* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download hypothalamic neuroanatomy and limbic inputs
Apical dendrite wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Causes of transsexuality wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Artificial general intelligence wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Synaptogenesis wikipedia , lookup
Neuroeconomics wikipedia , lookup
Single-unit recording wikipedia , lookup
Aging brain wikipedia , lookup
Subventricular zone wikipedia , lookup
Mirror neuron wikipedia , lookup
Neural coding wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neural oscillation wikipedia , lookup
Axon guidance wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Multielectrode array wikipedia , lookup
Electrophysiology wikipedia , lookup
Anatomy of the cerebellum wikipedia , lookup
Central pattern generator wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Development of the nervous system wikipedia , lookup
Nervous system network models wikipedia , lookup
Limbic system wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Synaptic gating wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Optogenetics wikipedia , lookup
Neuroanatomy wikipedia , lookup
Channelrhodopsin wikipedia , lookup
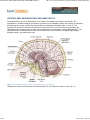
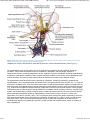
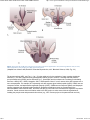
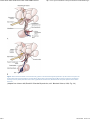
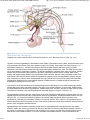
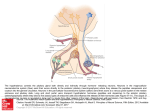
//HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS The hypothalamus is part of the diencephalon. It lies rostral to the midbrain and caudal to the forebrain. The hypothalamus is bounded dorsally by the thalamus, posteriorly by the mammillary bodies, and anteriorly by the lamina terminalis and optic chiasm, and the third ventricle splits the hypothalamus bilaterally (Figs. 1-1 and 1-2). The hypothalamus receives rich input from the autonomic areas and reticular nuclei of the brain stem, particularly the catecholaminergic cell groups (many of which have neuropeptides as cotransmitters, such as galanin and NPY). The hypothalamus also receives dense innervation from the limbic areas of the forebrain, including the hippocampus, amygdala, septum, and orbitofrontal cortex. Figure 1-1 Saggital section of the human brain, including the pituitary and pineal glands. (Adapted from Johnson MH, Everitt BJ. Essential Reproduction, ed 5. Blackwell Science, 2000, Fig. 6.1.) 1 de 6 02/03/2011 11:45 a.m. //HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... Figure 1-2 Schematic, three-dimensional view of the human hypothalamus, pituitary, and portal capillary system showing the approximate locations of the major nuclei. GnRH, gonadotropin-releasing hormone. (Adapted from Johnson MH, Everitt BJ. Essential Reproduction. Oxford, Blackwell Science, 2000, Fig. 6.2.) The hypothalamus serves as the primary site for the integration and regulation of many important physiologic processes. These include homeostatic control of temperature, metabolism, and body weight, aspects of cardiovascular function, physiologic adaptation to stress, regulation of growth, reproduction (including sexual behavior), and lactation. Although the regulation of these complex processes depends on the circuitry of the hypothalamus (and its afferent inputs), the control of these systems cannot be defined on the basis of strict anatomic criteria. The hypothalamus comprises distinct nuclei (collections of cell bodies), including the supraoptic, paraventricular, suprachiasmatic, ventro- and dorsomedial, and arcuate nuclei. The suprachiasmatic nucleus (SCN; Fig. 1-3) is the site of the brain's circadian “clock.” Cells in the SCN receive input from the retinohypothalamic pathway, through which the brain keeps track of the diurnal rhythm of light and dark and controls rhythmic cycles of activity and hormone secretion (e.g., sleep–wake and CRH/ACTH/cortisol rhythms). Subgroups of neurons in the SCN that express VIP and arginine vasopressin project to different parts of the hypothalamus to coordinate diverse physiologic functions, including activity rhythms and the preovulatory GnRH/LH surge (at least in rodent and ruminant species). Neurons in the SCN express genes that display endogenous rhythmicity, approximating a 24-hour period—hence, the term circadian, with components circa (“about”) and dian (“day”). The clock and per genes, among many others expressed by cells in the SCN, generate pacemaker activity, which may be entrained by external cues (e.g., light or activity) to help the body keep track of time. Cells in the SCN express estrogen receptor α (ERα) and are believed to be involved in the neuroendocrine regulation of gonadotropin secretion, at least in rodent and ruminant species, where the evidence is most compelling.[4-6] 2 de 6 02/03/2011 11:45 a.m. //HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... Figure 1-3 Rostral (A), mid (B), and caudal (C) coronal sections of the human hypothalamus. AHA, anterior hypothalamic area; LAT HYP, lateral hypothalamus; POA, preoptic area; VMN, ventromedial nucleus. (Adapted from Johnson MH, Everitt BJ. Essential Reproduction, ed 5. Blackwell Science, 2000, Fig. 6.3.) The arcuate nucleus (ARC; see Figs. 1-2 to 1-5) is the nodal point for the regulation of many complex physiologic functions.[7-9] The ARC comprises many phenotypically distinct groups of neurons—including cells that produce pro-opiomelanocortin (POMC) and its derivatives (e.g., β-endorphin and the melanocortin α-melanocyte–stimulating hormone, α-MSH), NPY, GHRH, kisspeptin, GALP, and dopamine. Most, if not all, neurons in the ARC express two or more neuropeptides. For example, NPY-expressing cells also express agouti-related peptide (AgRP). POMC neurons coexpress cocaine- and amphetamine-regulated transcript (CART). GHRH neurons coexpress galanin, and kisspeptin neurons coexpress both dynorphin and neurokinin B. Dopamine-containing neurons are concentrated in the tuberoinfundibular track within the ARC, and these cells play a critical role in the neuroendocrine regulation of prolactin secretion. Certain neurons whose cell bodies reside in the ARC project to other areas within the hypothalamus, including the preoptic area and paraventricular nucleus (e.g., NPY neurons project to the paraventricular nucleus). 3 de 6 02/03/2011 11:45 a.m. //HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... Figure 1-4 Schematic illustration of the neurosecretory systems in the human that regulate reproduction. A, The locations of oxytocin cell bodies, which reside in the hypothalamus, and their fibers, which project to the neurohypophysis. B, The primary locations of gonadotropinreleasing hormone (GnRH) cell bodies in the human hypothalamus and their axons, which terminate near portal capillaries in the median eminence. (Adapted from Johnson MH, Everitt BJ. Essential Reproduction, ed 5. Blackwell Science, 2000, Fig. 6.4.) 4 de 6 02/03/2011 11:45 a.m. //HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... Figure 1-5 Diagram of some of the neurotransmitter systems that are believed to play a role in regulating gonadotropin-releasing hormone (GnRH) secretion. GABA, γ-aminobutyric acid. (Adapted from Johnson MH, Everitt BJ. Essential Reproduction, ed 5. Blackwell Science, 2000, Fig. 6.18.) Together, the lateral hypothalamus, dorsomedial nucleus (DMN), ventromedial nucleus (VMN), and parvocellular region of the paraventricular nucleus (PVN) exert regulatory control over feeding, body weight, and activity rhythms.[10] In experimental animals (e.g., rats and cats) lesions of the VMN stimulate appetite and cause obesity, whereas stimulation of the VMN reduces feeding and body weight subsequently declines as a result. The VMN may also play a role in sexual behavior, particularly in females. The lateral hypothalamus comprises other unique cell groups, including neurons that produce orexins (also known as hypocretins), which have profound effects on sleep–wake cycles, feeding, and reward-seeking behavior, and can influence GnRH secretion. Neurons in the parvocellular region of the PVN produce TRH and CRH, which regulate the hypothalamic–pituitary–thyroid and hypothalamic–pituitary–adrenal axes, respectively, but both of these neuropeptides also play a critical role in the control of feeding and metabolism. CRH has been implicated in the stress-induced inhibition of GnRH secretion—perhaps through its interaction with β-endorphin–producing neurons in the ARC. Just rostral to the formal boundaries of the hypothalamus lies the medial preoptic area, which contains many GnRH neurons that project (along with GnRH neurons in the ARC) to the median eminence. (In the primate, GnRH neurons are widely dispersed in the anterior hypothalamus, medial preoptic area, and ARC, whereas in rodent species, GnRH cell bodies are restricted to the rostral hypothalamus and medial preoptic area [Figs. 1-4B and 1-5]). The hypothalamus receives input from many regions of the brain. Ascending noradrenergic projections arise from the medulla and pons and innervate many nuclear groups within the hypothalamus, including the medial preoptic area and ARC (see Fig. 1-5). Serotonin projections originate in the midbrain raphe and provide dense innervation of the hypothalamus, particularly the mammillary complex, periventricular nucleus, ARC, and SCN. The hypothalamus also receives descending input from several sources. These include projections from the basal forebrain, olfactory tubercle, piriform cortex, amygdala, and hippocampus. Hypophysiotropic neurons whose cell bodies reside within the hypothalamus send their projections to the median eminence, where their secretory products enter the portal vasculature and thus regulate anterior pituitary function. In the context of reproduction, GnRH neurons, with cell bodies in the preoptic area and ARC, send projections into the external zone of the median eminence. From nerve terminals in the median eminence, GnRH is secreted into the 5 de 6 02/03/2011 11:45 a.m. //HYPOTHALAMIC NEUROANATOMY AND LIMBIC INPUTS http://www.expertconsultbook.com/expertconsult/b/book.do?method=get... fenestrated capillaries and transported to the anterior pituitary (see Figs. 1-4B and 1-5). Kisspeptin neurons (whose cell bodies are in the ARC and rostral hypothalamus) interact with GnRH neurons by projecting directly to GnRH cell bodies, but may also send axoaxonal projections into the zona internal of the median eminence and thereby influence GnRH secretion by several different mechanisms (see Fig. 1-5). Magnocellular neurons in the PVN and supraoptic nucleus (SON) send long axons into the neurohypophysis, where they release vasopressin and oxytocin into the vasculature (see Fig. 1-4A). Copyright © 2011 Elsevier Inc. All rights reserved. Read our Terms and Conditions of Use and our Privacy Policy. For problems or suggestions concerning this service, please contact: [email protected] 6 de 6 02/03/2011 11:45 a.m.