South Pasadena · AP Chemistry
... stock 280 tables, 1750 chairs, 550 bookshelves, 300 china cabinets, and 325 sideboards. He asked his assistant to figure out how many dining room sets they could sell, how much money they would make if they sold all the sets possible, and what they would have left that could not be sold as part of t ...
... stock 280 tables, 1750 chairs, 550 bookshelves, 300 china cabinets, and 325 sideboards. He asked his assistant to figure out how many dining room sets they could sell, how much money they would make if they sold all the sets possible, and what they would have left that could not be sold as part of t ...
BIOLOGY 1 QUIZ REVIEW SHEET CHAPTER 4.4
... AGAIN AND _______H+___ IONS GO IN AND OUT OF THE INNER MITOCHONDRIAL MEMBRANE. THE _______H+___ IONS ADD TO OXYGEN TO MAKE ______H2O_________ WHICH IS A FINAL PRODUCT. THEN ATP, NADH AND FADH2 ARE USED TO MAKE MORE ATP…UP TO _____36_______ ATP. ...
... AGAIN AND _______H+___ IONS GO IN AND OUT OF THE INNER MITOCHONDRIAL MEMBRANE. THE _______H+___ IONS ADD TO OXYGEN TO MAKE ______H2O_________ WHICH IS A FINAL PRODUCT. THEN ATP, NADH AND FADH2 ARE USED TO MAKE MORE ATP…UP TO _____36_______ ATP. ...
Rest to Exercise Transition
... Why is the aerobic system activated so slowly – Theory 1 Inadequate oxygen molecules in mitochondria Oxygen can’t accept electrons in ETC – H2O If true, then oxygen molecules low in whole body ...
... Why is the aerobic system activated so slowly – Theory 1 Inadequate oxygen molecules in mitochondria Oxygen can’t accept electrons in ETC – H2O If true, then oxygen molecules low in whole body ...
Cellular Respiration
... 1. Pyruvic Acid is converted into Acetyl CoA. 2. This joins with oxaloacetic acid to form citric acid. 3. Citric Acid goes through a cycle where CO2 and electron carriers are formed. 4. The 2 original pyruvic acid molecules are completely broken down into CO2 ...
... 1. Pyruvic Acid is converted into Acetyl CoA. 2. This joins with oxaloacetic acid to form citric acid. 3. Citric Acid goes through a cycle where CO2 and electron carriers are formed. 4. The 2 original pyruvic acid molecules are completely broken down into CO2 ...
Chapter 3
... pathway to synthesize ATP in the absence of sufficient oxygen. 3. The third group of bacteria consists of the obligate anaerobes, such as Clostridium tetani, which cannot grow in the presence of oxygen because they lack either superoxide dismutase or catalase, or both. ● Obligate anaerobes vary in t ...
... pathway to synthesize ATP in the absence of sufficient oxygen. 3. The third group of bacteria consists of the obligate anaerobes, such as Clostridium tetani, which cannot grow in the presence of oxygen because they lack either superoxide dismutase or catalase, or both. ● Obligate anaerobes vary in t ...
Virginia Beach Department of EMS Oxygen and Oxygen
... The failure to ensure compliance with these measures may hamper the ability of the oxygen delivery equipment to work properly and, in some cases, may have the serious potential to cause a fire or explosion or deterioration of patient condition. Installed Oxygen Systems With Humidifiers The Departmen ...
... The failure to ensure compliance with these measures may hamper the ability of the oxygen delivery equipment to work properly and, in some cases, may have the serious potential to cause a fire or explosion or deterioration of patient condition. Installed Oxygen Systems With Humidifiers The Departmen ...
Powering the Cell: Cellular Respiration and Glycolysis/Practice!
... Critical Reading Read this passage from the lesson and answer the questions that follow. The Electron Transport Chain: ATP for Life in the Fast Lane At the end of the Krebs Cycle, energy from the chemical bonds of glucose is stored in diverse energy carrier molecules: four ATP, but also two FADH2 a ...
... Critical Reading Read this passage from the lesson and answer the questions that follow. The Electron Transport Chain: ATP for Life in the Fast Lane At the end of the Krebs Cycle, energy from the chemical bonds of glucose is stored in diverse energy carrier molecules: four ATP, but also two FADH2 a ...
Non-Metals
... It is a diatomic gas Cl2 It is a greenish-yellow gas with a choking odour . It is more dense than air and is slightly soluble in water . ...
... It is a diatomic gas Cl2 It is a greenish-yellow gas with a choking odour . It is more dense than air and is slightly soluble in water . ...
Hemoglobin and Myoglobin
... The energy in the formation of the Fe-O2 bond formation drives the T R transition. Hemoglobins O2 -binding Cooperativity derives from the T R Conformational shift. •The Fe of any subunit cannot move into its heme plane without the reorientation of its proximal His so as to prevent this residue ...
... The energy in the formation of the Fe-O2 bond formation drives the T R transition. Hemoglobins O2 -binding Cooperativity derives from the T R Conformational shift. •The Fe of any subunit cannot move into its heme plane without the reorientation of its proximal His so as to prevent this residue ...
Structure and Properties of Hemoglobin Learning Objectives What
... of ionic & hydrogen bonds b/w dimers and have more freedom of movement ...
... of ionic & hydrogen bonds b/w dimers and have more freedom of movement ...
File
... 3 Steps of Aerobic Cellular Respiration 2 Types of Fermentation 1 Main Goal of Cellular Respiration ...
... 3 Steps of Aerobic Cellular Respiration 2 Types of Fermentation 1 Main Goal of Cellular Respiration ...
Honors Chemistry Atomic Theory Reading
... A man named John Dalton, (to the left) discovered this limitation in the law of definite proportions in some of his experiments. Dalton was experimenting with several reactions in which the reactant elements formed different products, depending on the experimental conditions he used. One common reac ...
... A man named John Dalton, (to the left) discovered this limitation in the law of definite proportions in some of his experiments. Dalton was experimenting with several reactions in which the reactant elements formed different products, depending on the experimental conditions he used. One common reac ...
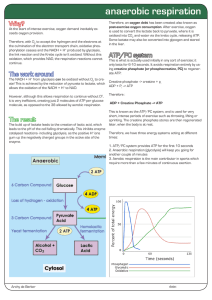
anaerobic respiration
... Some lactate may also be converted into glycogen and stored in the liver. ...
... Some lactate may also be converted into glycogen and stored in the liver. ...
Bacterial Growth and Nutrition
... • Bacteria acquire energy from oxidation of organic or inorganic molecules, or from sunlight. • Growth requires raw materials: some form of carbon. • Autotrophs vs. heterotrophs – Auto=self; hetero=other; troph=feeding. – Autotrophs use carbon dioxide – Heterotrophs use pre-formed organic compounds ...
... • Bacteria acquire energy from oxidation of organic or inorganic molecules, or from sunlight. • Growth requires raw materials: some form of carbon. • Autotrophs vs. heterotrophs – Auto=self; hetero=other; troph=feeding. – Autotrophs use carbon dioxide – Heterotrophs use pre-formed organic compounds ...
Photosynthesis and Cellular Respiration
... Plants capture energy by absorbing light and using it to form strong (covalent) chemical bonds between the atoms of carboncontaining (organic) molecules. These molecules can be used to assemble larger molecules. ...
... Plants capture energy by absorbing light and using it to form strong (covalent) chemical bonds between the atoms of carboncontaining (organic) molecules. These molecules can be used to assemble larger molecules. ...
BIO 219 Spring 2013 Outline for “Cell Metabolism” Energy (ATP
... NADH & FADH2 Oxygen (Why do we need it?) Net yield Fermentation Glycolysis Lactic acid production What about fats and proteins as an energy source? Objectives: By the end of lecture today you should be able to . . . (1) Describe the similarities and differences between aerobic and anaerobic metaboli ...
... NADH & FADH2 Oxygen (Why do we need it?) Net yield Fermentation Glycolysis Lactic acid production What about fats and proteins as an energy source? Objectives: By the end of lecture today you should be able to . . . (1) Describe the similarities and differences between aerobic and anaerobic metaboli ...
In organic chemistry, we studied a lot about the essential elements
... Phosphorus, Sulfur. And these elements play a very essential in living organism. HYDROGEN: It’s a very universal knowledge that hydrogen and oxygen forms water, an essential substance for life on Earth. However this element is very important in so much organic chemical compounds and an important ele ...
... Phosphorus, Sulfur. And these elements play a very essential in living organism. HYDROGEN: It’s a very universal knowledge that hydrogen and oxygen forms water, an essential substance for life on Earth. However this element is very important in so much organic chemical compounds and an important ele ...
Reacting Mass calculations
... What mass of iodine is produced when 7.1 g of chlorine reacts with excess potassium iodide? Cl2 + 2 KI 2 KCl + I2 ...
... What mass of iodine is produced when 7.1 g of chlorine reacts with excess potassium iodide? Cl2 + 2 KI 2 KCl + I2 ...
Cellular Respiration Releases Energy from Organic Compounds
... last set of reactions in electron transport So why is it so important??? ...
... last set of reactions in electron transport So why is it so important??? ...
Name: #: Cellular Respiration Review 2 Process Where does it
... 6. Write the complete overall chemical equation for cellular respiration using chemical symbols instead of words: 6O2 + C6H12O6 6H2O + 6CO2 + 36ATP 7. Why do we say there is a ‘net’ gain of 2 ATP at the end of glycolysis? Glycolysis produces 4ATP but since it needs 2 ATP to start, the cell only in ...
... 6. Write the complete overall chemical equation for cellular respiration using chemical symbols instead of words: 6O2 + C6H12O6 6H2O + 6CO2 + 36ATP 7. Why do we say there is a ‘net’ gain of 2 ATP at the end of glycolysis? Glycolysis produces 4ATP but since it needs 2 ATP to start, the cell only in ...
Cellular Respiration Name: Period: ______ Date: 1. Define cellular
... 32. The high energy electrons from the Krebs Cycle are passed to the _________________________________________ 33. What is the function of the electron transport chain? ___________________________________________________ 34. Where is the electron transport chain located in eukaryotes? ______________ ...
... 32. The high energy electrons from the Krebs Cycle are passed to the _________________________________________ 33. What is the function of the electron transport chain? ___________________________________________________ 34. Where is the electron transport chain located in eukaryotes? ______________ ...
Cellular Respiration
... the heart and vessels are responsible to pump and transport all nutrients to all parts of the body through tiny vessels called capillaries are things like glucose, oxygen and carbon dioxide able to enter or exit the tissue or vessel the only way to get glucose (from the intestine) and oxygen (from t ...
... the heart and vessels are responsible to pump and transport all nutrients to all parts of the body through tiny vessels called capillaries are things like glucose, oxygen and carbon dioxide able to enter or exit the tissue or vessel the only way to get glucose (from the intestine) and oxygen (from t ...
Cellular Respiration
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
Oxygen Metabolism and Oxygen Toxicity
... Glycogen stores are rapidly depleted, and lactic acid concentrations increase, lowering the cellular pH (lactic acidosis). Hypoxic cells begin to swell as they can no longer maintain their normal electrolyte concentrations. The mitochondria begin to swell and accumulate calcium which precipitates as ...
... Glycogen stores are rapidly depleted, and lactic acid concentrations increase, lowering the cellular pH (lactic acidosis). Hypoxic cells begin to swell as they can no longer maintain their normal electrolyte concentrations. The mitochondria begin to swell and accumulate calcium which precipitates as ...
Oxygen
Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements. Photosynthesis releases oxygen, and respiration consumes oxygen. Changes in phosphate are related to changes in oxygen concentrations.Oxygen was discovered independently by Carl Wilhelm Scheele, in Uppsala, in 1773 or earlier, and Joseph Priestley in Wiltshire, in 1774, but Priestley is often given priority because his work was published first. The name oxygen was coined in 1777 by Antoine Lavoisier, whose experiments with oxygen helped to discredit the then-popular phlogiston theory of combustion and corrosion. Its name derives from the Greek roots ὀξύς oxys, ""acid"", literally ""sharp"", referring to the sour taste of acids and -γενής -genes, ""producer"", literally ""begetter"", because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition.