Name: Chemistry Honors Date: Period: ____ Reduction/Oxidation
... Remember, oxidation and reduction (redox) occurs in every type of chemical reaction except double replacement. Model 1: In order to track electron movement we have to watch for changes in charge. But for molecular substances we must use oxidation numbers. Assigning oxidation number is a system devel ...
... Remember, oxidation and reduction (redox) occurs in every type of chemical reaction except double replacement. Model 1: In order to track electron movement we have to watch for changes in charge. But for molecular substances we must use oxidation numbers. Assigning oxidation number is a system devel ...
Effect of edge reconstruction and electron
... of the hole conductance than of the election conductance, see Fig. 1(b), as in Ref. 14. The effect of the Stone-Wales defect (Fig.1(c)) is more subtle because it doesn’t change number of carbon atoms at the ribbon’s edge. For periodically repeated StoneWales defects the aGNR unit cell is doubled and ...
... of the hole conductance than of the election conductance, see Fig. 1(b), as in Ref. 14. The effect of the Stone-Wales defect (Fig.1(c)) is more subtle because it doesn’t change number of carbon atoms at the ribbon’s edge. For periodically repeated StoneWales defects the aGNR unit cell is doubled and ...
Polaronic exciton in a parabolic quantum dot
... In polar semiconductors the excitonic properties are significantly altered due to the influence of the exciton – optical-phonon interaction. For instance, exciton binding energy is enhanced, depending on the strength of this interaction. A proper description of the nature of this coupling among elec ...
... In polar semiconductors the excitonic properties are significantly altered due to the influence of the exciton – optical-phonon interaction. For instance, exciton binding energy is enhanced, depending on the strength of this interaction. A proper description of the nature of this coupling among elec ...
Specular Reflection of Very Slow Metastable Neon Atoms from a
... reflection of this type has been called quantum reflection and was observed on the reflection of He and H atoms on liquid helium surface [16,17] and also indirectly from the sticking coefficient of hydrogen on liquid He [18,19]. The quantum reflection is also expected on solid surfaces. Kasevich et ...
... reflection of this type has been called quantum reflection and was observed on the reflection of He and H atoms on liquid helium surface [16,17] and also indirectly from the sticking coefficient of hydrogen on liquid He [18,19]. The quantum reflection is also expected on solid surfaces. Kasevich et ...
Electrochromic Shift of Chlorophyll Absorption in Photosystem I from
... spectra were mutually normalized to the same intensities at wavelengths above 700 nm, where we have observed no time evolution in DA after 200 ps. This normalization also leads to the same integrated intensities in both spectra, which is consistent with the fact that excitation and reduction states ...
... spectra were mutually normalized to the same intensities at wavelengths above 700 nm, where we have observed no time evolution in DA after 200 ps. This normalization also leads to the same integrated intensities in both spectra, which is consistent with the fact that excitation and reduction states ...
1b-Redox FIB notes and practice
... c) 4Al (s) + 3O2 (g) 2Al2O3 (s) ASSIGNING OXIDATION NUMBERS Oxidation numbers are a book keeping concept devised by chemists. An oxidation number is a ___________ or ___________ number assigned to an atom according to a set of arbitrary rules. Complete redox equations can be balanced by using oxid ...
... c) 4Al (s) + 3O2 (g) 2Al2O3 (s) ASSIGNING OXIDATION NUMBERS Oxidation numbers are a book keeping concept devised by chemists. An oxidation number is a ___________ or ___________ number assigned to an atom according to a set of arbitrary rules. Complete redox equations can be balanced by using oxid ...
coherent interaction of atoms and molecules with laser radiation
... electronic level, the vibrational number of the ground state was chosen v’’=0, of the excited state v’=0, and fluorescence was observed to the level v1’’=3. Rotational quantum numbers were chosen J’’=99J’=99J1’’=99 (Q-type transition) and J’’=105J’=106J1’’=107 (R-type transition). When developin ...
... electronic level, the vibrational number of the ground state was chosen v’’=0, of the excited state v’=0, and fluorescence was observed to the level v1’’=3. Rotational quantum numbers were chosen J’’=99J’=99J1’’=99 (Q-type transition) and J’’=105J’=106J1’’=107 (R-type transition). When developin ...
Stabilization of circular Rydberg atoms by circularly - BORA
... laser frequency, and if we used an in-plane linearly polarized laser field instead. Figure 3 shows the results for an x polarized laser pulse on a 5g (m = 4) initial state for pulses of 4, 6, and 8 optical cycles, and the results are, in fact, in qualitative agreement with the results in Fig. 1. The ...
... laser frequency, and if we used an in-plane linearly polarized laser field instead. Figure 3 shows the results for an x polarized laser pulse on a 5g (m = 4) initial state for pulses of 4, 6, and 8 optical cycles, and the results are, in fact, in qualitative agreement with the results in Fig. 1. The ...
INTERPLAY BETWEEN STRUCTURAL AND ELECTRONIC PROPERTIES OF CARBON NANOTUBES
... electrons have been considered as active. Our description of Pb nanowires within the jellium model follows Ref. 20 . We use rs = 2.30 a.u. for the WignerSeitz radius and the valence Z = 4 for Pb in the ground-state configuration [Xe]4f14 5d10 6s2 6p2 . The jellium background charge density of Bi, whi ...
... electrons have been considered as active. Our description of Pb nanowires within the jellium model follows Ref. 20 . We use rs = 2.30 a.u. for the WignerSeitz radius and the valence Z = 4 for Pb in the ground-state configuration [Xe]4f14 5d10 6s2 6p2 . The jellium background charge density of Bi, whi ...
Chapter 8 Concepts of Chemical Bonding
... electronegativity. The more polar bond will be the one between the atoms with the biggest differences in electronegativity. Consequently, the B—Cl bond is more polar; the chlorine atom carries the partial negative charge because it has a higher electronegativity. (b) In this example phosphorus is co ...
... electronegativity. The more polar bond will be the one between the atoms with the biggest differences in electronegativity. Consequently, the B—Cl bond is more polar; the chlorine atom carries the partial negative charge because it has a higher electronegativity. (b) In this example phosphorus is co ...
CHEMISTRY
... But zinc put in the reaction was 10 g (not 65,3) then to know how much ZnCl2 is formed, it is necessary to divide 10 / mw of Zinc (65,37) to have the number of moles (= 0,153). The same number of moles of ZnCl2 (see reaction coefficients). The mass of ZnCl2 will be = number of moles (0,153) x mm of ...
... But zinc put in the reaction was 10 g (not 65,3) then to know how much ZnCl2 is formed, it is necessary to divide 10 / mw of Zinc (65,37) to have the number of moles (= 0,153). The same number of moles of ZnCl2 (see reaction coefficients). The mass of ZnCl2 will be = number of moles (0,153) x mm of ...
Supported-Metal Catalysts
... and interlaboratory results were brought into much closer agreement. It was soon realized, however, that hydrogen chemisorption could not be used to determine metal dispersion in SMSI catalysts because these catalysts did not chemisorb hydrogen. In fact, suppression of hydrogen chemisorption was soo ...
... and interlaboratory results were brought into much closer agreement. It was soon realized, however, that hydrogen chemisorption could not be used to determine metal dispersion in SMSI catalysts because these catalysts did not chemisorb hydrogen. In fact, suppression of hydrogen chemisorption was soo ...
my title - Ohio University Physics and Astronomy
... across the surface to a random final location selected by the computer. For each bias, the atom was manipulated by using different current values (from 8 to 950 nA). For each set of ...
... across the surface to a random final location selected by the computer. For each bias, the atom was manipulated by using different current values (from 8 to 950 nA). For each set of ...
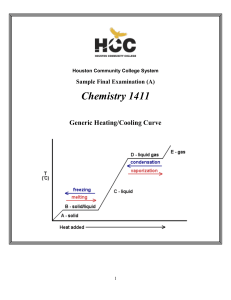
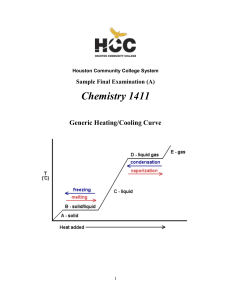
1411FINALSAMPLE+KEY - Houston Community College
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
ESO - ENCIGA
... tested by subsequent investigation and can be modified by its results. Science does not give statements of absolute eternal truth, it only provides theories. We know that those theories will probably be refined in the future, and some of them may even be discarded in favour of theories that make mor ...
... tested by subsequent investigation and can be modified by its results. Science does not give statements of absolute eternal truth, it only provides theories. We know that those theories will probably be refined in the future, and some of them may even be discarded in favour of theories that make mor ...
CH 4: Chemical Reactions
... were a monatomic ion. A. Hydrogen can be either +1 or –1. B. Oxygen usually has an oxidation number of –2. In peroxides, oxygen is –1. C. Halogens usually have an oxidation number of –1. • When bonded to oxygen, chlorine, bromine, and iodine have positive oxidation numbers. ...
... were a monatomic ion. A. Hydrogen can be either +1 or –1. B. Oxygen usually has an oxidation number of –2. In peroxides, oxygen is –1. C. Halogens usually have an oxidation number of –1. • When bonded to oxygen, chlorine, bromine, and iodine have positive oxidation numbers. ...
29_LectureOutlineAto..
... The Quantum-Mechanical Hydrogen Atom (cont’d) 3. The plane of the electron’s orbit can be tilted, but only at certain discrete angles. Each allowed angle is characterized by a quantum number m, which must be one of the values ...
... The Quantum-Mechanical Hydrogen Atom (cont’d) 3. The plane of the electron’s orbit can be tilted, but only at certain discrete angles. Each allowed angle is characterized by a quantum number m, which must be one of the values ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.