* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Duncan Watts, why are epidemics so unpredictable
Survey
Document related concepts
Hospital-acquired infection wikipedia , lookup
Epidemiology of HIV/AIDS wikipedia , lookup
Whooping cough wikipedia , lookup
History of biological warfare wikipedia , lookup
Oesophagostomum wikipedia , lookup
Great Plague of London wikipedia , lookup
Transcript
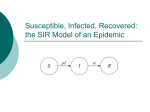
Duncan Watts, why are epidemics so unpredictable? Standard Model: S-I-R model: susceptible, infected, removed Mixing is uniformly random—mass action assumption Parameters of susceptibility, loss of immunity, recovery (HIV, 0) or death Sometimes extra category: “exposed” but not yet infectious; group to target—multiple exposures have additive effects on infection Mass action: epidemics depend only on total fraction of infectives and susceptibles Basic reproduction number, Ro, is average number of new infectives generated by a single infective If Ro > 1, exponential growth => epidemic threshold – bimodal outcome space, either epidemic or doesn’t catch on. If Ro < 1, epidemics never occur Shape of epidemic curve; number of new cases per day: slow growth, explosive Phases, followed by burnout (few remaining susceptibles to infect) Classical sigmoid curve 1918-9 Spanish flu, 500K deaths in US 1957-8 Asian flu, 70K deaths in Us 1968-9 Hong Kong flu, 34K deaths 2003 SARS, 800 deaths world wide All have about same Ro Why so different in size? 100 year history of Measles and Pertussis in Iceland: Log – log plot should be a straight line if a power law (isn’t) Over time a fluctuating incidence occurs: “resurgent” again and again; factors of immunity-recovery increase variability but don’t change this pattern; invading new populations gives rise to peaks in incidence, or “outbreaks.” Populations exhibit structure: Inhomogeneous population distribution Transportation and infrastructure networks Social, organizational and sexual networks => Uniform mixing only in local populations => Large networks are concatenations of many small epidemics Plague (Bubonic) unusual; fleas spread by humans and rats; spread at “walking rate,” 2-3 Km/day across Europe How to incorporate networks of human to human transmission into epidemic models? Compromise: Mass action holds locally; Local contexts are embedded in hierarchy of contexts (neighborhoods; cities; counties; states; regions; countries, continents); infection occurs (at least largely) in local contexts. Metapopulation models (global vs. local) not new but this has multiple embedded levels. (technically trivial but consequential nonetheless) Disease vs information spread network models: requires direct contact with all infected individuals. Multiscale populations generate variety of distributions for the same Ro; similar distributions for very different Ros Conclusions Ro>1 is still a necessary condition for an epidemic – but alone in a multiscale model it tells us very little about size or duration. Need also non-local mobility Po>1 These 2 conditions are sufficient for a non-local epidemic Reducing mobility and transport range (zeta) can be extremely effective in stopping an epidemic (e.g., WHO recommends against travel to China) Avg. size of epidemic more sensitive to Zeta than to volume of transport Po A very small number of people determine the size of an epidemic (e.g., rare events, by getting on a plane); 3 people doubled the size of one epidemic.