Document
... 5B. Suppose the purified PR is subjected to separation procedures with the following results: 1) Separation of the untreated protein by sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) results in 3 polypeptide bands of apparent MWs 180, 120 and 50. 2) Pretreatment of the protein w ...
... 5B. Suppose the purified PR is subjected to separation procedures with the following results: 1) Separation of the untreated protein by sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) results in 3 polypeptide bands of apparent MWs 180, 120 and 50. 2) Pretreatment of the protein w ...
2009 exam with answers
... Cellulose, as apposing strands are held together by H-bonds of the protruding hydroxyl at carbon 6. Such H-bonds are not possible for polyfucose as there is no hydroxyl on carbon 6. If you did not remember the protruding C6 hydroxyl’s role in particular, the same conclusion could be reached based on ...
... Cellulose, as apposing strands are held together by H-bonds of the protruding hydroxyl at carbon 6. Such H-bonds are not possible for polyfucose as there is no hydroxyl on carbon 6. If you did not remember the protruding C6 hydroxyl’s role in particular, the same conclusion could be reached based on ...
Functional Groups List
... Can form hydrogen bonds with other groups to weak polarity. Impt in making hydrogen bonds nearby chains in proteins. Found in sugars like fructose. ...
... Can form hydrogen bonds with other groups to weak polarity. Impt in making hydrogen bonds nearby chains in proteins. Found in sugars like fructose. ...
lecture 5
... - the cytoplasm has a reducing environment where disulfide bonds do not occur • disulfide-bonded proteins occur in the endoplasmic reticulum in eukaryotes; secreted proteins • periplasmic space in prokaryotes ...
... - the cytoplasm has a reducing environment where disulfide bonds do not occur • disulfide-bonded proteins occur in the endoplasmic reticulum in eukaryotes; secreted proteins • periplasmic space in prokaryotes ...
Practice Problems
... 12B. The ‘front’ end of a protein is better known as the ________________. 12C. An -helix (alpha-helix) is an example of a type of _____________ structure. 13. Peptide bonds are found in A. phospholipids. B. cholesterol. C. proteins. D. DNA. E. pyrocatechol. 14. Proteins can be polymerized A. on th ...
... 12B. The ‘front’ end of a protein is better known as the ________________. 12C. An -helix (alpha-helix) is an example of a type of _____________ structure. 13. Peptide bonds are found in A. phospholipids. B. cholesterol. C. proteins. D. DNA. E. pyrocatechol. 14. Proteins can be polymerized A. on th ...
Principles of Life
... After the tertiary structures of proteins were first shown to be highly specific, the question arose as to how the order of amino acids determined the three-dimensional structure. The second protein whose structure was determined was ribonuclease A, an enzyme from cows that was readily available fro ...
... After the tertiary structures of proteins were first shown to be highly specific, the question arose as to how the order of amino acids determined the three-dimensional structure. The second protein whose structure was determined was ribonuclease A, an enzyme from cows that was readily available fro ...
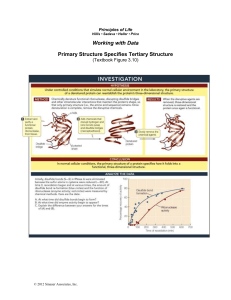
Primary Structure Specifies Tertiary Structure
... determined was ribonuclease A, an enzyme from cows that was readily available from pancreases at slaughterhouses. Because it works in the highly acidic environment of the cow stomach, RNase A was stable compared to most proteins and easy to purify. RNase A has 124 amino acids, among which are eight ...
... determined was ribonuclease A, an enzyme from cows that was readily available from pancreases at slaughterhouses. Because it works in the highly acidic environment of the cow stomach, RNase A was stable compared to most proteins and easy to purify. RNase A has 124 amino acids, among which are eight ...
Working with Data Primary Structure Specifies Tertiary Structure
... After the tertiary structures of proteins were first shown to be highly specific, the question arose as to how the order of amino acids determined the three-dimensional structure. The second protein whose structure was determined was ribonuclease A, an enzyme from cows that was readily available fro ...
... After the tertiary structures of proteins were first shown to be highly specific, the question arose as to how the order of amino acids determined the three-dimensional structure. The second protein whose structure was determined was ribonuclease A, an enzyme from cows that was readily available fro ...
L9 Protein cross links - e
... Application of sulfhydryl oxidase in food industry Production of dairy products The SOX was proved to remove the ‘burnt-off ’ flavor of milk caused by ultrahigh-temperature treatment (UHT); tested in a pilot scale with the enzyme immobilized on glass beads. This effect is suggested to be based on th ...
... Application of sulfhydryl oxidase in food industry Production of dairy products The SOX was proved to remove the ‘burnt-off ’ flavor of milk caused by ultrahigh-temperature treatment (UHT); tested in a pilot scale with the enzyme immobilized on glass beads. This effect is suggested to be based on th ...
here - Dr. Alan Dombkowski
... are the estimated χ3 torsion angle and an energy value in kcal/mol. χ3 angles of known disulfides have a bimodal distribution with peaks observed at +100 and -80 (Petersen et al., 1999), see figure in technical details. These values provide useful selection criteria for disulfide design. The estimat ...
... are the estimated χ3 torsion angle and an energy value in kcal/mol. χ3 angles of known disulfides have a bimodal distribution with peaks observed at +100 and -80 (Petersen et al., 1999), see figure in technical details. These values provide useful selection criteria for disulfide design. The estimat ...
week 2 montal answers
... a. Need Donor + Acceptor 2.) Ionic interactions a. Attraction = between opposite charges b. Repulsion = same charges. 3.) Hydrophobic interactions. 4.) Van der Waals interactions = two atoms in close proximity. Individually these forces are much weak, but many hundreds can add up to be as strong as ...
... a. Need Donor + Acceptor 2.) Ionic interactions a. Attraction = between opposite charges b. Repulsion = same charges. 3.) Hydrophobic interactions. 4.) Van der Waals interactions = two atoms in close proximity. Individually these forces are much weak, but many hundreds can add up to be as strong as ...
Determination of Amino Acid Sequence
... Sequencing of peptides Ordering peptide fragments Compare sequences generated from different cleavage methods Locating disulfide bonds Comparison of cleavage fragment with or without breaking disulfide bonds ...
... Sequencing of peptides Ordering peptide fragments Compare sequences generated from different cleavage methods Locating disulfide bonds Comparison of cleavage fragment with or without breaking disulfide bonds ...
Week 2 Handout with No answers
... a. Attraction = between opposite charges b. Repulsion = same charges. 3.) ___________________ 4.) __________________ = two atoms in close proximity. Individually these forces are much weak, but many hundreds can add up to be as strong as covalent bond. o Electrostatic + Hydrogen bonds are on the ord ...
... a. Attraction = between opposite charges b. Repulsion = same charges. 3.) ___________________ 4.) __________________ = two atoms in close proximity. Individually these forces are much weak, but many hundreds can add up to be as strong as covalent bond. o Electrostatic + Hydrogen bonds are on the ord ...
Serine Proteases Teaching Exercises
... c. Find phenylalanine, tyrosine, tryptophan and methionine. 4. When these enzymes are made, they are initially in an inactive precursor state, called zymogens. In order for the enzymes to become active, they will be cleaved to arrange the linear amino acid sequence in such a fashion as to orient thr ...
... c. Find phenylalanine, tyrosine, tryptophan and methionine. 4. When these enzymes are made, they are initially in an inactive precursor state, called zymogens. In order for the enzymes to become active, they will be cleaved to arrange the linear amino acid sequence in such a fashion as to orient thr ...
$doc.title
... proteins can assist proper protein folding as a protein matures, but once protein aggregates form generally they cannot be renatured to their individual, correct structure. Disulfide bond forming (DBF) enzyme is a chaperone protein related to the class of Sso7d proteins from the hyperthermophilic ba ...
... proteins can assist proper protein folding as a protein matures, but once protein aggregates form generally they cannot be renatured to their individual, correct structure. Disulfide bond forming (DBF) enzyme is a chaperone protein related to the class of Sso7d proteins from the hyperthermophilic ba ...
HWTP- test practice
... 6. The combinatorics of disulfide bond formation. A protein may contain several cysteines, which may pair together to form disulfide bonds as shown in Figure 1.17. If there is an even number n of cysteines, n/2 disulfide bonds can form. How many different disulfide pairing arrangements are possible ...
... 6. The combinatorics of disulfide bond formation. A protein may contain several cysteines, which may pair together to form disulfide bonds as shown in Figure 1.17. If there is an even number n of cysteines, n/2 disulfide bonds can form. How many different disulfide pairing arrangements are possible ...
DISULFIDE GROUPS Disulfide bonds in proteins are
... requires the presence of urea or another denaturant; limited and selective cleavage of a few disulfide bonds can frequently be obtained in the absence of such agents. Procedures have been described for limited reduction of disulfide bonds in proteins using dithiothreitol (Bewley and Li, 1969; Liu an ...
... requires the presence of urea or another denaturant; limited and selective cleavage of a few disulfide bonds can frequently be obtained in the absence of such agents. Procedures have been described for limited reduction of disulfide bonds in proteins using dithiothreitol (Bewley and Li, 1969; Liu an ...
CH 107 SI Summer 2015 Worksheet 13 Answers What are the two
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
Disulfide bridge assignment in complex proteins - HES
... Description Assigning disulfide bridges is an important component of the analytical strategy during recombinant protein production, for which mass spectrometry (MS) is an important technique. Venom proteins, such as the threefinger toxins, pose a particular challenge due to their complex arrangement ...
... Description Assigning disulfide bridges is an important component of the analytical strategy during recombinant protein production, for which mass spectrometry (MS) is an important technique. Venom proteins, such as the threefinger toxins, pose a particular challenge due to their complex arrangement ...
MCB100A/CHEM130A In-Section Quiz #2 (Aathavan Karunakaran)
... 2. Consider Ribonuclease-A from Anfinsen’s original experiment. You have mutated one of the cysteines into a serine (there are now only 7 cysteines), but the mutant protein is just as active as the wild-type. You now repeat the Anfinsen experiment on this mutant protein: i.e, unfolded the protein w ...
... 2. Consider Ribonuclease-A from Anfinsen’s original experiment. You have mutated one of the cysteines into a serine (there are now only 7 cysteines), but the mutant protein is just as active as the wild-type. You now repeat the Anfinsen experiment on this mutant protein: i.e, unfolded the protein w ...
第五屆生物物理新知研討會
... Department of Biological Science & Technology,Institute of Bioinformatics, National Chiao Tung University, HsinChu, Taiwan ...
... Department of Biological Science & Technology,Institute of Bioinformatics, National Chiao Tung University, HsinChu, Taiwan ...
Disulfide

In chemistry and biology a disulfide refers to a functional group with the general structure R–S–S–R. The linkage is also called an SS-bond or a disulfide bridge and is usually derived by the coupling of two thiol groups. In formal terms, the connection is a persulfide, in analogy to its congener, peroxide (R–O–O–R), but this terminology is rarely used, except in reference to hydrodisulfides (R–S–S–H or H–S–S–H compounds).In inorganic chemistry disulfide usually refers to the corresponding anion S22−, or −S–S−, for example in disulfur dichloride.