USATestprep, Inc.
... C) must occur before plants are able to carry out photosynthesis. D) is performed only by organisms that are incapable of photosynthesis. 2) What are the reactants of cellular respiration? A) ATP and water B) sugar and oxygen C) NADPH and hydrogen D) carbon dioxide and water 3) On average, how many ...
... C) must occur before plants are able to carry out photosynthesis. D) is performed only by organisms that are incapable of photosynthesis. 2) What are the reactants of cellular respiration? A) ATP and water B) sugar and oxygen C) NADPH and hydrogen D) carbon dioxide and water 3) On average, how many ...
Nitrous Oxide/Oxygen
... • Blended mixture of 50% nitrous oxide and 50% oxygen • Also known as “laughing gas” • Produces sedation and analgesia • Colorless, odorless, heavier than air • Nonexplosive, nonflammable • Readily diffuses through membranes (rapid onset, short duration after inhalation is stopped) ...
... • Blended mixture of 50% nitrous oxide and 50% oxygen • Also known as “laughing gas” • Produces sedation and analgesia • Colorless, odorless, heavier than air • Nonexplosive, nonflammable • Readily diffuses through membranes (rapid onset, short duration after inhalation is stopped) ...
Nitrous Oxide Guidlines
... • Blended mixture of 50% nitrous oxide and 50% oxygen • Also known as “laughing gas” • Produces sedation and analgesia • Colorless, odorless, heavier than air • Nonexplosive, nonflammable • Readily diffuses through membranes (rapid onset, short duration after inhalation is stopped) ...
... • Blended mixture of 50% nitrous oxide and 50% oxygen • Also known as “laughing gas” • Produces sedation and analgesia • Colorless, odorless, heavier than air • Nonexplosive, nonflammable • Readily diffuses through membranes (rapid onset, short duration after inhalation is stopped) ...
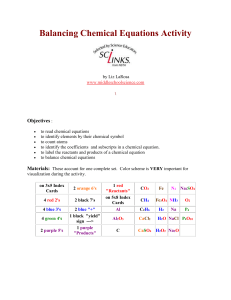
Balancing Chemical Equations Activity by Liz LaRosa www
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
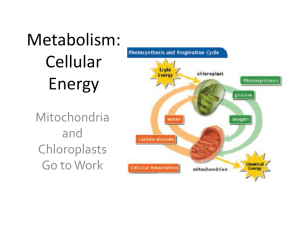
Cellular Energy
... Photosynthesis • Plants make enough glucose to be used during the night and on cloudy days when they don’t get sunlight. • The extra glucose is stored in the cells of the plant’s leaves. • When needed, the glucose travels to the mitochondria to be used in cellular respiration for the production of ...
... Photosynthesis • Plants make enough glucose to be used during the night and on cloudy days when they don’t get sunlight. • The extra glucose is stored in the cells of the plant’s leaves. • When needed, the glucose travels to the mitochondria to be used in cellular respiration for the production of ...
Word and Skeleton Equations Practice (ws Fall 2010)
... hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and iodine, I2(s). 1. Gaseous hydrogen and oxygen react to form gaseous water. 2. Solid sodium metal reacts with liquid water to form an aqueous solution of sodium hydroxide and hydrogen gas. 3. Solid ...
... hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and iodine, I2(s). 1. Gaseous hydrogen and oxygen react to form gaseous water. 2. Solid sodium metal reacts with liquid water to form an aqueous solution of sodium hydroxide and hydrogen gas. 3. Solid ...
Oxygen - CriticalCareMedicine
... interferes with regional blood flow and promotes shunting from vulnerable microcirculatory units. Inhomogeneous expression of endothelial adhesion molecules also contribute through their effects on WBC kinetics. ...
... interferes with regional blood flow and promotes shunting from vulnerable microcirculatory units. Inhomogeneous expression of endothelial adhesion molecules also contribute through their effects on WBC kinetics. ...
TOX - UCSF Fresno
... dissociate into oxygen bubbles in a reaction enhanced by the enzyme catalase: 2 [H2O2] > 2[H2O] + O2 (gas). A lot of oxygen can be liberated – it estimated that just 1 mouthful (30 cc) of concentrated hydrogen peroxide releases 3.5 LITERS of oxygen gas. The real problem with all this oxygen is that ...
... dissociate into oxygen bubbles in a reaction enhanced by the enzyme catalase: 2 [H2O2] > 2[H2O] + O2 (gas). A lot of oxygen can be liberated – it estimated that just 1 mouthful (30 cc) of concentrated hydrogen peroxide releases 3.5 LITERS of oxygen gas. The real problem with all this oxygen is that ...
Writing Chemical Equations KClO3 O2 (g) + KCl (s) Balancing
... Would read: Solid sodium hydrogen carbonate reacts with aqueous hydrochloric acid to produce aqueous sodium chloride, liquid water and gaseous carbon dioxide. Example2: Oxygen gas can be made by heating potassium chlorate in the presence of the catalyst manganese(IV) oxide. Potassium chloride is lef ...
... Would read: Solid sodium hydrogen carbonate reacts with aqueous hydrochloric acid to produce aqueous sodium chloride, liquid water and gaseous carbon dioxide. Example2: Oxygen gas can be made by heating potassium chlorate in the presence of the catalyst manganese(IV) oxide. Potassium chloride is lef ...
Anaerobic Energy Systems
... Post Exercise Oxygen Consumption (EPOC) When aerobic exercise begins, the oxygen transport system does not immediately supply the needed quantity of oxygen to the active muscles because oxygen consumption requires several minutes to reach steady state. Because oxygen needs and oxygen supply dif ...
... Post Exercise Oxygen Consumption (EPOC) When aerobic exercise begins, the oxygen transport system does not immediately supply the needed quantity of oxygen to the active muscles because oxygen consumption requires several minutes to reach steady state. Because oxygen needs and oxygen supply dif ...
MS Word Version
... The tertiary structure of myoglobin is that of a typical water soluble globular protein. Its secondary structure is unusual in that it contains a very high proportion (75%) of a-helical secondary structure. A myoglobin polypeptide is comprised of 8 separate right handed ahelices, designated A throug ...
... The tertiary structure of myoglobin is that of a typical water soluble globular protein. Its secondary structure is unusual in that it contains a very high proportion (75%) of a-helical secondary structure. A myoglobin polypeptide is comprised of 8 separate right handed ahelices, designated A throug ...
Chemistry PowerPoint
... a. The total mass of the reactants is greater than the total mass of the products b. The total mass of the reactants is less than the total mass of the products c. The total mass of the reactants equals the total mass of the products d. Mass can be created and destroyed ...
... a. The total mass of the reactants is greater than the total mass of the products b. The total mass of the reactants is less than the total mass of the products c. The total mass of the reactants equals the total mass of the products d. Mass can be created and destroyed ...
Carbon Cycle
... 2. Animals and plants cannot directly use all the nitrogen found in our ________________. 3. Only special bacteria can directly use nitrogen in our atmosphere and “fix” it so other organisms can benefit. These bacteria are called ____________-_________ bacteria. 4. Higher organisms use nitrogen to m ...
... 2. Animals and plants cannot directly use all the nitrogen found in our ________________. 3. Only special bacteria can directly use nitrogen in our atmosphere and “fix” it so other organisms can benefit. These bacteria are called ____________-_________ bacteria. 4. Higher organisms use nitrogen to m ...
Group 16: The Oxygen Family - Chemwiki
... The latter allotrope, ozone, is a pale-blue poisonous gas with a strong odor. It is a very good oxidizing agent, stronger than dioxygen, and can be used as a substitute for chlorine in purifying drinking water without giving the water an odd taste. However, because of its unstable nature it disappea ...
... The latter allotrope, ozone, is a pale-blue poisonous gas with a strong odor. It is a very good oxidizing agent, stronger than dioxygen, and can be used as a substitute for chlorine in purifying drinking water without giving the water an odd taste. However, because of its unstable nature it disappea ...
Acetaldehyde2
... include this sour product. • Ethanal is a colorless liquid that has a stong “green apple” smell and without it we might not have this flavor for our candy, gum, etc. ...
... include this sour product. • Ethanal is a colorless liquid that has a stong “green apple” smell and without it we might not have this flavor for our candy, gum, etc. ...
Chapter 1 Homework - due Tuesday, Sept
... a) electron transport chain - these protein complexes pump protons into the intermembrane space while passing electrons between them b) proton gradient - so that hydrogen ions will diffuse through the ATP synthase channels down their concentration gradient c) c) ATP synthase complex – as hydrogen io ...
... a) electron transport chain - these protein complexes pump protons into the intermembrane space while passing electrons between them b) proton gradient - so that hydrogen ions will diffuse through the ATP synthase channels down their concentration gradient c) c) ATP synthase complex – as hydrogen io ...
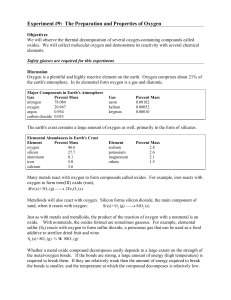
Experiment # 9 Properties of Oxygen
... Experiment #9: The Preparation and Properties of Oxygen Objectives We will observe the thermal decomposition of several oxygen-containing compounds called oxides. We will collect molecular oxygen and demonstrate its reactivity with several chemical elements. Safety glasses are required for this expe ...
... Experiment #9: The Preparation and Properties of Oxygen Objectives We will observe the thermal decomposition of several oxygen-containing compounds called oxides. We will collect molecular oxygen and demonstrate its reactivity with several chemical elements. Safety glasses are required for this expe ...
IntroRedoxDCIAns
... combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or appearance of an element. The other characteristic is the change in the number of ...
... combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or appearance of an element. The other characteristic is the change in the number of ...
Introduction to Oxidation Reduction
... combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or appearance of an element. The other characteristic is the change in the number of ...
... combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or appearance of an element. The other characteristic is the change in the number of ...
Exercise and Respiration Paloma
... • Myoglobin: a respiratory pigment found in some muscles. It acts as an oxygen store by combining with O2 in areas of high concentration and breaks down when the oxygen levels are low. • used for oxygen storage, for use during exercise ...
... • Myoglobin: a respiratory pigment found in some muscles. It acts as an oxygen store by combining with O2 in areas of high concentration and breaks down when the oxygen levels are low. • used for oxygen storage, for use during exercise ...
Oxidation Number Rules
... Chemistry 1211 Rules for Establishing Oxidation Numbers 1. Any uncombined atom, or any atom in a molecule of an element, is assigned an oxidation number of zero. Examples: N2, Cl2, C, Sn, S8 2. The oxidation number of a simple, monatomic ion is the same as the charge on the ion. Examples: Na+ is +1, ...
... Chemistry 1211 Rules for Establishing Oxidation Numbers 1. Any uncombined atom, or any atom in a molecule of an element, is assigned an oxidation number of zero. Examples: N2, Cl2, C, Sn, S8 2. The oxidation number of a simple, monatomic ion is the same as the charge on the ion. Examples: Na+ is +1, ...
Science of running
... • Increase stroke volume – amount of blood that can be pushed from the heart • Increase the amount of oxygen in the blood (Iron) • Increase the amount of capillaries that move the blood to the muscles ...
... • Increase stroke volume – amount of blood that can be pushed from the heart • Increase the amount of oxygen in the blood (Iron) • Increase the amount of capillaries that move the blood to the muscles ...
Oxygen
Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements. Photosynthesis releases oxygen, and respiration consumes oxygen. Changes in phosphate are related to changes in oxygen concentrations.Oxygen was discovered independently by Carl Wilhelm Scheele, in Uppsala, in 1773 or earlier, and Joseph Priestley in Wiltshire, in 1774, but Priestley is often given priority because his work was published first. The name oxygen was coined in 1777 by Antoine Lavoisier, whose experiments with oxygen helped to discredit the then-popular phlogiston theory of combustion and corrosion. Its name derives from the Greek roots ὀξύς oxys, ""acid"", literally ""sharp"", referring to the sour taste of acids and -γενής -genes, ""producer"", literally ""begetter"", because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition.