Midterm Review Project Ch 5
... phospholipids make cell membrance: hydrophilic head on outside and hydrophobic tail on inside of bilayer steroids, cholesterol ...
... phospholipids make cell membrance: hydrophilic head on outside and hydrophobic tail on inside of bilayer steroids, cholesterol ...
Protein folding
... As they are synthesized they assume secondary and tertiary structure. Activity of proteins depend on the integrity of its final tertiary structure also reffered as the native form. The native form of protein is not very stable structure and it vulnerable to change by heat, high salt, reducing agents ...
... As they are synthesized they assume secondary and tertiary structure. Activity of proteins depend on the integrity of its final tertiary structure also reffered as the native form. The native form of protein is not very stable structure and it vulnerable to change by heat, high salt, reducing agents ...
Amino Acids - Clydebank High School
... (substrate) molecule which have the correct shape and orientation. They form an intermediate complex. The new substance forms and then leaves the enzyme surface – which can then be reused. When an enzyme is denatured the shape of the protein molecules, of the enzyme, has changed and the reacta ...
... (substrate) molecule which have the correct shape and orientation. They form an intermediate complex. The new substance forms and then leaves the enzyme surface – which can then be reused. When an enzyme is denatured the shape of the protein molecules, of the enzyme, has changed and the reacta ...
Power Point
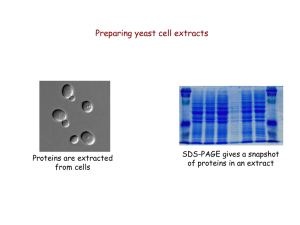
... “Quick and dirty” protein extraction from yeast 1. Collect cells by centrifugation 2. Wash cells with deionized water ...
... “Quick and dirty” protein extraction from yeast 1. Collect cells by centrifugation 2. Wash cells with deionized water ...
Cellular Chemical Reactions
... What are the four main types of large molecules and what characteristic do they share? What is an important property of lipids? What are some things that proteins do? What are the subunits of nucleic acids called? Use pg 537 to describe how a cell membrane is ...
... What are the four main types of large molecules and what characteristic do they share? What is an important property of lipids? What are some things that proteins do? What are the subunits of nucleic acids called? Use pg 537 to describe how a cell membrane is ...
File
... • These interactions may include hydrogen bonds, disulphide bridges, ionic interactions, polar associations, etc. • The affinity or repulsion of side chains will affect the overall shape of the polypeptide chain and are determined by the position of specific amino acids within a sequence. ...
... • These interactions may include hydrogen bonds, disulphide bridges, ionic interactions, polar associations, etc. • The affinity or repulsion of side chains will affect the overall shape of the polypeptide chain and are determined by the position of specific amino acids within a sequence. ...
IFITM3 Peptide PRODUCT DATA SHEET Bioworld Technology CO., Ltd.
... family of proteins. IFITM proteins are induced by type I and type II interferons and contain multiple interferon (IFN)-stimulated response elements (ISREs) in their promoter regions. IFITM proteins play important roles in many cellular processes and their expression requires the presence of the ...
... family of proteins. IFITM proteins are induced by type I and type II interferons and contain multiple interferon (IFN)-stimulated response elements (ISREs) in their promoter regions. IFITM proteins play important roles in many cellular processes and their expression requires the presence of the ...
protein Synthesis
... 3. DNA has the information to make what? 4. What do proteins do? What do enzymes do? 5. Where is DNA located? What are genes? 6. What units are proteins made from? Where are proteins synthesized? 7. How does the DNA information get from nucleus to cytoplasm? 8. What is transcription? What is transla ...
... 3. DNA has the information to make what? 4. What do proteins do? What do enzymes do? 5. Where is DNA located? What are genes? 6. What units are proteins made from? Where are proteins synthesized? 7. How does the DNA information get from nucleus to cytoplasm? 8. What is transcription? What is transla ...
Proteins for Growth and Repair
... Too much protein can harm your body. If you eat too much protein the excess will be stored as body fat. Over-consumption of protein can also cause constipation, diarrhea, excessive gas, dehydration, dizziness and bad breath. ...
... Too much protein can harm your body. If you eat too much protein the excess will be stored as body fat. Over-consumption of protein can also cause constipation, diarrhea, excessive gas, dehydration, dizziness and bad breath. ...
Übung: Monte Carlo, Molecular Dynamics
... dependent dielectric constant. Write it simply as a function of the distance between two particles (no need for vectors). Use nomenclature consistent with the question above. ...
... dependent dielectric constant. Write it simply as a function of the distance between two particles (no need for vectors). Use nomenclature consistent with the question above. ...
Overview
... Micro 201 Yuan Lecture 2, Class 24: Protein Folding and Molecular Chaperones April 20th, 2017 Overview The intracellular concentration of protein in bacterial cells can be estimated to be ~135 mg/ml. In this session, we will explore how bacteria employ a suite of molecular machines collectively know ...
... Micro 201 Yuan Lecture 2, Class 24: Protein Folding and Molecular Chaperones April 20th, 2017 Overview The intracellular concentration of protein in bacterial cells can be estimated to be ~135 mg/ml. In this session, we will explore how bacteria employ a suite of molecular machines collectively know ...
3-in-1: A novel approach to study membrane protein pharmacology
... mediates crucial functions in the nervous system and has been implicated a numerous diseases. Most LGICs are molecular assemblies of more than one subunit, but conventional methods to study these proteins cannot easily address the contribution of individual subunits within such a protein complex. Re ...
... mediates crucial functions in the nervous system and has been implicated a numerous diseases. Most LGICs are molecular assemblies of more than one subunit, but conventional methods to study these proteins cannot easily address the contribution of individual subunits within such a protein complex. Re ...
Ch. 5. Protein Purification and Characterization Techniques
... Salting Out • After Proteins solubilized, they can be purified based on solubility (usually dependent on overall charge, ionic strength, polarity • Ammonium sulfate (NH4SO4) commonly used to “salt out” ...
... Salting Out • After Proteins solubilized, they can be purified based on solubility (usually dependent on overall charge, ionic strength, polarity • Ammonium sulfate (NH4SO4) commonly used to “salt out” ...
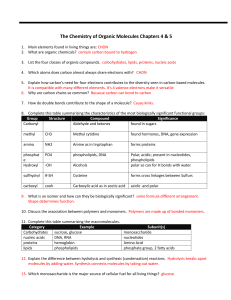
04-05 Biochem review sheet answers ws
... 17. Explain the difference between a saturated and unsaturated fatty acid. Unsaturated have some double carbon bonds. 18. List some of the many functions of proteins. transport, hormones, receptors, defense, etc. 19. How do amino acids differ? R group 20. What is a peptide bond? covalent bond betwee ...
... 17. Explain the difference between a saturated and unsaturated fatty acid. Unsaturated have some double carbon bonds. 18. List some of the many functions of proteins. transport, hormones, receptors, defense, etc. 19. How do amino acids differ? R group 20. What is a peptide bond? covalent bond betwee ...
1. Protein Interactions
... Size: Larger molecules have more active sites Structure: the stability (strength of intramolecular bonds) and molecule unfolding rate ...
... Size: Larger molecules have more active sites Structure: the stability (strength of intramolecular bonds) and molecule unfolding rate ...
Chemical Compounds Overview
... d. Dehydration synthesis- Reaction in which water in removed to form a bond, creating a polymer. e. Hydrolysis- Reverse of dehydration synthesis. Polymers are broken down into monomers by adding water. 2. Lipids a. Store energy. Form some membranes. b. Monomer- Glycerol + 3 fatty acids c. Polymer- f ...
... d. Dehydration synthesis- Reaction in which water in removed to form a bond, creating a polymer. e. Hydrolysis- Reverse of dehydration synthesis. Polymers are broken down into monomers by adding water. 2. Lipids a. Store energy. Form some membranes. b. Monomer- Glycerol + 3 fatty acids c. Polymer- f ...
Protein Engineering
... Usually found in extracellular proteins, not intracellular Cross link between chains or in chains formed by oxidation of cysteine residues ...
... Usually found in extracellular proteins, not intracellular Cross link between chains or in chains formed by oxidation of cysteine residues ...
In Biology, Molecular Shape Matters
... Its not just chemical formula, it’s the shape of the molecule that lets it do its “job”. ...
... Its not just chemical formula, it’s the shape of the molecule that lets it do its “job”. ...
Self Test Quiz-1 Given below are some questions related to protein
... Given below are some questions related to protein and enzymes in general. Each statement is followed by 4 choices. Choose a single correct answer for each question. 1. How many different types of amino acid are used to make proteins? a. 4 b. 20 c. 23 d. 38 2. Amino acids contain carbon, hydrogen, ox ...
... Given below are some questions related to protein and enzymes in general. Each statement is followed by 4 choices. Choose a single correct answer for each question. 1. How many different types of amino acid are used to make proteins? a. 4 b. 20 c. 23 d. 38 2. Amino acids contain carbon, hydrogen, ox ...
Proteins - mrsmaineswiki
... tripeptide. Follow the same basic instructions as you did to make the dipeptide. Complete an equation on your drawing paper. 8. Explain how 2 different proteins could be made from the same 3 amino acids. 9. Proteins usually consist of a long chain of amino acids. The chain is ____________, _________ ...
... tripeptide. Follow the same basic instructions as you did to make the dipeptide. Complete an equation on your drawing paper. 8. Explain how 2 different proteins could be made from the same 3 amino acids. 9. Proteins usually consist of a long chain of amino acids. The chain is ____________, _________ ...
Cyclol

The cyclol hypothesis is the first structural model of a folded, globular protein. It was developed by Dorothy Wrinch in the late 1930s, and was based on three assumptions. Firstly, the hypothesis assumes that two peptide groups can be crosslinked by a cyclol reaction (Figure 1); these crosslinks are covalent analogs of non-covalent hydrogen bonds between peptide groups. These reactions have been observed in the ergopeptides and other compounds. Secondly, it assumes that, under some conditions, amino acids will naturally make the maximum possible number of cyclol crosslinks, resulting in cyclol molecules (Figure 2) and cyclol fabrics (Figure 3). These cyclol molecules and fabrics have never been observed. Finally, the hypothesis assumes that globular proteins have a tertiary structure corresponding to Platonic solids and semiregular polyhedra formed of cyclol fabrics with no free edges. Such ""closed cyclol"" molecules have not been observed either.Although later data demonstrated that this original model for the structure of globular proteins needed to be amended, several elements of the cyclol model were verified, such as the cyclol reaction itself and the hypothesis that hydrophobic interactions are chiefly responsible for protein folding. The cyclol hypothesis stimulated many scientists to research questions in protein structure and chemistry, and was a precursor of the more accurate models hypothesized for the DNA double helix and protein secondary structure. The proposal and testing of the cyclol model also provides an excellent illustration of empirical falsifiability acting as part of the scientific method.