1 Mole
... How many moles of O2 are needed to react with 4 moles of sodium? How many moles of O2 are needed to react with 2 moles of sodium? How many moles of Na2O are produced when 2 moles of sodium are used? ...
... How many moles of O2 are needed to react with 4 moles of sodium? How many moles of O2 are needed to react with 2 moles of sodium? How many moles of Na2O are produced when 2 moles of sodium are used? ...
Wednesday, Aug. 30, 2006
... • Establish limited number of fundamental laws, usually Theory with mathematical expressions • Predict nature’s courses ⇒Theory and Experiment work hand-in-hand ⇒Theory works generally under restricted conditions ⇒Discrepancies between experimental measurements and theory presents opportunities to q ...
... • Establish limited number of fundamental laws, usually Theory with mathematical expressions • Predict nature’s courses ⇒Theory and Experiment work hand-in-hand ⇒Theory works generally under restricted conditions ⇒Discrepancies between experimental measurements and theory presents opportunities to q ...
Theoretical Modeling of Transport in Nanostructures June 02, 2009
... elastic scattering. Contrary to the most of the ab initio studies, using the trial and errormethod by applying different molecules and various substitutions, in my talk I propose a way of a systematic study of the junctions, in which the asymmetry necessary for the molecule to act as a diode can be ...
... elastic scattering. Contrary to the most of the ab initio studies, using the trial and errormethod by applying different molecules and various substitutions, in my talk I propose a way of a systematic study of the junctions, in which the asymmetry necessary for the molecule to act as a diode can be ...
Vacuum Ultraviolet Spectroscopy and Photochemistry of Zinc
... complete agreement with the earlier1 DFT findings. Thus the ground state equilibrium bond length was found to be 1.5413 Å, and the fundamental vibrational frequencies of ν1, the symmetric (σg), and ν3, the asymmetric (σu) stretches were 1915.5 and 1926.8 cm−1, respectively, while ν2, the bending (πu) ...
... complete agreement with the earlier1 DFT findings. Thus the ground state equilibrium bond length was found to be 1.5413 Å, and the fundamental vibrational frequencies of ν1, the symmetric (σg), and ν3, the asymmetric (σu) stretches were 1915.5 and 1926.8 cm−1, respectively, while ν2, the bending (πu) ...
M. Sc. in Industrial Chemistry Centre for Applied Chemistry Syllabus
... exactly solvable system, particle in a box, harmonic oscillator and hydrogen atom, shapes of the atomic orbitals, orbital angular momenta, tunnelling. Approximate methods of quantum mechanics, vibrational principle, perturbation theory up to second order in energy, applications, Born interpretation, ...
... exactly solvable system, particle in a box, harmonic oscillator and hydrogen atom, shapes of the atomic orbitals, orbital angular momenta, tunnelling. Approximate methods of quantum mechanics, vibrational principle, perturbation theory up to second order in energy, applications, Born interpretation, ...
Frustrated Quantum Magnetism with Laser-Dressed Rydberg Atoms
... Hertz (or few nK) regime [3] (see however [4]). Below we take an alternative route, and show that laser-excited interacting Rydberg atoms [5] provide us not only with a complete toolbox to design and realize the complex spin1/2 models of interest, but also give rise to energy scales much larger than ...
... Hertz (or few nK) regime [3] (see however [4]). Below we take an alternative route, and show that laser-excited interacting Rydberg atoms [5] provide us not only with a complete toolbox to design and realize the complex spin1/2 models of interest, but also give rise to energy scales much larger than ...
Chemical-Principles-7th-Edition-Zumdahl-Test-Bank
... 18. An element's most stable ion forms an ionic compound with chlorine having the formula XCl2. If the mass number of the ion is 24 and it has 10 electrons, what is the element and how many neutrons does it have? A) Mg, 12 neutrons B) Ne, 16 neutrons C) O, 16 neutrons D) Ne, 14 neutrons E) Na, 11 ne ...
... 18. An element's most stable ion forms an ionic compound with chlorine having the formula XCl2. If the mass number of the ion is 24 and it has 10 electrons, what is the element and how many neutrons does it have? A) Mg, 12 neutrons B) Ne, 16 neutrons C) O, 16 neutrons D) Ne, 14 neutrons E) Na, 11 ne ...
2.6 M - Thierry Karsenti
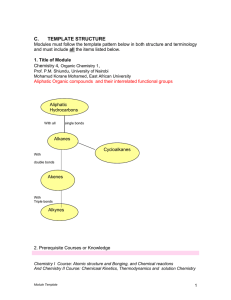
... This module in organic chemistry is designed to prepare students that wish to become teachers : Student teachers must have knowledge of the key concepts and classification tools of organic chemistry that include functional groups of hydrocarbons,alcohols and ethers,aldehydes and ketones,alkyl halide ...
... This module in organic chemistry is designed to prepare students that wish to become teachers : Student teachers must have knowledge of the key concepts and classification tools of organic chemistry that include functional groups of hydrocarbons,alcohols and ethers,aldehydes and ketones,alkyl halide ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
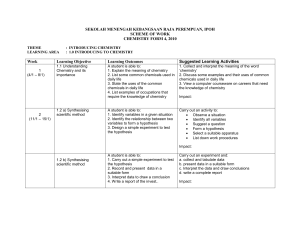
... Gather information and discuss a. the similarities in chemical properties of lithium, sodium and potassium b. the relationship between the chemical properties of Group 1 elements and their electron arrangements Study the reaction of lithium, sodium and potassium with chlorine and bromine through com ...
... Gather information and discuss a. the similarities in chemical properties of lithium, sodium and potassium b. the relationship between the chemical properties of Group 1 elements and their electron arrangements Study the reaction of lithium, sodium and potassium with chlorine and bromine through com ...
File
... The mass of the products is always greater than the mass of the reactants. b. The mass of the reactants equals the mass of the products. c. The mass of the reactants increases. d. The mass of the products is always less than the mass of the reactants. ...
... The mass of the products is always greater than the mass of the reactants. b. The mass of the reactants equals the mass of the products. c. The mass of the reactants increases. d. The mass of the products is always less than the mass of the reactants. ...
CfE Advanced Higher Chemistry Unit 2: Organic
... formed when atomic orbitals lie perpendicular to the bond and overlap side on. End to end overlap is more efficient than side on overlap and therefore σ bonds are stronger than π bonds. ...
... formed when atomic orbitals lie perpendicular to the bond and overlap side on. End to end overlap is more efficient than side on overlap and therefore σ bonds are stronger than π bonds. ...
QIPC 2011
... – restrict to “low energy sector”. Still exponentially big in number of electrons – now Fock vectors are in terms of orbitals, not positions – identify orthogonal states that differ slightly , i.e., electron moved from one orbital to another, or one spin flipped. This pair is a good candidate for ...
... – restrict to “low energy sector”. Still exponentially big in number of electrons – now Fock vectors are in terms of orbitals, not positions – identify orthogonal states that differ slightly , i.e., electron moved from one orbital to another, or one spin flipped. This pair is a good candidate for ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.