* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Thiazide and Thiazide
Toxicodynamics wikipedia , lookup
Nicotinic agonist wikipedia , lookup
Discovery and development of tubulin inhibitors wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
Drug discovery wikipedia , lookup
Development of analogs of thalidomide wikipedia , lookup
Discovery and development of direct Xa inhibitors wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Discovery and development of angiotensin receptor blockers wikipedia , lookup
Metalloprotease inhibitor wikipedia , lookup
Neuropharmacology wikipedia , lookup
Psychopharmacology wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Drug interaction wikipedia , lookup
Oral rehydration therapy wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
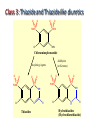
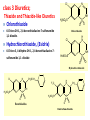
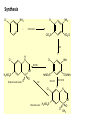
Lec-3 Class 3:Thiazide and Thiazide-like diuretics Thiazides and Thiazide-like diuretics • They were synthesized accidentally in an attempt to create more potent carbonic anhydrase inhibitors. • They act by inhibiting sodium reabsorption at the cortex level specifically at just distal to the loop of Henle. • They exert their effect from the luminal side of the nephron membrane thus, must be filtered to reach the site of action. Class 3: Thiazide and Thiazide-like diuretics O O O O H2NS SNH2 Cl NH2 Chloraminophenamide Aldehydes (or Ketones) Acylating Agents O O O H2NS O O O H2NS S O O S NH NH Cl N Thiazides R Cl N H R Hydrothiazides (Hydrochlorothiazide) Chloraminophenamide became a key intermediate in the development of diuretics that lack the undesirable properties of the CA inhibitors (???). When Chloraminophenamide was treated with acylating agents, cyclization resulted in the formation of Thiazides. The use of aldehydes or ketones in place of the acylating reagents yielded the corresponding dihydro derivatives. The products of these reactions became known as thiazides and hydrothiazides, respectively. The thiazides were the first orally effective saluretic agents diuretic activity was not influenced by the patient’s acid-base status. 4 Mechanism of Action inhibit Na+ and Cl- transporter in distal convoluted tubules increased Na+ and Cl- excretion increased K+/Mg2+ excretion decrease Ca2+ excretion Diuretics Thiazide and Thiazide-like Diuretics Mechanism of Action Actions: • Acts in the distal convoluted tubule – Inhibit tubular resorption of sodium, chloride, and potassium ions – Result: water, sodium, and chloride are excreted • Potassium is also excreted to a lesser extent • Dilate the arterioles by direct relaxation • Results: – Lowered peripheral vascular resistance – Sodium, water, chloride and potassium are excreted Overview of the group They are generally medium potency diretics. The first thiazide in the market is chlorthiazide suffered poor GI absorption and low bioavailability. Hydrochlorthiazide is the second member introduced to the market with higher bioavailability. The main side effects are the possibility of inducing slight hyperglycemia and hyperlipidemia. Overview of the group continued • Thiazide like drugs don’t have true thiazide ring but share the same mechanism of action and chemically related. • All members are available in oral forms except chlorthiazide ( oral and parentral): no clinical significance for their parentral forms. Cl class 3 Diuretics; Thiazide and Thiazide-like Diuretics Chlorothiazide N NH H2NO2S S 6-Chloro-2H-1, 2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Chlorothiazide H N Cl Hydrochlorothiazide, (Esidrix) 6-Chloro-3, 4-dihydro-2H-1, 2,4-benzothiadiazine-7- NH H2NO2S sulfonamide 1,1 -dioxide O O S O Hydrochlorothiazide Cl N CH2SCH2 F3C H N H2NO2S S CH2 NH H2NO2S S O NH O Benzthiazides O O Bendroflumethiazide O Structure-Activity Relationships: 4 5 Cl N 6 7 H2NO2S 3 2 S 8 O 1) The 2-position can tolerate small alkyl groups as CH3. 2) Substitutents in the 3-position determine the potency and duration of action of the thiazides. 3) Saturation of C-N bond between the 3 and 4 positions of the benzothiadiazine-1,1-dioxide nucleus increases the potency of this class of diuretics approximately 3-10 fold. 4) Direct substitution of the 4-, 5-, or 8-position with an alkyl group usually results in diminished diuretic activity, 5) Substitution of the 6-position with an activating group is essential for diuretic activity. The best substituent include Cl-, Br-, CF3-, and NO2- groups. 6) The sulfamoyl group in the 7-position is essential for diuretic activity. 1 NH O R Synthesis: Cl NH2 Cl NH2 ClSO2OH + ClO2S SO2Cl NH3 Cl H N Cl NH2 HCHO NH H2NO2S Hydrochlorothiazide O H2NO2S S O HCOCl H2 SO2NH2 HCOOH Cl N NH Chlorothiazide H2NO2S S O O OH2 Diuretics Thiazide and Thiazide-like Diuretics Indications • Hypertension – first line drug for HTN • Edematous states • Idiopathic hypercalciuria (condition characterized by recurrent stone formation in the kidneys due to excess calcium excretion) because thiazide diuretics used to prevent calcium loss and protect the kidneys • Diabetes insipidus Diuretics Thiazide and Thiazide-like Diuretics Adverse Effects Body System Adverse Effects CNS Dizziness, headache, blurred vision, paresthesias, decreased libido GI Anorexia, nausea, vomiting, diarrhea Integumentary Urticaria, photosensitivity Metabolic Hypokalemia, glycosuria, hyperglycemia, hyperuricemia Thiazide-like diuretics • The sulfamoyl group para to the activating group of thiazides could be replaced by several other electronegative groups (X-) with retention of diuretic activity (as R = amide, carbonyl, carboxyl groups, etc) in the meta-disulfamoylbenzene, • These diuretics known as thiazide-like diuretics. • Their site of action, efficacy, electrolyte excretion pattern, and adverse effects resemble the thiazides. X H2NO2S R' SO2NH2 X R' H2NO2S R Quinazoline derivatives 1- Metolazone •is a thiazide like drug • It is primarily used to treat congestive hear failure and HTN • Metolazone indirectly decreases the amount of water reabsorbed into the bloodstream by the kidney, so that blood volume decreases and urine volume increases. This lowers blood pressure and prevents excess fluid accumulation in heart failure. • Metolazone is sometimes used together with loop diuretics such as furosemide, but these highly effective combinations can lead to dehydration and electrolyte abnormalities. Quinazoline derivatives 2-Chlorthalidone • A diuretic with actions and uses similar to those of the thiazide diuretics even though it does not contain a thiazide ring system • A benzenesulfonamide-phthalimidine that tautomerizes to a BENZOPHENONES form. It is considered a thiazide-like diuretic.