Article Evolution of a Genome-Encoded Bias in Amino Acid
... toward fewer cognate amino acids in certain amino acid biosynthetic pathways and a profile of bias across amino acids that differed between the two groups, suggesting a possible correlation with the organisms’ ecological niches. The cognate bias hypothesis was recently tested and confirmed using a f ...
... toward fewer cognate amino acids in certain amino acid biosynthetic pathways and a profile of bias across amino acids that differed between the two groups, suggesting a possible correlation with the organisms’ ecological niches. The cognate bias hypothesis was recently tested and confirmed using a f ...
Amino Acids [PDF:247KB]
... crustaceans, and Meat): isoleucine, leucine, lysine, sulfur-containing amino acids (methionine, cystine), aromatic amino acids (phenylalanine, tyrosine), threonine, tryptophan, valine, and histidine as essential amino acids that cannot be synthesized in the body at all or sufficiently, and arginine, ...
... crustaceans, and Meat): isoleucine, leucine, lysine, sulfur-containing amino acids (methionine, cystine), aromatic amino acids (phenylalanine, tyrosine), threonine, tryptophan, valine, and histidine as essential amino acids that cannot be synthesized in the body at all or sufficiently, and arginine, ...
CALPAIN: TRANSITIONING FROM THE USE OF THE
... substrate, PLFMER, is cut at the intended scissile bond between F and M. Alanine substitutions at each position optimized the sequence to PLFAAR, which has a 2.3-fold higher turnover rate. The set of substrates derived from this study provided a tool for profiling the activity of calpain isoforms. O ...
... substrate, PLFMER, is cut at the intended scissile bond between F and M. Alanine substitutions at each position optimized the sequence to PLFAAR, which has a 2.3-fold higher turnover rate. The set of substrates derived from this study provided a tool for profiling the activity of calpain isoforms. O ...
The Three-Dimensional Structure of Aspergillus niger Pectin Lyase
... amino acid sequence. PLB shares 46 to 65% amino acid sequence identity with other A. niger pectin lyases. Unlike the Pels, pectin lyases do not require Ca21 for enzymatic activity; nevertheless, Ca21 and Na1 are known to stimulate enzymatic activity near the pH optimum of 8.5 (Kester and Visser, 199 ...
... amino acid sequence. PLB shares 46 to 65% amino acid sequence identity with other A. niger pectin lyases. Unlike the Pels, pectin lyases do not require Ca21 for enzymatic activity; nevertheless, Ca21 and Na1 are known to stimulate enzymatic activity near the pH optimum of 8.5 (Kester and Visser, 199 ...
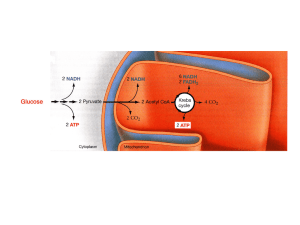
Pentose P Path
... NADPH, a product of the Pentose Phosphate Pathway, functions as a reductant in anabolic (synthetic) pathways, e.g., fatty acid synthesis. NAD+ serves as electron acceptor in catabolic pathways, in which metabolites are oxidized. ...
... NADPH, a product of the Pentose Phosphate Pathway, functions as a reductant in anabolic (synthetic) pathways, e.g., fatty acid synthesis. NAD+ serves as electron acceptor in catabolic pathways, in which metabolites are oxidized. ...
Theoretical studies on pyridoxal 5’-phosphate- catalyzed reactions of biological relevance 2014
... of as much as 1.1 billion years (Radzicka1996, Snider2000, Wolfenden2001). This may question the possibility of life formation when considering the age of the Earth (~4.5 billion years). However, taking into account the number of residues of each protein and the average number of proteins in the cel ...
... of as much as 1.1 billion years (Radzicka1996, Snider2000, Wolfenden2001). This may question the possibility of life formation when considering the age of the Earth (~4.5 billion years). However, taking into account the number of residues of each protein and the average number of proteins in the cel ...
Journal of Bacteriology
... lowed by enzyme assay at 30 C. The results are given in Table 2. Although the L-alanine adding enzyme gave a linear curve at 42 C, a considerable amount of activity was lost during incubation of the enzyme for 10 min at this temperature, probably because cofactors or substrates or both, which may pr ...
... lowed by enzyme assay at 30 C. The results are given in Table 2. Although the L-alanine adding enzyme gave a linear curve at 42 C, a considerable amount of activity was lost during incubation of the enzyme for 10 min at this temperature, probably because cofactors or substrates or both, which may pr ...
C H A P
... of the lipase(s) produced by this bacterium remain almost unknown, since only related enzymes such as H. pylori phospholipases A2 and C have been cloned and/or characterized (Weitkamp et al., 1993; Dorrell et al., 1999; General Introduction 3.4.2). Therefore, this chapter is focused on the isolation ...
... of the lipase(s) produced by this bacterium remain almost unknown, since only related enzymes such as H. pylori phospholipases A2 and C have been cloned and/or characterized (Weitkamp et al., 1993; Dorrell et al., 1999; General Introduction 3.4.2). Therefore, this chapter is focused on the isolation ...
Glycogen Metabolism
... Glycogenin initiates glycogen synthesis. • Glycogenin is an enzyme that catalyzes attachment of a glucose molecule to one of its own tyrosine residues. • Glycogenin is a dimer, and evidence indicates that the 2 copies of the enzyme glucosylate one another. ...
... Glycogenin initiates glycogen synthesis. • Glycogenin is an enzyme that catalyzes attachment of a glucose molecule to one of its own tyrosine residues. • Glycogenin is a dimer, and evidence indicates that the 2 copies of the enzyme glucosylate one another. ...
Structure, mechanism and regulation of pyruvate carboxylase
... catalyses the HCO3 − - and MgATP-dependent carboxylation of pyruvate to form oxaloacetate. This is a very important anaplerotic reaction, replenishing oxaloacetate withdrawn from the tricarboxylic acid cycle for various pivotal biochemical pathways. PC is therefore considered as an enzyme that is cr ...
... catalyses the HCO3 − - and MgATP-dependent carboxylation of pyruvate to form oxaloacetate. This is a very important anaplerotic reaction, replenishing oxaloacetate withdrawn from the tricarboxylic acid cycle for various pivotal biochemical pathways. PC is therefore considered as an enzyme that is cr ...
Biochemistry 304 2014 Student Edition Amino Acid Metabolism
... hydrolytic enzymes (cathepsins). Degrade substances taken up by endocytosis. Recycle intracellular constituents enclosed within vacuoles. In “well nourished cells” protein degradation is nonselective. In starving cells a selective pathway is activated that imports and degrades proteins that contain ...
... hydrolytic enzymes (cathepsins). Degrade substances taken up by endocytosis. Recycle intracellular constituents enclosed within vacuoles. In “well nourished cells” protein degradation is nonselective. In starving cells a selective pathway is activated that imports and degrades proteins that contain ...
Structure of the ordered hydration of amino acids
... overlooking of unconventional water–protein interactions and of the connection between the main chain and side chains. For example, the nitrogen heteroatoms of tryptophan and histidine side chains have been shown to participate in an off-plane interaction with water (Stollar et al., 2004), with wate ...
... overlooking of unconventional water–protein interactions and of the connection between the main chain and side chains. For example, the nitrogen heteroatoms of tryptophan and histidine side chains have been shown to participate in an off-plane interaction with water (Stollar et al., 2004), with wate ...
Biochemical Thermodynamics - Illinois Institute of Technology
... Assume the protonated species is H3O+ rather than H+ Then the reaction is 2 H2O H3O+ + OH keq = [H3O+][OH-] / ([H2O]2) At pH=7, [H3O+] = [OH-] = 10-7M Dilute solution: [H2O] = 55.5M, so ...
... Assume the protonated species is H3O+ rather than H+ Then the reaction is 2 H2O H3O+ + OH keq = [H3O+][OH-] / ([H2O]2) At pH=7, [H3O+] = [OH-] = 10-7M Dilute solution: [H2O] = 55.5M, so ...
Raines, ChemRev 1998
... its integrity and solubility under drastic conditions: first, 0.25 N sulfuric acid at 5 °C, and then, pH 3.0 at 95-100 °C.84 The final step in this protocol calls for crystallization of the enzyme. The three-dimensional structure of RNase A is fully encoded by its amino acid sequence.85-89 This disc ...
... its integrity and solubility under drastic conditions: first, 0.25 N sulfuric acid at 5 °C, and then, pH 3.0 at 95-100 °C.84 The final step in this protocol calls for crystallization of the enzyme. The three-dimensional structure of RNase A is fully encoded by its amino acid sequence.85-89 This disc ...
Amino Acid Catabolism: N
... Hereditary deficiency of any of the Urea Cycle enzymes leads to hyperammonemia - elevated [ammonia] in blood. Total lack of any Urea Cycle enzyme is lethal. ...
... Hereditary deficiency of any of the Urea Cycle enzymes leads to hyperammonemia - elevated [ammonia] in blood. Total lack of any Urea Cycle enzyme is lethal. ...
Scholarly Interest Report
... defined, and specific kinetic mechanisms have been proposed and examined. A second area of current concern is the structure and reactivity of the binuclear center comprising cytochrome a3 and CuB and which is the catalytic site for the reduction of oxygen to water. This binuclear center is being stu ...
... defined, and specific kinetic mechanisms have been proposed and examined. A second area of current concern is the structure and reactivity of the binuclear center comprising cytochrome a3 and CuB and which is the catalytic site for the reduction of oxygen to water. This binuclear center is being stu ...
Laboratory Evolution of Cytochrome P450 BM-3 Monooxygenase for Organic Cosolvents
... Point mutagenesis at low error rates explores only a very limited set of (primarily conservative) amino acid substitutions. We and others have observed that saturation mutagenesis performed at sites identified by error-prone PCR often generates further improvements. Taking double-mutant F87AB5 as th ...
... Point mutagenesis at low error rates explores only a very limited set of (primarily conservative) amino acid substitutions. We and others have observed that saturation mutagenesis performed at sites identified by error-prone PCR often generates further improvements. Taking double-mutant F87AB5 as th ...
Effect of Alanine-293 Replacement on the Activity, ATP Binding, and
... leucylation of tRNALeu. Several mutants in the CP1 domain of Escherichia coli LeuRS were obtained by introduction of restriction endonuclease sites into its gene, leuS. Of these mutants, only LeuRS-A293F had decreased activity (46%) compared to the native enzyme. To investigate the effect of A293 on ...
... leucylation of tRNALeu. Several mutants in the CP1 domain of Escherichia coli LeuRS were obtained by introduction of restriction endonuclease sites into its gene, leuS. Of these mutants, only LeuRS-A293F had decreased activity (46%) compared to the native enzyme. To investigate the effect of A293 on ...
REDESIGN OF CARNITINE ACETYLTRANSFERASE SPECIFICITY BY PROTEIN ENGINEERING UNIVERSIDAD DE BARCELONA
... yeast and analysed its effect on ester production during alcoholic fermentation. Compared with control cells, overexpression of CrAT caused a significant reduction in the production of some esters, including the important flavour components ethyl acetate and 3-methyl-butyl acetate (isoamyl acetate). ...
... yeast and analysed its effect on ester production during alcoholic fermentation. Compared with control cells, overexpression of CrAT caused a significant reduction in the production of some esters, including the important flavour components ethyl acetate and 3-methyl-butyl acetate (isoamyl acetate). ...
Catalytic triad

A catalytic triad refers to the three amino acid residues that function together at the centre of the active site of some hydrolase and transferase enzymes (e.g. proteases, amidases, esterases, acylases, lipases and β-lactamases). An Acid-Base-Nucleophile triad is a common motif for generating a nucleophilic residue for covalent catalysis. The residues form a charge-relay network to polarise and activate the nucleophile, which attacks the substrate, forming a covalent intermediate which is then hydrolysed to regenerate free enzyme. The nucleophile is most commonly a serine or cysteine amino acid, but occasionally threonine. Because enzymes fold into complex three-dimensional structures, the residues of a catalytic triad can be far from each other along the amino-acid sequence (primary structure), however, they are brought close together in the final fold.As well as divergent evolution of function (and even the triad's nucleophile), catalytic triads show some of the best examples of convergent evolution. Chemical constraints on catalysis have led to the same catalytic solution independently evolving in at least 23 separate superfamilies. Their mechanism of action is consequently one of the best studied in biochemistry.
![Amino Acids [PDF:247KB]](http://s1.studyres.com/store/data/002278939_1-1b03d0bc0e7fdaaa14234846bba6ef6c-300x300.png)