Enzyme Foldable
... a. Draw and label the enzyme and the substrate. Explain the role of an enzyme in the body. 3. Enzyme Vocabulary a. These are the key words in the reading packet. 4. Graphs a. Draw and label the 4 graphs associated with enzymes. i. Temperature ii. PH iii. Concentrations iv. Activation Energy b. Write ...
... a. Draw and label the enzyme and the substrate. Explain the role of an enzyme in the body. 3. Enzyme Vocabulary a. These are the key words in the reading packet. 4. Graphs a. Draw and label the 4 graphs associated with enzymes. i. Temperature ii. PH iii. Concentrations iv. Activation Energy b. Write ...
Enzymes HW Key
... 1. What is the difference between the lock and key hypothesis and the induced fit model? Lock and key is that the enzyme and substrate are rigid and have complementary shapes and fit together like a lock and a key. The induced fit hypothesis is more like a hand in a glove. Both enzyme and substrate ...
... 1. What is the difference between the lock and key hypothesis and the induced fit model? Lock and key is that the enzyme and substrate are rigid and have complementary shapes and fit together like a lock and a key. The induced fit hypothesis is more like a hand in a glove. Both enzyme and substrate ...
Non-competitive
... May be a metal ion such as Zn2+ of Mg2+ May also be an organic molecule such as vitamin B or heme – called a coenzyme Substrate – the molecule an enzyme acts on Activation – any process that initiates or increases the action of an enzyme Inhibition – any process that inactivates an enzyme or reduces ...
... May be a metal ion such as Zn2+ of Mg2+ May also be an organic molecule such as vitamin B or heme – called a coenzyme Substrate – the molecule an enzyme acts on Activation – any process that initiates or increases the action of an enzyme Inhibition – any process that inactivates an enzyme or reduces ...
Enzymes in Action Kit – In Brief
... enzymatic action by binding to another part of the enzyme. This second site, known as the allosteric site, is the place on an enzyme where a molecule that is not a substrate may bind, thus changing the shape of the enzyme and influencing its ability to be active. ...
... enzymatic action by binding to another part of the enzyme. This second site, known as the allosteric site, is the place on an enzyme where a molecule that is not a substrate may bind, thus changing the shape of the enzyme and influencing its ability to be active. ...
Reversible Competitive Inhibitor
... Of the three types of reversible inhibitors, we will focus most on competitive inhibitors. Competitive inhibitors are very common in medicinal chemistry. The reason is because if you know the natural substrate of an enzyme, then you also know the same of a molecule that binds the enzyme's active sit ...
... Of the three types of reversible inhibitors, we will focus most on competitive inhibitors. Competitive inhibitors are very common in medicinal chemistry. The reason is because if you know the natural substrate of an enzyme, then you also know the same of a molecule that binds the enzyme's active sit ...
7.6 Enzymes – summary of mark schemes
... shape / (intramolecular) bonding / conformation of the protein / enzyme is altered; shape / properties of active site altered; substrate no longer fits the active site / no enzyme-substrate / ES complex formed; no enzyme activity / works more slowly (until the inhibitor dissociates); eg CN inhibitio ...
... shape / (intramolecular) bonding / conformation of the protein / enzyme is altered; shape / properties of active site altered; substrate no longer fits the active site / no enzyme-substrate / ES complex formed; no enzyme activity / works more slowly (until the inhibitor dissociates); eg CN inhibitio ...
Characterization of α-galactosidase belonging to family-4 glycoside hidrolases Bacillus halodurans
... protein of 434 amino acids with a predicted molecular weight of 49,761. It was assigned to family 4 of glycoside hidrolases. Almost all of the enzyme was produced as inclusion bodies at 37oC. In order to reduce the expression level, induction temperature was decreased to 20oC so that the enzyme coul ...
... protein of 434 amino acids with a predicted molecular weight of 49,761. It was assigned to family 4 of glycoside hidrolases. Almost all of the enzyme was produced as inclusion bodies at 37oC. In order to reduce the expression level, induction temperature was decreased to 20oC so that the enzyme coul ...
Chapter 4 Enzymes and Energy
... one product binds with the enzyme and prevents it from binding with the substrate. The product may bind with the enzyme at the allosteric site. This is negative feedback. inhibition animation ...
... one product binds with the enzyme and prevents it from binding with the substrate. The product may bind with the enzyme at the allosteric site. This is negative feedback. inhibition animation ...
Enzymes
... 2. Cofactors and Coenzymes • Inorganic substances (zinc, iron) and vitamins (respectively) are sometimes need for proper enzymatic activity. • Example: Iron must be present in hemoglobin in order for it to pick up oxygen. ...
... 2. Cofactors and Coenzymes • Inorganic substances (zinc, iron) and vitamins (respectively) are sometimes need for proper enzymatic activity. • Example: Iron must be present in hemoglobin in order for it to pick up oxygen. ...
8.4 Enzymes speed up metabolic reactions by
... function at one site is affected by the binding of a regulatory molecule to a separate site - results in either inhibition or stimulation of activity Cooperativity: mechanism that amplifies the response of enzymes to substrates Feedback inhibition: a metabolic pathway is switched off by the inhibito ...
... function at one site is affected by the binding of a regulatory molecule to a separate site - results in either inhibition or stimulation of activity Cooperativity: mechanism that amplifies the response of enzymes to substrates Feedback inhibition: a metabolic pathway is switched off by the inhibito ...
Incredible Enzymes - Mrs. Tuma`s Biology Page
... One enzyme (the lock) has a specific shape for one set of substrates (the key) and is very rigid ...
... One enzyme (the lock) has a specific shape for one set of substrates (the key) and is very rigid ...
Enzymeregulation
... Suppose supply of E in cell increases (e.g. eat a meal rich in E). How to shut down synthesis of E? Cell's answer: Enzyme 1 is reversibly inhibited by E. Note that E is not the substrate, and chemically so different that it cannot bind to active site. How does E shut down Enzyme 1? Enz 1 is a specia ...
... Suppose supply of E in cell increases (e.g. eat a meal rich in E). How to shut down synthesis of E? Cell's answer: Enzyme 1 is reversibly inhibited by E. Note that E is not the substrate, and chemically so different that it cannot bind to active site. How does E shut down Enzyme 1? Enz 1 is a specia ...
Investigation of the enzymatic processes depending on the ty
... Some metabolic processes are regulated by enzymes that exist in different molecular forms - isoenzymes Isoenzymes - multiple forms of an enzyme which differ in amino acid sequence but catalyze the same ...
... Some metabolic processes are regulated by enzymes that exist in different molecular forms - isoenzymes Isoenzymes - multiple forms of an enzyme which differ in amino acid sequence but catalyze the same ...
DO NOW Monday 2/12
... Are specialized proteins that help: – Catalyze (“speed up”) reactions – Break down molecules – Build up new molecules And can be affected by changes in the enzyme’s environment – their activity or effectiveness ...
... Are specialized proteins that help: – Catalyze (“speed up”) reactions – Break down molecules – Build up new molecules And can be affected by changes in the enzyme’s environment – their activity or effectiveness ...
Chapter 2-ROLE OF ENZYMES
... 21. Is this inhibitor able to do this because it is similar or different to the molecular structure of the substrate? 22. At low concentrations of competitive inhibitor the reaction rate is still high as few active sites are blocked by the inhibitor leaving the substrates no difficulty in finding fr ...
... 21. Is this inhibitor able to do this because it is similar or different to the molecular structure of the substrate? 22. At low concentrations of competitive inhibitor the reaction rate is still high as few active sites are blocked by the inhibitor leaving the substrates no difficulty in finding fr ...
Section 6 – Catalysis
... Inhibitor is similar in structure and electrical charge to substrate It binds to the active site An increase in the substrate can result in an increase of product formation (inhibitor is out competed) Competitive inhibition can be reversible or irreversible (depending on mechanism of binding) ...
... Inhibitor is similar in structure and electrical charge to substrate It binds to the active site An increase in the substrate can result in an increase of product formation (inhibitor is out competed) Competitive inhibition can be reversible or irreversible (depending on mechanism of binding) ...
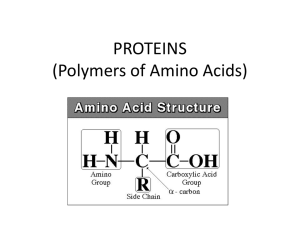
PROTEINS (Polymers of Amino Acids)
... • Substrate – substance being broken down • Active site – area where substrate/enzyme connect ...
... • Substrate – substance being broken down • Active site – area where substrate/enzyme connect ...
Properties of Enzymes
... Vmax – maximal velocity of a reaction. Addition of more substrate will not increase the rate of the reaction. Km – The concentration of substrate at which the rate of the reaction is half-maximal ...
... Vmax – maximal velocity of a reaction. Addition of more substrate will not increase the rate of the reaction. Km – The concentration of substrate at which the rate of the reaction is half-maximal ...
Mechanism of enzyme action, kinetic of enzymatic catalysis
... •The enzyme cannot differentiate between the two compounds •When inhibitor binds, prevents the substrate from binding •Inhibitor can be released by increasing substrate concentration ...
... •The enzyme cannot differentiate between the two compounds •When inhibitor binds, prevents the substrate from binding •Inhibitor can be released by increasing substrate concentration ...
Cofactors and Inhibitors
... the activity of an enzyme. If the inhibitor attaches to the enzyme the enzyme will change shape making it denatured and so the reaction will not occur. And example of a non competitive inhibitor is Sarin. Sarin is a nerve gas and if inhaled in large amounts, can be deadly. Sarin paralizes your muscl ...
... the activity of an enzyme. If the inhibitor attaches to the enzyme the enzyme will change shape making it denatured and so the reaction will not occur. And example of a non competitive inhibitor is Sarin. Sarin is a nerve gas and if inhaled in large amounts, can be deadly. Sarin paralizes your muscl ...
Enzyme Puzzle Activity
... and enzymes work together like puzzles. Only one active site of an enzyme will fit in like a puzzle piece with a specific substrate. Thus, they are very specific. Every enzyme has its optimum pH (= pH where it works best). Most enzymes work in almost neutral media. When the temperature increases, mo ...
... and enzymes work together like puzzles. Only one active site of an enzyme will fit in like a puzzle piece with a specific substrate. Thus, they are very specific. Every enzyme has its optimum pH (= pH where it works best). Most enzymes work in almost neutral media. When the temperature increases, mo ...
LAB 8: ENZYMES AS DRUG TARGETS.
... enzyme inhibition. The rate of an enzyme reaction (V) varies with substrate (S) concentration. Increasing S increased V until Vmax is reached. Km, concentration of substrate is the concentration at which reaction rate is half maximal., represents a measure of how tightly the substrate is bound. i.e. ...
... enzyme inhibition. The rate of an enzyme reaction (V) varies with substrate (S) concentration. Increasing S increased V until Vmax is reached. Km, concentration of substrate is the concentration at which reaction rate is half maximal., represents a measure of how tightly the substrate is bound. i.e. ...
Enzyme inhibitor

An enzyme inhibitor is a molecule that binds to an enzyme and decreases its activity. Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used in pesticides. Not all molecules that bind to enzymes are inhibitors; enzyme activators bind to enzymes and increase their enzymatic activity, while enzyme substrates bind and are converted to products in the normal catalytic cycle of the enzyme.The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction. Inhibitor binding is either reversible or irreversible. Irreversible inhibitors usually react with the enzyme and change it chemically (e.g. via covalent bond formation). These inhibitors modify key amino acid residues needed for enzymatic activity. In contrast, reversible inhibitors bind non-covalently and different types of inhibition are produced depending on whether these inhibitors bind to the enzyme, the enzyme-substrate complex, or both.Many drug molecules are enzyme inhibitors, so their discovery and improvement is an active area of research in biochemistry and pharmacology. A medicinal enzyme inhibitor is often judged by its specificity (its lack of binding to other proteins) and its potency (its dissociation constant, which indicates the concentration needed to inhibit the enzyme). A high specificity and potency ensure that a drug will have few side effects and thus low toxicity.Enzyme inhibitors also occur naturally and are involved in the regulation of metabolism. For example, enzymes in a metabolic pathway can be inhibited by downstream products. This type of negative feedback slows the production line when products begin to build up and is an important way to maintain homeostasis in a cell. Other cellular enzyme inhibitors are proteins that specifically bind to and inhibit an enzyme target. This can help control enzymes that may be damaging to a cell, like proteases or nucleases. A well-characterised example of this is the ribonuclease inhibitor, which binds to ribonucleases in one of the tightest known protein–protein interactions. Natural enzyme inhibitors can also be poisons and are used as defences against predators or as ways of killing prey.