* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Sequence of events in formation of eukaryotic mRNA
X-inactivation wikipedia , lookup
Genomic imprinting wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Non-coding DNA wikipedia , lookup
Ridge (biology) wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Gene regulatory network wikipedia , lookup
RNA interference wikipedia , lookup
Genome evolution wikipedia , lookup
Molecular evolution wikipedia , lookup
Messenger RNA wikipedia , lookup
Community fingerprinting wikipedia , lookup
Gene expression profiling wikipedia , lookup
Alternative splicing wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
RNA polymerase II holoenzyme wikipedia , lookup
RNA silencing wikipedia , lookup
Eukaryotic transcription wikipedia , lookup
Polyadenylation wikipedia , lookup
Promoter (genetics) wikipedia , lookup
Transcriptional regulation wikipedia , lookup
Gene expression wikipedia , lookup
Silencer (genetics) wikipedia , lookup
Non-coding RNA wikipedia , lookup
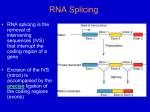
Study Guide/Outline—RNA Processing RNA Processing: rRNA genes, tRNA genes, self-splicing, spliceosomal Structural genes Pre-RNA Processing •What kind of processing must occur to pre-mRNA in eukaryotes? Where does this take place? •What happens to the RNA molecule if it does not undergo processing? •Purpose of each RNA processing step –What is a methyl guanine “cap”? –How is a polyA tail added? How does this tail contribute to the stability of the mRNA? •How are nucleotides numbered in genes with exons and introns? Spliceosomes •What is a spliceosome and what class of genes use spliceosomes? •What consensus sequences are needed in introns in order for correct splicing to occur? What would happen if there was a mutation in a splice site consensus sequence? •What is the significance of the lariat structure in splicing out introns? mRNA and protein synthesis are coupled in bacteria In eukaryotes mRNA must be processed and transported out of nucleus for translation iGenetics, 1st ed. Russell Prokaryotes vs. Eukaryotes Prokaryotic • Polycistronic (one promoter, multiple genes) • Introns thought to be nonexistent in prokaryotes until very recently • Transcription and translation can occur concordantly • Exceptions: archaebacteria, bacteriaphage (virus), mitochondria, chloroplasts Eukaryotic • Monocistronic (one promoter, one gene) • Introns are common • High amounts of “junk DNA” in genome. • RNA requires significant processing • Size of introns is roughly correlated with complexity of the organism. Structure of the methylguanine cap 5 O 4 1 2 3 5 O 4 1 3 2 5 O 4 1 3 2 Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. RNA polymerase II transcribes a gene past the polyA signal sequence. 5′ 3′ PolyA signal sequence The RNA is cleaved just past the polyA signal sequence. RNA polymerase continues transcribing the DNA. 5′ 3′ 5′ 3′ Torpedo model: An exonuclease binds to the 5′ end of the RNA that is still being transcribed and degrades it in a 5′ to 3′ direction. Allosteric model: After passing the polyA signal sequence, RNA polymerase II is destabilized due to the release of elongation factors or the binding of termination factors (not shown). Termination occurs. 5′ 3′ 5′ 3′ 5′ 3′ Exonuclease catches Exonuclease up to RNA polymerase II and causes termination. 3′ 5′ 3′ Figure 14.15 14 - 47 Polyadenylation signal sequence 5′ 3′ AAUAAA Consensus sequence in higher eukaryotes 5′ Endonuclease cleavage occurs about 20 nucleotides downstream from the AAUAAA sequence. AAUAAA PolyA-polymerase adds adenine nucleotides to the 3′ end. 5′ AAUAAA AAAAAAAAAAAA.... 3′ PolyA tail Brooker Figure 14.22 Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display Termination of RNA synthesis in (eukaryotic) RNA Pol II 3’ UTR Regular transcript Peter J. Russell, iGenetics: Copyright © Pearson Education, Inc., publishing as Benjamin Cummings. Site of cleavage and addition of polyA tail Animation of cap addition and poly-A tail addition http://vcell.ndsu.nodak.edu/animations/ http://vcell.ndsu.nodak.edu/animations/ Length of poly-A Tail and Stability (half-life of mRNA) Prokaryotic Degradation at 5’ end begins immediately (before 3’ transcription is completed) Eukaryotic cFOS mRNA (cell Half-life: 10-30 cycle gene) min Hemoglobin Short poly-A tail Half-life: 24 hours Long poly-A tail Major types of introns Type of intron Gene type Splicing Mechanism Enzymatic tRNAs and rRNAs Nuclear (premRNA) tRNA genes Protein-encoding genes in nuclear chromosomes Spliceosomal Group I Some rRNA genes Self-splicing Group II Protein-encoding genes in mitochondria Self-splicing Processing of ribosomal RNA Promoter 18S 5.8S 28S Transcription 5′ 45S rRNA transcript 18S 5.8S 28S Cleavage (the light pink regions are degraded) This processing occurs in the nucleolus 18S rRNA 5.8S rRNA 28S rRNA Functional RNAs that are key in ribosome structure Brooker Figure 14.16 Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display 3′ Processing of tRNAs Endonuclease 5′ Endonuclease (RNaseP) mG 3′ A C C Exonuclease (RNaseD) T T P Covalently modified bases P mG IP = Methylguanosine P = Pseudouridine T = 4-Thiouridine Brooker Figure 14.17 Anticodon IP = 2-Isopentenyladenosine Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display RNA-DNA hybrid reveals intron sequences as they “loop out” Splice site consensus sequences Exon 5′ Intron A/ GGU C Pu AGUA 5′ splice site Exon UACUUAUCC Py12N Py AGG Branch site 3′ splice site Brooker Figure 14.19 Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display 3′ Rough overview of splicing mechanism (formation of lariat structure) Mechanism of Spliceosome Mechanism of Spliceosome (Brooker) Exon 1 Exon 2 A GU 5′ AG 5′ splice site Branch site 3′ 3′ splice site U1 binds to 5′ splice site. U2 binds to branch site. U1 snRNP U2 snRNP A 5′ Intron loops out and exons brought closer together 3′ U4/U6 and U5 trimer binds. Intron loops out and exons are brought closer together. A U2 U4/U6 snRNP U1 5′ U5 snRNP 3′ Brooker, Fig 14.20 Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display Mechanism of Spliceosome (Brooker), cont. 5′ splice site is cut. 5′ end of intron is connected to the A in the branch site to form a lariat. U1 and U4 are released. U1 U4 U2 A U6 U5 5′ 3′ 3′ splice site is cut. Exon 1 is connected to exon 2. The intron (in the form of a lariat) is released along with U2, U5, and U6 (intron will be degraded). Intron will be degraded and the snRNPs used again U2 A Intron plus U2, U5, and U6 U6 5′ Exon 1 U5 Exon 2 3′ Two connected exons Brooker, Fig 14.20 Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display Rare mutations in intron sequences can cause a phenotype (e.g. in the large gene, Dystrophin) Duchenne’s Muscular Dystrophy (X-linked) •Caused by mutations in Dystrophin gene: •2+ million nt gene (many exons) •Relatively high incidence (1/3500 males) due to large size of gene and hemizygosity in males Advanced Duchenne’s Muscular Dystrophy Fig from Medical Genetics, Jorde et al., 3rd ed. Major types of introns Type of intron Gene type Splicing Mechanism Enzymatic tRNAs and rRNAs Nuclear (premRNA) tRNA genes Protein-encoding genes in nuclear chromosomes Spliceosomal Group I Some rRNA genes Self-splicing Group II Protein-encoding genes in mitochondria Self-splicing Self splicing Introns Self-splicing introns (relatively uncommon) CH2OH O H 3′ G H Intron Guanosine binding site G Intron H OH OH Guanosine 5′ A Exon 1 G Exon 2 G 3′ 5′ Exon 1 HH O O (a) Group I P O CH2 O HH O O 5′ RNA 3′ 3′ P H 2′ P 3′ Exon 2 P O CH2 O A 5′ P 3′ OH G 5′G O H 2′ 5′ 3′ OH HH OH P 5′ O H 2′ A 3′ 5′ G P O CH2 P 3′ RNA (b) Group II Brooker Fig 14.18a and b Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display Go over lecture outline at end of lecture