* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 5 PowerPoint
Peptide synthesis wikipedia , lookup
Western blot wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Gene expression wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Phosphorylation wikipedia , lookup
Metalloprotein wikipedia , lookup
Point mutation wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Genetic code wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Protein structure prediction wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Proteolysis wikipedia , lookup
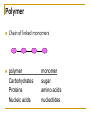
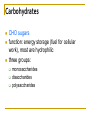
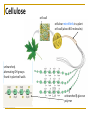
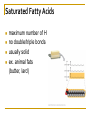
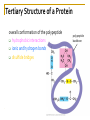
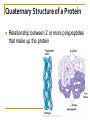
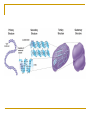
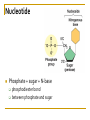
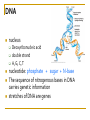
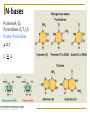
Chapter 5 Macromolecules Macromolecules Four Classes 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic acids Polymer Chain of linked monomers polymer Carbohydrates Proteins Nucleic acids monomer sugar amino acids nucleotides Composite molecule Lipids glycerol + fatty acid(s) Bonds macromolecule Carbohydrates Lipids Proteins Nucleic acids bond/link type glycosidic linkage ester linkage peptide bond phosphodiester bond Chemical Reactions Dehydration synthesis (Condensation) - remove a water molecule, polymerize Requires enzymes HO 1 2 3 H H OH short polymer H20 HO 1 2 longer polymer 3 4 H Chemical Reactions Hydrolysis - add a water molecule, depolymerize HO H H2O HO H OH H Metabolism Anabolism - synthesize molecules polymerization dehydration Catabolism - breakdown molecules depolymerization hydrolysis Carbohydrates CHO sugars function: energy storage (fuel for cellular work), most are hydrophilic three groups: monosaccharides disaccharides polysaccharides Monosaccharides empirical formula = (CH2O) n n = 3 to 7 n=5 pentose n=6 hexose Examples of hexoses (C6H12O6) glucose fructose galactose Disaccharides monosaccharides glucose + glucose = glucose + galactose = glucose + fructose = disaccharide maltose lactose sucrose formed through dehydration Formation of Disaccharaides glucose glucose glucose fructose maltose sucrose Polysaccharide Covalently bonded glucose units Glycosidic linkage - covalent bond between glucose units Glycogen (animals) Starch (plants) glycogen granules amylose (unbranched) amylopectin Cellulose cell wall cellulose microfibrils in a plant cell wall (about 8O molecules) unbranched, alternating OH groups found in plant cell walls unbranched β glucose polymer Chitin Present in exoskeleton of arthropods Glucose plus a nitrogen apendage used to make strong and flexible surgical thread Lipids CHO Function: long-term energy storage fats other lipids: phospholipids, steroids, pigments water-insoluble, nonpolar Fats Glycerol + fatty acid (s) monoglyceride diglyceride triglyceride monoglyceride (palmitic acid) glycerol - 3C alcohol trigryceride (triacylglycerol) Fats, cont. ester linkage - bond between carboxyl (f.a.) and hydroxyl (glycerol) fatty acids saturated unsaturated esther linkage Saturated Fatty Acids maximum number of H no double/triple bonds usually solid ex. animal fats (butter, lard) Unsaturated Fatty Acids usually liquid 1 double bond more than 1 double bond monounsaturated polyunsaturated ex. Oils (nut and seed) hydrogenation causes it to solidify peanut oil peanut butter fats yeald 9 cal/gr oleic acid Other Lipids Phospholipids Steroids phosphate + 2 f.a. + glycerol Hormones and cholesterol Pigments chlorophylls xanthophylls carotenes anthocyanins hydrophilic hydrophobic choline phosphate glycerol fatty acids Proteins CHON Function: diverse structures, contraction, storage, defense, transport, messengers, enzymes monomer: amino acid Proteins cont. amino acids (20): most contain -central C -amino group -carboxyl group -H atom -functional group (R) R-group Non polar glycine alanine methionine valine phenylalanine leucine tryptophan isoleusine proline polar serine threonine cysteine tyrosine aspargine glutamine acidic basic electrically charged asparctic ac. glutamic acid lysine arginine histine Proteins cont. *Peptide bonds *between 2 amino acids *dehydration reaction protein synthesis side chain backbone Levels of Protein Structure The following determine the shape of the protein (polypeptides): primary secondary tertiary quaternary Primary Structure of a Protein Start: N-terminus (amino) End: C-terminus (carboxyl) +H3N (amino end) amino acid subunits (carboxyl end) O C OH- Secondary Structure of a Protein Bending and H-bonding to form coils and folds forming alpha helices (coils) and beta pleated sheets (folds) β pleated sheet α- helix Tertiary Structure of a Protein overall conformation of the polypeptide hydrophobic interactions ionic and hydrogen bonds disulfide bridges polypeptide backbone Quaternary Structure of a Protein Relationship between 2 or more polypeptides that make up the protein Protein Conformation Altering any level will change the final conformation denaturation - unravel the protein caused by several different factors Nucleic Acids CHONP Function: store information monomer - nucleotide ex. DNA, RNA RNA Genomics - branch of biotech concerned with mapping and sequencing DNA (genomes) Nucleotide Phosphate + sugar + N-base phosphodiester bond between phosphate and sugar DNA nucleus Deoxyribonucleic acid double strand A, G, C, T nucleotide: phosphate + sugar + N-base The sequence of nitrogenous bases in DNA carries genetic information stretches of DNA are genes RNA cytoplasm Ribonucleic acid single strand RNA nucleotide: phosphate + sugar + A, G, C, U N-base N-bases Purines (A, G) Pyrimidines (C, T, U) Purine-Pyrimidine =T C=G A Activity: DNA What will the other strand be? 5' A 3' DNA ? G ? C ? T ? DNA Activity What will the RNA strand be? 5' A 3' DNA The End ? G ? C ? T ? RNA