* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Biochemistry PPT
Survey
Document related concepts
Oxidative phosphorylation wikipedia , lookup
Point mutation wikipedia , lookup
Butyric acid wikipedia , lookup
Citric acid cycle wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Peptide synthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Photosynthesis wikipedia , lookup
Protein structure prediction wikipedia , lookup
Genetic code wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Proteolysis wikipedia , lookup
Transcript
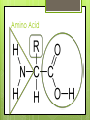
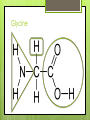
Biochemistry Chapter 2 Sections 3 & 4 Carbon Why is carbon so special? Atomic # 6 4 valence electrons Electrons for bonds in outermost shell that are available Can make up to 4 covalent bonds When Building electrons are shared block for the biomolecules of life! 4 Macromolecules Proteins People Lipids Love Carbohydrates Chicken Nucleic NuggetsAcids Macromolecules Every macromolecule has Monomer Single unit of a whole Polymer Many units or monomers bonded together DELICIOUS CARBOHYDRATES Carbohydrates Made of carbon, hydrogen, & oxygen Unique 1:2:1 pattern Used for energy mostly and sometimes for structure in plants Monomer: monosaccharide 2 monomers: disaccharide Polymer: polysaccharide Units of Carbs There Glucose – our energy source Fructose - sweetener Galactose – not as common There are 3 monosaccharides are 3 polysaccharides Glycogen – how we store carbs Starch – how plants store carbs Cellulose – structure in plants Links https://www.wisc- online.com/learn/generaleducation/anatomy-andphysiology1/ap13104/biomolecules-thecarbohydrates http://www.tv411.org/science/tv411whats-cooking/carbohydrates-sciencelesson/activity/1/1 LIPIDS (FATS & OILS) Lipids Made of carbon & hydrogen Does not dissolve in water Four types of lipids Triglycerides Phospholipids Waxes Steroids Triglycerides - FATS Saturated Have no double bonds in their fatty acid chains – maximum # of H atoms Straight Fat chain Usually animal fats Solid at room temperature Triglycerides Triglycerides – OILS Unsaturated Have at least one double bond in their fatty acid chain Forms Fat a kink in the chain Usually oils and from plants Liquid at room temperature Phospholipid Phospholipid Two fatty acid chains Found in all cells Makes up the cell membrane 2 layers of phospholipids lipid bilayer Phospholipid Bilayer Wax One fatty acid chain to a glycerol Makes cells waterproof Create protective layer in animals and plants Steroids No fatty acid chain Four carbon rings linked together Usually our hormones Ex: Cholesterol is found in our cell membranes Links http://www.hhmi.org/biointeractive/obesi ty/obesity_molecular/01.html http://www.wisconline.com/objects/ViewObject.aspx?ID= AP13204 NUCLEIC ACID Nucleic Acids Contains carbon, hydrogen, oxygen, nitrogen, and phosphorus Nucleic Acids Monomer = nucleotide 5 carbon sugar Phosphorous group Nitrogenous base Properties Store genetic information Two kinds of nucleic acids DNA RNA Protein Macromolecules with carbon, hydrogen, oxygen, and nitrogen Monomer: amino acid 2 monomers: dipeptide Polymer: polypeptide Amino Acids Each amino acid has three main groups Amino group Carboxyl group R group (changes) Change the shape and properties of the amino acid Amino Acid Glycine R group The R group makes each amino acid different There are 20 possible amino acids based off this one group The R group is = variable Proteins Covalent bond in proteins is called a peptide bond Links amino acids together For every peptide bond, one molecule of water is formed Links http://www.youtube.com/watch?v=va0D NJId_CM https://www.wisconline.com/learn/generaleducation/anatomy-andphysiology1/ap13304/biomolecules---theproteins Chemical Reaction A process the changes one set of chemicals into another Reactants Elements or compounds that enter or start a chemical reaction Products Elements or compounds that leave or end a chemical reaction Reactants Products Energy Sources Organisms need to carry our reactions that require energy Plants make their energy from the sun, animals get their energy from foods eaten Activation energy The energy needed to start a reaction Enzymes Enzyme catalyst is a type of protein that is a Catalysts start a reaction Without enzymes, reactions would take happen too slow to sustain life’s functions Three functions Start a reaction Speed up a reaction Lower activation energy Enzymes Each enzyme has an area known as the active site Where a specific molecule binds and a reaction occurs The molecule binding is called a substrate Similar to a lock and key Enzyme conditions Enzymes work at a specific conditions Temperature pH If not in desired conditions, this causes enzyme to change shape and lose its function Called denaturation http://www.sumanasinc.com/webconten t/animations/content/proteinstructure.ht ml Fold it! Help scientists figure out protein folding This game is open to the public The first hundred puzzles are known proteins But many proteins are not decoded and scientists are asking for our help to figure them out http://fold.it/