* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Principles of Metabolic Regulation
Western blot wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Gene regulatory network wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Transcriptional regulation wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Paracrine signalling wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Proteolysis wikipedia , lookup
Ultrasensitivity wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Lipid signaling wikipedia , lookup
Blood sugar level wikipedia , lookup
Mitogen-activated protein kinase wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metabolomics wikipedia , lookup
Biochemical cascade wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Citric acid cycle wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
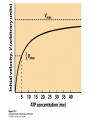
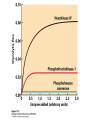
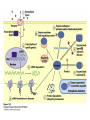
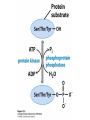
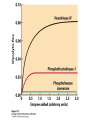
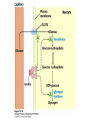
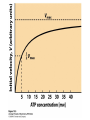
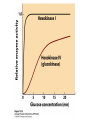
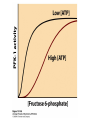
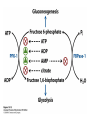
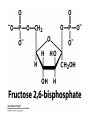
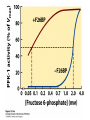
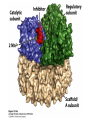
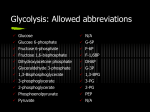
CHAPTER 15 Principles of Metabolic Regulation Key topics: – Principles of regulation in biological systems – Glycolysis vs. gluconeogenesis? Metabolic Pathways • The biochemical reactions in the living cell — the metabolism — is organized into metabolic pathways • The pathways have dedicated purposes – Some are dedicated to extraction of energy – Some are dedicated to storage of fuels – Some are dedicated for synthesis of important building blocks – Some are dedicated to elimination of waste materials • The pathways can be represented as a map – Follow the fate of metabolites and building blocks – Identify enzymes that act on these metabolites – Identify points and agents of regulation – Identify sources of metabolic diseases Map of Metabolic Pathways Homeostasis • Organisms maintain homeostasis by keeping the concentrations of most metabolites at steady state • In steady state, the rate of synthesis of a metabolite equals the rate of breakdown of this metabolite Principles of Regulation • The flow of metabolites through the pathways is regulated to maintain homeostasis • Sometimes, the levels of required metabolites must be altered very rapidly – Need to increase the capacity of glycolysis during the action – Need to reduce the capacity of glycolysis after the action – Need to increases the capacity of gluconeogenesis after successful action Feedback Inhibition • In many cases, ultimate products of metabolic pathways directly or indirectly inhibit their own biosynthetic pathways – ATP inhibits the commitment step of glycolysis Reactions Far From Equilibrium are Common Points of Regulation • Living systems thrive by keeping some metabolic reactions far from equilibrium while the levels of metabolites are in steady state Rates of a Biochemical Reactions • Rates of a biochemical reactions depend on many factors • Concentration of reactants • Activity of the catalyst – Concentration of the enzyme – Intrinsic activity of the enzyme • Concentrations of effectors – Allosteric regulators – Competing substrates – pH, ionic environment • Temperature Factors that Determine the Activity of Enzymes Active Protein Molecules have a Finite Lifespan • Different proteins in the same tissue have very different half-lives – less than an hour to about a week for liver enzymes – The stability correlates with the sequence at Nterminus • Some proteins are as old as you are – Crystallins in the eye lens Phosphorylation of Enzymes Affects their Activity • Protein phosphorylation is catalyzed by protein kinases • Dephosphorylation is spontaneous, or catalyzed by protein phosphatases • Typically, hydroxyl groups of Ser, Thr, or Tyr are phosphorylated Some Enzymes in the Pathway Limit the Flux of Metabolites More than Others • Hexokinase and phosphofructokinase are appropriate targets for regulation of glycolytic flux – Increased hexokinase activity enables activation of glucose – Increased phosphofructokinase-1 activity enables catabolism of activated glucose via glycolysis Control of Glycogen Synthesis • Insulin signaling pathway – increases glucose import into muscle – stimulates the activity of muscle hexokinase – activates glycogen synthase • Increased hexokinase activity enables activation of glucose • Glycogen synthase makes glycogen for energy storage Regulation of Hexokinase IV by Sequestration Rate of Reaction Depends on the Concentration of Substrates • The rate is more sensitive to concentration at low concentrations – Frequency of substrate meeting the enzyme matters • The rate becomes insensitive at high substrate concentrations – The enzyme is nearly saturated with substrate Isozymes may Show Different Kinetic Properties • Isozymes are different enzymes that catalyze the same reaction • They typically share similar sequences • Their regulation is often different Glycolysis vs. Gluconeogenesis Regulation of Phosphofructokinase-1 • The conversion of fructose-6-phosphate to fructose 1,6-bisphosphate is the commitment step in glycolysis • ATP is a negative effector – Do not spend glucose in glycolysis if there is plenty of ATP Phosphofructokinase-1 (PFK-1) Regulation of Phosphofructokinase 1 and Fructose 1,6-Bisphosphatase • Go glycolysis if AMP is high and ATP is low • Go gluconeogenesis if AMP is low Regulation by Fructose 2,6Bisphosphate • F26BP activates phosphofructokinase (glycolytic enzyme) • F26BP inhibits fructose 1,6-bisphosphatase (gluconeogenetic enzyme) Regulation by Fructose 2,6Bisphosphate • Go glycolysis if F26BP is high • Go gluconeogenesis if F26BP is low Regulation of 2,6-Bisphosphate Levels Molecular Origin of Enzyme Regulation • Regulation of catalysis typically involves – Binding of inhibitors, often to the active site – Binding of regulatory protein subunits Regulation of Pyruvate Kinase • Signs of abundant energy supply allosterically inhibit all pyruvate kinase isoforms • Signs of glucose depletion (glucagon) inactivate liver pyruvate kinase via phosphorylation – Glucose from liver is exported to brain and other vital organs Two Alternative Fates for Pyruvate • Pyruvate can be a source of new glucose – Store energy as glycogen – Generate NADPH via pentose phosphate pathway • Pyruvate can be a source of acetyl-CoA – Store energy as body fat – Make ATP via citric acid cycle • Acetyl-CoA stimulates glucose synthesis by activating pyruvate carboxylase Epinephrine and Glucagon Stimulate Breakdown of Glycogen