Adobe Photoshop PDF - Perimeter Institute
... the board and let it ‘snap’ back down. The bungee cord exerts a force on the toy making it accelerate. 3. Demonstrate Einstein’s model of gravity by removing the bungee cord, holding the toy in the air and accelerating the board up to hit it. “According to Einstein, gravity is not a force. The toy d ...
... the board and let it ‘snap’ back down. The bungee cord exerts a force on the toy making it accelerate. 3. Demonstrate Einstein’s model of gravity by removing the bungee cord, holding the toy in the air and accelerating the board up to hit it. “According to Einstein, gravity is not a force. The toy d ...
On the Extra Anomalous Gyromagnetic Ratio of the Electron and
... existence of anti-particles. In its bare form, the Dirac equation provided us with an impressive and accurate description of the Electron hence it being referred in most of the literature as the “Dirac Equation for the Electron”. It also accounts very well for quarks and other spin-1/2 particles al ...
... existence of anti-particles. In its bare form, the Dirac equation provided us with an impressive and accurate description of the Electron hence it being referred in most of the literature as the “Dirac Equation for the Electron”. It also accounts very well for quarks and other spin-1/2 particles al ...
DEPARTMENT OF CHEMISTRY, CFS, IIUM
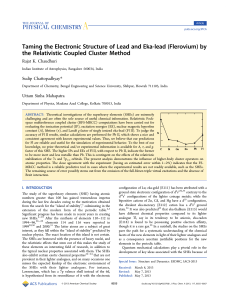
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
Electron dephasing scattering rate in two
... The first term is the interference contribution due to triplet state and the second term due to the singlet state. b = B / B and bso = Bso / B, where B and Bso are the dephasing and spin-orbit scattering fields, respectively. Ft共b , bso兲 is given in the works of Minkov et al.15 and Iordanskii et ...
... The first term is the interference contribution due to triplet state and the second term due to the singlet state. b = B / B and bso = Bso / B, where B and Bso are the dephasing and spin-orbit scattering fields, respectively. Ft共b , bso兲 is given in the works of Minkov et al.15 and Iordanskii et ...
введение в общую introductio to the general ch ведение в общую
... the number of chemical bonds a given atom has formed in a given molecule. The number of bonds formed by a given element was originally thought to be a fixed chemical property. In fact, in most cases this is not true. For example, phosphorus often has a valence of three, but can also have other valen ...
... the number of chemical bonds a given atom has formed in a given molecule. The number of bonds formed by a given element was originally thought to be a fixed chemical property. In fact, in most cases this is not true. For example, phosphorus often has a valence of three, but can also have other valen ...
WORD - SSS Chemistry
... ___________________________ devised the Scattering Experiment, which showed that all atoms had a small dense __________________________. ...
... ___________________________ devised the Scattering Experiment, which showed that all atoms had a small dense __________________________. ...
Far-infrared Rydberg-Rydberg transitions in a magnetic field: Deexcitation of antihydrogen atoms
... shown. The initial state, respectively 28s, 29s 关Fig. 3共a兲兴 and 27d 关Fig. 3共b兲兴, is excited with a far infrared photon 共90– 110 m兲 to final states near the n-mixing regime. We indicated the position of the p and f states for a field-free situation in Fig. 3. In a one-photon transition the populatio ...
... shown. The initial state, respectively 28s, 29s 关Fig. 3共a兲兴 and 27d 关Fig. 3共b兲兴, is excited with a far infrared photon 共90– 110 m兲 to final states near the n-mixing regime. We indicated the position of the p and f states for a field-free situation in Fig. 3. In a one-photon transition the populatio ...
Document
... The Pauli exclusion principle states that the joining of atoms to form a system (crystal) does not alter the total number of quantum states The discrete energy must split into a band of energies in order that each electron can occupy a distinct quantum state ...
... The Pauli exclusion principle states that the joining of atoms to form a system (crystal) does not alter the total number of quantum states The discrete energy must split into a band of energies in order that each electron can occupy a distinct quantum state ...
MEASUREMENT OF LIFETIMES OF EXCITED STATES OF THE
... IN measurement of excitation cross sections of energetic ions or atoms in collisions with gas molecules, we must consider the following fact. In the apparatus usually used for this type of experiment, a monoenergetic beam of the ions or atoms being studied enters a collision chamber filled with gas ...
... IN measurement of excitation cross sections of energetic ions or atoms in collisions with gas molecules, we must consider the following fact. In the apparatus usually used for this type of experiment, a monoenergetic beam of the ions or atoms being studied enters a collision chamber filled with gas ...
Quantum Monte Carlo Methods Chapter 14
... only, adds a simple term to the local energy operator. In our discussion below, we base our numerical Monte Carlo solution on the Metropolis algorithm. The implementation is rather similar to the one discussed in connection with the Ising model, the main difference resides in the form of the probabi ...
... only, adds a simple term to the local energy operator. In our discussion below, we base our numerical Monte Carlo solution on the Metropolis algorithm. The implementation is rather similar to the one discussed in connection with the Ising model, the main difference resides in the form of the probabi ...
Problems Chapter 9
... and put j = 0, i.e. kill y-component of B field. From now on we adopt this convention and write DÎ R instead of ∆. Let us note that we can also choose j = Π, than have in effect both positive and negatives vaules for D. A priori the two choices are distinct and this will have some consequence below. ...
... and put j = 0, i.e. kill y-component of B field. From now on we adopt this convention and write DÎ R instead of ∆. Let us note that we can also choose j = Π, than have in effect both positive and negatives vaules for D. A priori the two choices are distinct and this will have some consequence below. ...
class XI CHEMISTRY - Kendriya Vidyalaya No.1 Harni Road
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
- Kendriya Vidyalaya No. 2 Raipur
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
Ex: -F, -Cl, -Br
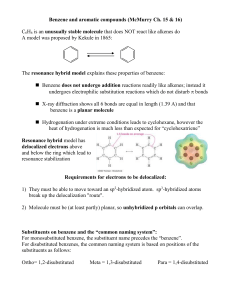
... X-ray diffraction shows all 6 bonds are equal in length (1.39 A) and that benzene is a planar molecule Hydrogenation under extreme conditions leads to cyclohexane, however the heat of hydrogenation is much less than expected for “cyclohexatriene” Resonance hybrid model has delocalized electrons ...
... X-ray diffraction shows all 6 bonds are equal in length (1.39 A) and that benzene is a planar molecule Hydrogenation under extreme conditions leads to cyclohexane, however the heat of hydrogenation is much less than expected for “cyclohexatriene” Resonance hybrid model has delocalized electrons ...
Term 111, Final Exam (All correct choices are A): 1. What is the
... 12. Bonds with higher ionic character form between (note: Ei = ionization energy and Eea = electron affinity) A) an element with a large Ei and an element with a small negative Eea B) an element with a small Ei and an element with a small negative Eea C) elements with equal values of Ei and Eea D) a ...
... 12. Bonds with higher ionic character form between (note: Ei = ionization energy and Eea = electron affinity) A) an element with a large Ei and an element with a small negative Eea B) an element with a small Ei and an element with a small negative Eea C) elements with equal values of Ei and Eea D) a ...
Class 1
... Multiple-charged ions are formed if there are many ionizable sites in the molecule, as in peptides and proteins, so that the formula masses of large molecules can be determined by ESI – another big advantage over EI. Most analyzers have limits on the size of m/z that can be measured with acceptable ...
... Multiple-charged ions are formed if there are many ionizable sites in the molecule, as in peptides and proteins, so that the formula masses of large molecules can be determined by ESI – another big advantage over EI. Most analyzers have limits on the size of m/z that can be measured with acceptable ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.