. They are very specific locks and the compounds they work with are the special keys. Here's the deal... There ar ...
... You need a key that is just the right shape to fit in that lock. Otherwise you're stuck in the cold. Enzymes work in a similar way. Enzymes complete very specific jobs and do nothing else. They are very specific locks and the compounds they work with are the special keys. Here's the deal... There ar ...
Enzymes - Solon City Schools
... of the chemical bonds that maintain protein structure b. pH: The same is true of the extra hydrogen ions at very low pH 1. Optimal pH for most enzymes near neutral ...
... of the chemical bonds that maintain protein structure b. pH: The same is true of the extra hydrogen ions at very low pH 1. Optimal pH for most enzymes near neutral ...
Exam practice answers
... molecules are broken. The active site changes shape and the starch no longer fits in the active site. The amylase is denatured. ...
... molecules are broken. The active site changes shape and the starch no longer fits in the active site. The amylase is denatured. ...
Getting the inside running on enzymes
... “There is a possibility of using advanced protein engineering techniques for sensing applications or understanding the way in which we can turn molecules or biochemical pathways on and off. These findings may also inform the design of new antibiotic therapies.” chemical reactions and the rate at whi ...
... “There is a possibility of using advanced protein engineering techniques for sensing applications or understanding the way in which we can turn molecules or biochemical pathways on and off. These findings may also inform the design of new antibiotic therapies.” chemical reactions and the rate at whi ...
Three Dimensional Protein Structures
... and the catalyzed rate is as fast as molecules collide. ...
... and the catalyzed rate is as fast as molecules collide. ...
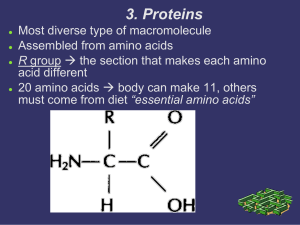
3. Proteins
... Increase the rate of chemical reactions without being used in the reaction Lower the amount of energy required to initiate the reaction ...
... Increase the rate of chemical reactions without being used in the reaction Lower the amount of energy required to initiate the reaction ...
enzymes 194 kb enzymes
... Long term control can be achieved with by genetic control. Excess tryptophan represses the production of all the enzymes in the tryptophan biosynthesis pathway, using negative feedback to prevent the cell wasting energy. Allosteric enzymes are very important and do not obey Michaelis-Menten kinetics ...
... Long term control can be achieved with by genetic control. Excess tryptophan represses the production of all the enzymes in the tryptophan biosynthesis pathway, using negative feedback to prevent the cell wasting energy. Allosteric enzymes are very important and do not obey Michaelis-Menten kinetics ...
Enzyme Activity
... necessary way of making sure that the reaction does not proceed too fast, at other times, it is undesirable ...
... necessary way of making sure that the reaction does not proceed too fast, at other times, it is undesirable ...
Enzymes - myndrs.com
... • Endergonic - Refers to a chemical reaction that consumes energy. (anabolic) • Exergonic - Describes a chemical reaction that releases energy in the form of heat, light, etc. (catabolic) ...
... • Endergonic - Refers to a chemical reaction that consumes energy. (anabolic) • Exergonic - Describes a chemical reaction that releases energy in the form of heat, light, etc. (catabolic) ...
Enzymes are proteins which control biochemical reactions in cells
... o Enzymes catalyse a reaction at max. rate at an optimum state * Induced fit theory o Enzyme's shape changes when substrate binds to active site o Amino acids are moulded into a precise form to perform catalytic reaction effectively o Enzyme wraps around substrate to distort it o Forms an enzyme-sub ...
... o Enzymes catalyse a reaction at max. rate at an optimum state * Induced fit theory o Enzyme's shape changes when substrate binds to active site o Amino acids are moulded into a precise form to perform catalytic reaction effectively o Enzyme wraps around substrate to distort it o Forms an enzyme-sub ...
Slide 1
... • Enzymes are biological molecules that catalyze(i.e., increase the rates of) chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need ...
... • Enzymes are biological molecules that catalyze(i.e., increase the rates of) chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need ...
Enzyme Kinetics Lab
... Enzymes are a class of proteins that greatly speed up (catalyze) reactions between specific substances, usually at their functional groups. The substances that each type of enzyme acts upon are called its substrates. Enzymes have four common features: 1. They don’t make anything happen that couldn’t ...
... Enzymes are a class of proteins that greatly speed up (catalyze) reactions between specific substances, usually at their functional groups. The substances that each type of enzyme acts upon are called its substrates. Enzymes have four common features: 1. They don’t make anything happen that couldn’t ...
Aim: Why are Enzymes necessary for our survival?
... Is formed when the substrate is bound to the enzyme ...
... Is formed when the substrate is bound to the enzyme ...
Enzyme
... degree of cooperativity and the more sigmoidal will be the plot of vi versus [S]. • A perpendicular dropped from the point where the y term log vi/(Vmax − vi) is zero intersects the x axis at a substrate concentration termed S50, • the substrate concentration that results in halfmaximal velocity. • ...
... degree of cooperativity and the more sigmoidal will be the plot of vi versus [S]. • A perpendicular dropped from the point where the y term log vi/(Vmax − vi) is zero intersects the x axis at a substrate concentration termed S50, • the substrate concentration that results in halfmaximal velocity. • ...
CHM 365 Name: Exam 2 Oct. 13, 2004 Do all of the questions. Part I
... a) Enzymes can be inhibited by the products they produce. b) Enzymes can be inactivated by the addition of a functional group. c) Coenzyme and substrate availability can regulate enzyme reaction rate. d) The reaction rate slows as equilibrium is approached. e) The activity of an enzyme is covalently ...
... a) Enzymes can be inhibited by the products they produce. b) Enzymes can be inactivated by the addition of a functional group. c) Coenzyme and substrate availability can regulate enzyme reaction rate. d) The reaction rate slows as equilibrium is approached. e) The activity of an enzyme is covalently ...
ENZYMES - Bio12.com
... The are usually specific and they work at low concentrations They block the enzyme but they do not usually destroy it Many drugs and poisons are inhibitors of enzymes in the nervous system ...
... The are usually specific and they work at low concentrations They block the enzyme but they do not usually destroy it Many drugs and poisons are inhibitors of enzymes in the nervous system ...
Aim: Why are Enzymes necessary for our survival?
... What is a Catalyst? A catalyst is any substance that speeds up a chemical reaction without itself being changed. ...
... What is a Catalyst? A catalyst is any substance that speeds up a chemical reaction without itself being changed. ...
ENZYMES
... Some substances can inhibit enzyme function – Inhibitors Some substances can enhance enzyme function – activators ...
... Some substances can inhibit enzyme function – Inhibitors Some substances can enhance enzyme function – activators ...
Enzyme inhibitor

An enzyme inhibitor is a molecule that binds to an enzyme and decreases its activity. Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used in pesticides. Not all molecules that bind to enzymes are inhibitors; enzyme activators bind to enzymes and increase their enzymatic activity, while enzyme substrates bind and are converted to products in the normal catalytic cycle of the enzyme.The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction. Inhibitor binding is either reversible or irreversible. Irreversible inhibitors usually react with the enzyme and change it chemically (e.g. via covalent bond formation). These inhibitors modify key amino acid residues needed for enzymatic activity. In contrast, reversible inhibitors bind non-covalently and different types of inhibition are produced depending on whether these inhibitors bind to the enzyme, the enzyme-substrate complex, or both.Many drug molecules are enzyme inhibitors, so their discovery and improvement is an active area of research in biochemistry and pharmacology. A medicinal enzyme inhibitor is often judged by its specificity (its lack of binding to other proteins) and its potency (its dissociation constant, which indicates the concentration needed to inhibit the enzyme). A high specificity and potency ensure that a drug will have few side effects and thus low toxicity.Enzyme inhibitors also occur naturally and are involved in the regulation of metabolism. For example, enzymes in a metabolic pathway can be inhibited by downstream products. This type of negative feedback slows the production line when products begin to build up and is an important way to maintain homeostasis in a cell. Other cellular enzyme inhibitors are proteins that specifically bind to and inhibit an enzyme target. This can help control enzymes that may be damaging to a cell, like proteases or nucleases. A well-characterised example of this is the ribonuclease inhibitor, which binds to ribonucleases in one of the tightest known protein–protein interactions. Natural enzyme inhibitors can also be poisons and are used as defences against predators or as ways of killing prey.