* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 52142_present
Cluster chemistry wikipedia , lookup
Hydroformylation wikipedia , lookup
Metalloprotein wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Metal carbonyl wikipedia , lookup
Spin crossover wikipedia , lookup
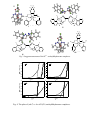
1 PtIV-oxaloyldihydrazone complexes: synthesis, characterization, optical properties and corrosion inhibition on carbon-steel in hydrochloric acid solution A.M. Hassana, Ayman H. Ahmeda*, Hosni A. Gumaaa, Bassem H. Mohamedb, Ahmed M. Erakyb aDepartment of Chemistry, Faculty of science, Al-Azhar University. Nasr City, Cairo, Egypt. bCairo E-mail: Oil refining Company (CORC), Cairo, Egypt. * Author to whom correspondence should be addressed; e-mail address: [email protected] Tel: +2022629357 Fax: +2022629358 Abstract Platinium(IV) hydrazone complexes derived from oxaloyldihydrazide and selected aldehydes, namely hydroxyacetophenone salicyaldehyde and and o-methoxybenzaldehyde 2-hydroxynaphthaldehyde, o- have been synthesized and characterized. The mode of bonding as well as the structure of the isolated compounds have been ascertained on the basis of emental analyses and spectral (UV-Vis., IR, mass, 1 H NMR) measurements. The synthesized dihydrazones coordinate to the metal center either dibasic or tetrabasic hexadentate ligands forming binuclear complexes in all cases. The metal center in all investigated complexes has octahedral stereochemistry. Ligands L1 and L3 bind with Pt(IV) ion in diketo forms while L2 and L4 employed their dienol skeletons. Optical transmission spectra were recorded in the range 190–2100 nm and optical parameters such as indirect optical band gap and refractive index have been determined. The inhibitive and adsorptive properties of all ligands for the corrosion of 2 carbon-steel in 1 M HCl solutions were investigated using weight loss technique. The results pointed out that the ligands have reasonable inhibitions towards the corrosion of carbon-steel. The adsorption of the inhibitors on metal surface was found to be spontaneous and consistent with the mechanism of physical adsorption. The adsorption data fitted well to Freundlich, Langmuir and Frumkin adsorption isotherms. Keyword: Dihydrazone complexes; Physico-chemical studies; Energy band gap; Corrosion inhibition. 1. Introduction Hydrazones played a central role in the development of coordination chemistry. Hydrazones obtained by the condensation of o-hydroxy or methoxy aldehydes and ketones with hydrazides are considered potential polynucleating ligands because they posses amide, azomethine and phenol or methoxy functions thus offer a variety of bonding possibilities in metal complexes [1]. Extensive studies have revealed that the lone pair on trigonally hybridized nitrogen atom of the azomethine group is responsible for the chemical and biological activity [2-4]. Several complexes derived from hydrazones have been reported [5-8] and compounds of this type have a great biological activity as antitumoral and antiviral agents [9,10]. In fact, little papers have been reported for oxaloyldihydrazones or their complexes due to their weak solubility in most organic solvents. Oxaloyldihydrazones are soluble only in high polar solvents as DMF and DMSO which required much effort to purify or isolate them. Nevertheless CoII, CuII, NiII, MnII, MoIV ..etc complexes of oxaloyldihydrazone have been studied [11-13], PtIV complexes of the selected oxaloyldihydrazones have not bee separated yet. Worthy mention, some hydrazone complexes have been encapsulated in zeolite-Y by analogous methods and the resulting materials were inferred by various physicochemical characterization techniques [14-18]. The corrosion problem is of a great problems, which faced the world from the last years until now, we can’t hide this problem from our live but we can reduce 3 “inhibit” it in the metals by several methods as the environment need. For this purpose, corrosion inhibitors are employed to interact with a metallic surface and thus protect the metal against corrosion specially in acidic environments [19]. Several reports have documented the use of many heterocyclic compounds, such as thiophene derivatives, mercaptobenzoazoles, crown ethers, Schiff base, hydrazones, pyridine derivatives and thio-compound [2,20,21] as corrosion inhibitors. The context of this work aims to describe some types of oxaloyldihydrazones and their Pt(IV) complexes. The study was also extended to investigate the adsorptive and inhibition efficiency for all ligands on carbon-steel (C-steel) in hydrochloric acid pickling solution and some thermodynamic parameters have been estimated. Moreover, the energy gap and refractive index of the isolated compounds have been determined to describe their optical and electronic properties. 2. Experimental 2.1 Materials and methods The selected metal salts, diethyl oxalate, hydrazine monohydrate and ammonium hexachloropalatinate (IV), (NH4)2[PtCl6], were purchased from Sigma-Aldrich. The employed aldehydes were of E-Merck grade. Other chemicals and solvents were of highest purity and used without further purification. The 1H NMR spectra were recorded on a Jeol-FX-90Q Fourier NMR spectrometer at 25 ºC using DMSO solvent and TMS as an internal standard. Mass spectra of the ligands were performed by a Shimadzu-GC-MSQP1000 EX using the direct inlet system. Metal contents (%wt) were estimated complexometrically by EDTA using xylenol orange as indicator and solid hexamine as buffer (pH = 6). Elemental analyses (CHN) and spectral (UV-Vis., FT-IR) measurements were carried out as reported [22]. The optical band gap energy (Eg) of product compounds was calculated from Tuac’s equations [23,24]. Carbon-steel (C-steel) specimen was of the chemical compositions (wt %) depicted in Table 1. Prior to the experiment, the specimens were polished with emery papers (220-800 grades) until the 4 surface appears free from any scratches and other apparent defects, then washed with distilled water followed by degreasing in absolute ethanol and acetone, dried at room temperature, weighed and finally stored in a moisture free desiccators prior to use. 2.2. Preparations 2.2.1 Preparation of oxalicdihydrazide Oxalicdihydrazide (H2NNHCO-CONHNH2) was isolated by the reaction of diethyloxalate (15 ml, 0.11 mole) and excess hydrazine monohydrate (9.8 ml, 0.22 mole) under continuous stirring in ice. The formed compound was filtered off, washed several times by water and ethanol, recrystallized from distilled water-ethanol mixed solvent including washing the solids repeatedly by ethanol to remove any excess of hydrazine. Finally, the isolated crystals were dried in oven at 80 °C for 8 hrs. The resulting product was melted at 240 °C (Lit. m.p= 240 °C), soluble in hot water and insoluble in alcohol. 2.2.2. Preparation of oxaloyldihydrazone ligands The dihydrazone ligands; bis(salicyaldehyde)oxaloyldihydrazone (L1), bis(2-hydroxy1-naphthaldhyde)oxaloyldihydrazone (L2), bis(2-hydroxyacetophenone) oxaloyldihydrazone (L3) and bis(2-methoxybenzaldhyde)oxaloyldihydrazone (L4); were prepared by general condensation procedure. Oxalicdihydrazide (0.01 mol) dissolved first in hot water (20 cm3) followed by adding methanol (40 cm3) was mixed with the appropriate aldehydes [salicyaldehyde, 2-hydroxy-1-naphthaldehyde, 2- hydroxyacetophenone and 2-methoxybenzaldehyde] (0.02 mol) in absolute methanol. The resulting mixture was refluxed for 3 hrs under constant stirring. The product separated out on concentrating the solution to half of its volume and cooling. The crystals of the desired ligand was collected by filtration through a Buchner funnel and dried in the oven at 50 °C for 2 h. After that the ligand was recrystallized from DMF-MeOHaq mixed solvent, collected, washed thoroughly on filter paper by acetone to remove any excess of DMF and then dried in an electric oven at 50 °C for 2 h. The authenticity of the ligands was proved by elemental analyses, (IR, mass and 1H NMR) spectroscopy (Table 2,3). Taking L1 and L2 as examples for mass fragmentation (Fig. 1), patterns of 5 bis(salicyaldehyde)oxaloyldihydrazone (L1) showed ion peaks at m/e = 326.45 (0.81%, C16H14N4O4) as molecular ion peak, 233.2 (0.39%, M+(C10H9N4O3)), 216.2 (0.39%, M+(C10H9N4O2)), 77.2 (100%, M+(C6H6) as base peak), 191.05 (0.27%, M+(C9H8N3O2)), 120.25 (72.6%, M+(C7H7N2)), 105.1 (33.79%, M+(C7H6N)), 163.25 (19.24%, M+(C8H7N2O2)), 135.2 (10%, M+(C7H7N2O)) and 66.05 (10.75%, M+(C5H6)). Mass spectrum of the bis(2-hydroxy-1-naphthaldhyde)oxaloyldihydrazone (L2) exhibited ion peaks at m/e = 427 (1.51%, (C16H14N4O4) as molecular ion peak, 410 (2.26%, M+(C24H18N4O3)), 270 (2.73%, M+(C13H10N4O3)), 157 (100%, M+(C11H9O) as base peak), 257 (4.34%, M+( C13H10N3O3)), 242 (2.02%, M+(C13H9N2O3)), m/e = 175 (56.99%, M+(C11H9NO)), 141 (6.39%, M+(C11H9)), 128 (19.76%, M+(C10H8), while the ion peak at m/e = 79 (11.31%) points to M+(C6H8), 64.95 (33.82%, M+(C5H5) and 50 (88.69%, M+(C4H4)). 2.2.3. Preparation of solid complexes The dihydrazone ligand (1 mmol) was dissolved in a minimum amount of DMF (20 ml) and then 60 ml methanol was added. The resulting solution was added slowly to a methanolic solution of Pt(IV) (ammonium hexachloropalatinate (IV)). The platinum salt was dissolved first in a minimum amount of distilled water (20 ml) before adding the methanol (40 ml) to make its solution. The resulting mixture was heated under reflux for 3-4 hrs and then reduced to 15 cm3 by evaporation on hot plate. The resulting reaction mixture was cooled down to room temperature and the colored solid complexes were filtered off, washed several times with successive portions of hot solvents (dimethylformamide, methanol and acetone, respectively) to remove any excess of the unreacted dihydrazone ligand and finally dried in an electric furnace at 80 °C for 3 hrs. 2.3. Corrosion inhibition 2.3.1 Weight loss measurement 6 In the weight loss experiment, 100 ml beakers containing 1M HCl solutions were employed at room temperature (28 °C). Stock solutions of the inhibitors were prepared in DMF. All test solutions contained 10 ml (20 vol.%) of DMF to maintain inhibitors completely soluble. The total volume of the test solution is 50 ml. The coupons were suspended in the beakers with the aid of glass hooks. In the absence of inhibitors, the coupons were retrieved from their corrodent solutions at 60 minutes interval for 240 minutes. Further measurements were carried out after introducing the additives (L1-L4) of concentrations 5×10-4, 1×10-4, 5×10-5 and 1×10-5 M (at room temperature) for the same duration. Coupons were dipped into saturated ammonium acetate solution at room temperature, to terminate the corrosion reaction. They were washed by scrubbing with a light brush, dried in acetone and then in an electric oven maintained at 80 °C. The loss of weight of coupons is evaluated in grams as the difference in weight of the coupons before and after the test [25], W =Wi - Wf , where: W = weight loss of coupon, Wi = Initial weight of coupon, Wf = Final weight of coupon. The value of corrosion rate, ρ (in gm.cm-2.min-1) was calculated from ρ = W/(A×t) equation [26], where: W = weight loss to the nearest 0.0001gm, A = area of specimen to the nearest 0.01 cm2 (2 × area of one surface/face) and t = exposure time (minutes). The percentage inhibition efficiency (%IE), and a parameter surface coverage (θ) which represents the part of the surface covered by the inhibitor molecules, were calculated using % = [1- ρ1/ρ2] X 100% and θ = [1- ρ1/ρ2] the equations [26], where: % = corrosion inhibition efficiency, ρ1 = corrosion rate in the presence of inhibitors, ρ2 = corrosion rate in the absence of inhibitor at the same temperature and θ = surface coverage. 3. Results and discussion The entire oxaloyldihydrazones (L1-L4) and their solid complexes with Pt(IV) have been isolated in a pure form. Analytical, physical and spectroscopic data of the hydrazones and their complexes are given in Tables 2,3. The complexes are insoluble in alcohols, Et2O, CHCl3, acetone, CCl4 as well as benzene meanwhile are nearly insoluble in DMF and DMSO. The insolubility feature of the isolated complexes discouraged the 7 execution of their 1H NMR or X-ray analysis. Comparison of the elemental analysis for both the calculated and found percentages indicates that the compositions of the isolated solid complexes coincide well with the proposed formulae. 3.1. IR Spectra The significant IR bands of dihydrazones and their platinum complexes are summarized in Table 2. Ligands: Oxloyldihydrazones (L1-L4) can exist either in the trans (staggered) configuration or cis-configuration, Fig. 2, [27]. In cis- configuration, the dihydrazone can adopt either anti-cis- configuration or syn-cis-configuration. NMR spectroscopy is considered a powerful technique employed to determine their skeleton. Infrared spectra showed strong bands at 1602-1617 cm-1 assignable to the azomethine group (νC=N). The observation of these bands emphasized the interaction of dihydrazides with aldehydes forming azomethine linkages. The bands of ν(OH) phenolic/naphthoic, ν(NH) and ν(C=O) for L1-L3 were noticed at (3149, 3204 and 1666), (3476, 3166 and 1705(m) +1660(v.s)) and (3448, 3293 and 1689(m) +1651(v.s)) cm-1, respectively. Meanwhile, L4 which does not contain o-hydroxy group revealed ν(NH) at 3202 cm-1 and ν(C=O) at1653 cm-1. The appearance of both ν(C=O) and ν(NH) simultaneously in the IR spectra of L1-L4 indicates the presence of keto forms. In fact, IR spectroscopy contributed with NMR technique in identifying the ligands skeleton. The appearance of carbonyl groups (C=O) near 1660 cm-1 as one band in case of L1 and L4 and pair of bands near (1660, 1700 cm-1) for each L2 and L3 indicates the trans (staggered-structure, Fig. 2) configuration for (L1 and L4) while mixture of [cis(syn/anti-cis-structure) + trans (staggered-structure), Fig. 2] isomers for (L2 and L3). This suggestion results from the field effect criteria which can be elucidated as follows. When 2C=O:: groups are in the same direction (cis configuration), the non-bonding electrons present on oxygen atoms cause electrostatic repulsion. This cause a change in the state of hybridization of C=O group and also make it to go out of the plane of the double bond. Thus, the configuration diminished and absorption occurs at higher wave number. Thus, cis is absorbed (due to the field effect) at higher frequency as compared to trans isomer. Thereby, L2 and L3 showed two bands for C=O, one of them 8 (at high frequency) associated with syn/anti-cis-structure while that at lower frequency is related to staggered configuration. The low intensity of high frequency bands (near 1700 cm-1) compared with that at low frequency (near 1660 cm-1) indicates the domination of staggered structure. Worthy mention, NMR spectroscopy is in good agreement with this suggestion. The observation of OH (phenolic) group in L1 at lower position (3149 cm-1) is taken as evidence to the persistence of interamolecular H-bonding (O–H….N) between the phenolic-OH and azomethine group in solid state [28]. The proposition of intermolecular H-bond (O–H….O-H) between associated ligand molecules is excluded owing to the sharpness of this band. The remarkable downward frequency shift indicates the strength of this bond. In fact, 1H NMR did not distinguish this H-bond in its spectra because execution of NMR analysis in high polar solvent (DMSO) led to break these bonds. The phenolic-OH group did not form H-bonding in ligands L2 and L3 where it is observed after 3400 cm-1. Furthers, the existence of two bands at 3278 cm-1 (L1) and 3227 cm-1 (L4) may be attributed to ν(OH)enolic in H-bond bonding with azomethine group. The lower shift for these two bands than the normal value asserts the weakness of this bond. The obscure of enolic OH in NMR spectra of L1 and L4 may be due to two factors: (1) its lower concentrations. Minute molecules may change one or two of its C=O groups into enolized configuration but the keto form is still dominated, (2) dilution by DMSO disrupted these H-bonds returning the modified molecules into keto forms. The significant ν(C-O) groups associated to the aromatic ring of the dihydrazones L1-L4 were observed at 1275, 1287, 1246 and 964 cm-1, respectively [28,29]. All of the investigated hydrazones ligands showed 3-4 bands in the range 1400-1600 cm-1 related to ν(C=C) of the aromatic ring. Also, each ligand exhibited strong band in the region 700-800 cm-1 corresponding to the out of plane deformation of the aromatic ring. The positions of other bands assigned to υ(CH)aromatic near 2900 cm-1 and υ(CH)aliphatic near 3050 cm-1 are demonstrated in Table 3. Pt(IV) complexes: The metal - ligand interaction in case of both I and III as well as II and IV are synonymous. For I-III, disappearance of the (OH)phenolic and (NH) accompanied with the negative shift of (C=O) of the related free ligand, upon complexation, referred to the diketo structure and indicated the coordination of the free dihydrazones through C=O and 9 deprotonated (NH and phenolic OH) groups. Foundation of C=O as one band in III, 1607 cm-1, instead of two bands in L3 (1689, 1651 cm-1) agrees with busyness of the oxygen electrons of the two carbonyl (O=C-C=O) groups in coordination with the central metal ion even though the foundation of cis direction of the two carbonyl groups. On the other hand, the higher frequency shift of the band assigned to (C-O) phenolic by chelation besides issuing of (M-Ophenolic/carbonyl) manifest the bonding with deprotonated phenolic hydroxyl in addition to carbonyl groups [30]. The remaining of the azomethine (C=N) group at nearly the same positions upon complexation substantiated the uninvolvement of the azomethine in coordination. In case of II and IV, obscurity of both ν(NH) and ν(C=O) after chelation with splitting of ν(C=N) and frequency shift of ν(C-Onaphthoic/methoxy) evidenced that the corresponding ligand binds to the platinum(IV) in dienol form via the azomethine and ohydroxyl/methoxy groups [30,31]. Finding of two bands assignable to ν(OHphenolic), 3439 cm-1, and ν(M-OHphenolic), 522 cm-1, supports this suggestion. Also, the two new bands attributed to ν(M-Oenolic), 525 cm-1 [32], and ν(M-Omethoxy), 460 cm-1, manifest the formation of PtIV complexes where the chlorides compensate the positive charge on the metal ions [33,34]. On the other hand, crystalline and coordinated water in all platinum (IV) species (I, III) were supposed based on the appearance of the broad bands within the range 3300-3500 cm-1, Table 3, [34,35]. Finally, The slight changes in the positions of ν(C=C)ph for all free ligands upon complexation result from metal-ligand interaction. 3.2. 1H NMR Spectra The assignment of the main signals in 1H NMR spectra of the ligands are given in Table 2. 1H NMR spectra of all ligands exhibited multiple signals of the aromatic protons in the 6.5–8.5 ppm region. The signals of equal integration observed in L1, L2 and L3 at δ (12.6, 11), (12.8, 12.6) and (12.9, 11.8) ppm downfield of TMS have been assigned to NH and ortho-OH protons, respectively. On the other hand, L4 revealed a signal at 12.3 ppm attributed to secondary NH group. Upon the addition of D2O then OH and NH signals were obscured. Furthers, the existence of the δOH (phenolic/naphthoic) at its 10 normal frequency excluded any instructive intramolecular hydrogen bond between orthoOH and the CH=N groups (C=N…..H-O). The azomethine signals, δ(CH=N), observed only in L1, L2 and L4 have been assigned at 8.75, 9.73 and 8.95 ppm, respectively. As reported in literature [32], if the dihydrazone exists in the syn-cis-configuration or staggered configuration, the δOH, δNH and δCH=N, resonances, each should appear as a singlet. However, the appearance of these signals in the form of six signals (doublet of doublet) indicates anti-cis (chair) configuration. Actually, the features of the 1H NMR spectra of the dihydrazones are in consistent with the syn-cis- or staggered configuration. According to IR interpretation mentioned above, the staggered configuration is well defined/dominated for all ligands (L1-L4). 1H NMR spectra of L3 and L4 differ from other ligands spectra where they showed signals at 2.55 and 3.9 ppm downfield of TMS due the methyl (CH3) and methoxy (OCH3) protons, respectively [1,8]. The 1H NMR spectra of the diamagnetic Pt(IV) complexes 1H NMR spectra of the complexes are not given owing to their remarkable insolubility feature in CHCl3 or DMSO. 3.3. Electronic spectra and magnetic studies The assignments of the observed electronic absorption bands of the oxaloylhydrazones and their platinum complexes [34,36,37] as well as the magnetic data of the formed chelates are shown in Table 3. The electronic data of the studied ligands in DMF exhibited five absorption bands at λmax (nm) equals 350-392 (n-π*, C=N), 338-375 (n-π*, C=O), 306-340 (π-π*, C=N), 292-315 (π-π*, C=O), 280-303 (π-π*, aromatic ring)]. The essential features of the spectrum of the complex are entirely different when compared with its corresponding fee ligand, which supports the coordination of the ligands to the metal ions. The electronic spectra of PtIV complexes in DMF (Table 3) revealed two d-d broad bands at 525-570 and 410-520 nm related to 1A1g→3T1g and 1A1g→1T1g transitions, respectively. Another broad band was observed in PtIV-L1 (I) at 450 nm attributed to 1 Ag→1T2g transition. The observation of these bands suggested an octahedral geometry around the Pt(IV) ion. Pt(IV)-complexes have a diamagnetic nature. In the light of the forgoing results, the most reasonable structures of the metal complexes can be represented by Fig. 3. 11 3.4. Optical properties The optical and the electrical properties of metal complexes have recently interesting due to their great potential application in Schottky diode, solid state devices and optical sensor [38]. To clarify the conductivity type of the isolated complexes, the optical band gap (Eg) of oxaloyldihydrazone ligands and their Pt(IV) complexes have been calculated from the following equations: The measured transmittance (T) was used to calculate approximately the absorption coefficient (α) using the relation, α = 1/d ln(1/T), where d is the width of the cell. The optical band gap was estimated using Tuac’s equations [23–24]: αhʋ = A (hʋ-Eg)m , where m is equal to 1/2 and 2 for direct and indirect transition, respectively and A is an energy independent constant. The values of α calculated from the first equation was used to plot (αhʋ)2 vs. hʋ from which an indirect band gap was found by extrapolating the linear portion of the curve to (αhʋ)2 = 0. The values of indirect optical band gap Eg were determined and given in Table 2. The Eg values of oxaloyldihydrazone ligands L(L1-L4) and Pt(IV)-L complexes in DMF solvent were found to be at 3.06-3.42 and 2.52-3.20 eV, respectively as indicated in Fig. 4. As seen in Table 2, optical band gap values of all ligands (Eg) were reduced upon complexation. As reported in literature [39] it is suggested that after complexation, metal leads to raise mobilization of the ligand electrons by accepting them in its shell. It can be evaluated that after formation of the complex, the chemical structure of the ligands is changed, the width of the localized levels is expanded and in turn, the band gap is smaller. This result is very significant in applications of electronic and optoelectronic devices, because of the lower optical band gap of the materials [40]. Small band gap facilitates electronic transitions between the HOMO-LUMO energy levels and makes the 12 molecule more electro-conductive [41]. Upon excitation, some of the electrons get enough energy to jump the band gap in one go. Once they are in the conduction band, they can conduct electricity, as can the hole they left behind in the valence band. The hole is an empty state that allows electrons in the valence band some degree of freedom. In essence, the obtained band gap values suggest that these complexes are semiconductors [39] and lie in the same range of highly efficient photovoltaic materials. So, the present compounds could be considered potential materials for harvesting solar radiation in solar cell applications [39,42]. The little difference in the optical band gap Eg values between all studied complexes may be due to their synonymous chemical structures. As the refractive index is an important parameter for optical applications, it is important to determine it for the ligands and their complexes. The evaluation of refractive indices of optical materials is of considerable importance for applications in integrated optic devices such as switches, filters and modulators. Besides, the refractive index is an important physical parameter, which is widely used in chemistry to identify the liquid, or its purity. For this purpose, optical properties were investigated for the compounds by spectrophotometric measurement of transmittance, T, and reflectance, R, in the wavelength range of 190–900 nm. The optical reflectance spectra (R) for oxaloyldihydrazone ligands and their Pt(IV) complexes as a function of wavelength (λ,nm) are shown in Fig. 5. The reflectance has been estimated using the relationship, R+T+A=1, for normal reflectance, the refractive index value (n) of the compounds may be approximately expressed by the relation [43], n = (1+√R) / (1-√R), where R is the normal reflectance. The variations in the refractive index (n) with wavelength (λ,nm) is shown in Fig. 6. The variation of n values with frequency range indicates that some interactions take place between photons and electrons [44]. Moreover, Fig. 6 indicates that the complexeation affects the refractive index of the free ligands where there is a difference in the n values upon connection. 3.5. Corrosion inhibition 13 Weight loss data of C-steel in 1M HCl in absence and presence of various concentrations of inhibitors were obtained and depicted in Table 4. It is obvious from this table that the weight loss decreases as the concentrations of different compounds increase. Also, it was observed that inhibition efficiencies increased with increasing of inhibitor concentrations. This behavior resulted from the adsorption of hydrazones on the metal coupons/solution interface where the adsorbed species mechanically screen the coated part of the metal surface from the action of the corrosive medium. This adsorption could be attributed to physico-chemical properties of the investigated ligands molecules as functional groups (OH, NH, C=O and C=N), quite large molecular size, high molecular weight, aromaticity, electron density at the donor atoms (N:, :O:) and also the -orbital character of azomethine, carbonyl and phenyl groups. The order of inhibition efficiency of hydrazone compounds is: L4 > L3 > L1 > L2 and this trend can be interpreted as follows. The difference in the inhibition efficiencies of the four compounds lies in their steric factor and electron density at the donor atoms. Nevertheless, all hydrazone ligands can attach to the metal surface, the persistence of methoxy (OMe) group in (L4) which is an efficient electron donating group than phenolic-OH group present in other ligands (L1-L3) facilitates the adsorption on the metal surface. Positive mesomeric effect (+M effect) and negative inductive effect (–I effect) of -OCH3 leads to increase the electron density at phenyl ring which pushes the electrons towards the donor atoms in O=C-NH-N=CH part by virtue of resonance and subsequently enhances the bond strength between the molecule and the corroding metal [49]. Under these circumstances, the inhibition efficiency of (L4) should be higher than other ligands. L3 comes after L4 due to the electron donating nature of methyl group attached to the azomethine (-N=C(CH3)) which increases the electron density on the azomethine group causing high bond strength between ligand and metal surface. Perhaps, L1 comes before L2 because the electron pair on the ortho-OH group reachs directly to the azomethine group upon delocolization with one phenyl ring while in case L2 the existence of two naphthyl rings at each side of molecule may cause crowding (steric factor) that encourage incomplete coating besides they can pull or withdraw the electron pair from the ortho-OH group to themselves under the resonance effect. The order of 14 increasing inhibition efficiency of the hydrazone derivatives set out in Table 4. The obtained data provided a convincing evidence for the application of the used ligands specially L4 as corrosion inhibitors. The maximum efficiency for L4 is 47% for steel, which indicates an acceptable inhibition for C-steel. 3.5.1. Adsorption isotherms and Gibb’s free energy Values of (θ, degree of surface coverage) calculated from weight loss measurements have been used to study the adsorption characteristics of the inhibitors on the surface of metal (C- steel). The data obtained at room temperature nearby 303 K were tested to fit curves for different adsorption isotherms including Freundlich, Temkin, Langmuir, Frumkin, and Flory-Huggins, and the correlation coefficients (R2) were used to determine the best fits [46]. The Gibb’s free energy of adsorption ΔGads at room temperature, which can characterize the interaction of adsorbed molecules and metal surface, for all choice isotherms are calculated according to the relation [47]: ∆Gads= -RTln55.5Kads, where: T is the temperature (in K) 3.5.1.1. Freundlich isotherm Freundlich isotherm is given by the relation [48] lnθ = lnKads + 1/nlnC, where C is the inhibitor bulk concentration in mol/L, Kads.(mol-1L) is the equilibrium constant of adsorption and n is Freundlich heterogeneity factor. Figure 7 shows the plot of surface coverage θ versus logarithmic concentration of all ligands. The linear plot with acceptable correlation coefficient were obtained indicating that the adsorption of the inhibitors onto different metal surface can be described by Freundlich isotherm. The adsorption parameters obtained for the Freundlich adsorption isotherm are tabulated in Table 5. In the present study for different inhibitors, n values are greater than unity implying favored adsorption process [49]. 3.5.1.2. Langmuir adsorption 15 This isotherm may be formulated as [50]. (C/θ) = (1/K) + C, where: K is the equilibrium constant for adsorption process. Figure 7 represents the plot of logC/θ versus log C for different compounds relative to C-steel. The obtained linear plots with high correlation coefficients indicates that the adsorption of the inhibitors onto different metal surface follows Langmuir adsorption isotherm. From the resulting data one can postulates that there is no interaction between the adsorbed species. The equilibrium constant (K) for adsorption desorption can be calculated from the reciprocal of anti logarithm of the intercept. The thermodynamic parameters for adsorption process, Kads and ΔGads, associated to the investigated compounds were inserted in Table 5. 3.5.1.3. Frumkin adsorption Frumkin adsorption isotherm is given by [51] LogC[θ/(1-θ)] = 2.303 Log k + 2αθ, where: K is the adsorption-desorption constant and α is the lateral interaction term describing the interaction in adsorbed layer. Plots of LogC[θ/(1-θ)] against θ as presented in Fig. 7 were linear with acceptable correlation coefficients suggesting the applicability of Frumkin isotherm. The values for Frumkin adsorption parameters are recorded in Table 5. The results pointed out that values of the adsorption parameter α are positive suggesting the attractive behavior of the inhibitor on the surface of carbon steel [52]. 3.5.1.4. Adsorption consideration The inhibition effect of inhibitor compound is ascribed to adsorption of the molecule on metal surface. This adsorption may be physical or chemical depending on the adsorption strength. For a physical adsorption mechanism, the value of the free energy (ΔGads) of adsorption should be less negative than -40 kJ/mol. The negative values of ΔGads set out in Table 5 suggest spontaneous adsorption of the inhibitor and characterize their strong 16 interaction with the metal surface [53]. The magnitudes of ΔGads (>-40 KJ/mol) indicate that adsorption on metal surface is in good agreement with physical adsorption mechanism [53,54]. Subsequently, the results support the assertion that the mechanism of corrosion inhibition is due to the formation of a protective film on the metal surface where the additive covers both the anodic and cathodic sites. 4. Conclusion A series of oxaloyldihydrazone ligands (L1–L4), was prepared essentially by the usual condensation reaction between oxaloyldihydrazide and different aldehydes such as salicylaldehyde, 2-hydroxy-1-naphthaldhyde, 2-hydroxyacetophenone and 2- methoxybenzaldhyde in 1L:2M molar ratio. The formed compounds were purified by crystallization to give bis(salicaldhyde) oxaloyldihydrazone (L1), bis(2-hydroxy-1naphthaldhyde) oxaloyldihydrazone hydroxyacetophenone)oxaloyldihydrazone(L3), (L2), bis(2bis(2-methoxy- benzaldhyde)oxaloyldihydrazone (L4). The isolated oxaloyldihydrazones (L1-L4) and their four solid complexes with Pt(IV) ion can be prepared by normal condensation method. Comparison of the elemental analysis for both the calculated and found percentages indicates that the compositions of the solid complexes coincide well with the proposed formulae. Elemental analyses (CHN), UV–Vis, FT-IR spectroscopy and 1H NMR spectra evinced the formation of the intended dihydrazones and their Pt(IV)complexes. The dihydrazone is bonded in I and III as a dibasic diketo hexdentate ligand via phenolate and carbonyl oxygens (C=O) in addition to deprotonated secondary amine giving dinuclear complexes. In II and IV, the ligand acted as a tetrabasic dienol hexadentate ligand through oxygen of ortho-OH/OMe, azomethine nitrogen and enolized carbonyl (C-OH) giving dinuclear complexes else. On the basis of various physicochemical data presented and discussed above, the complexes may tentatively be suggested to have octahedral structures. The indirect band gap energy (Eg) for all separated compounds lies in the range of semiconductor materials. The results of corrosion inhibition presented in this work points to the ability of all ligands to inhibit the corrosion of C-steel in HCl solution to an acceptable extent. The inhibition efficiencies of 17 the inhibitors increase with increasing the inhibitor concentration and followed the trend: L4 > L3 > L1 > L2. The obtained values of Gibb’s free energy is attributed to physical adsorption of the ligands on the surface of the C-steel. The experimental data fit Freundlich, Langmuir and Frumkin adsorption isotherms for all investigated oxaloyldihydrazones inhibitors. References [1] E. M. Sherif, A.H. Ahmed, Synth React Inorg Met-Org Chem. 40 (2010) 365–372. [2] M. Ghasemian, A. Kakanejadifard, F. Azarbani, A. Zabardasti, S. Shirali, Z. Saki, S. Kakanejadifard, Spectrochim. Acta 38 (2015) 643-647. [3] A.H. Ahmed, E. Ewais, J. Chem. Pharm. Res. 4(7) (2012) 3349-3360. [4] J.M. Price, Federation Proc. 20(3) (1961) 223–226. [5] V.P. Singh, A. Katiyar, Pestic. Biochem. Phys. 92(1) (2008) 8–14. [6] V. P. Singh, P. Singh, A. K. Singh, Inorg. Chim. Acta 379(1) (2011) 56–63. [7] M. Challaa, T. S. Reddy, African Journal of Pure and Applied Chemistry 5(13) (2011) 442–447. [8] H. Abd El-Wahab, M. Abd El-Fattah, A.H. Ahmed, A.A. Elhenawy, N.A. Alian, J. Organomet. Chem. 791 (2015) 99–106. [9] J. Yuan, D. B. Lovejoy, D.R. Richardson, Blood 104(5) (2004) 1450–1458. [10] S. Rollas, S. G. Küçükgüzel, Molecules 12(8) (2007) 1910–1039. [11] M. Salavati-Niasari, A. Sobhani, J. Mol. Catal. A: Chemical 285 (2008) 58–67. [12] R.A. Lal, S. Adhikari, A. Kumar, J. Chakraborty, S. Bhaumik, Synth. React. Inorg. Met.-Org. Chem. 32(1) (2002) 81–96. 18 [13] R.A. Lal, D. Basumatary, S. Adhikari, A. Kumar, Spectrochim. Acta A: 69(3) (2008) 706–714. [14] A.H. Ahmed, Rev. Inorg. Chem. 34(3) (2014) 153-175. [15] A.H. Ahmed, M.S. Thabet, Syn. and Rea. in Ino. and Metal-Org. Chem., (2013), In press. [16] A.H. Ahmed, A.G. Mostafa, Mate. Sci. Eng. C: 29(3) (2009) 877–883. [17]. A.H. Ahmed, M.S. Thabet, J. Mol. Struct. 1006 (2011) 527–535. [18] M.S. Thabet, A.H. Ahmed, J. Porous Mater. 20 (2013) 319–330. [19] N.O. Eddy, S.A. Odoemelam, J. Surface Sci. Technol. 24 (2008)1-14. [20] S. Nega, J. Aionso, A. Diazj, F. Junquere, J. Heterocycl. Chem., 27(2) (1990) 269273. [21] A.S. Fouda, M. Abdallah , S.M. Al-Ashrey, A.A. Abdel Fattah, Desalination, (2010) 250-538 [22] T.M. Salama, A.H. Ahmed, Z.M. El-Bahy, Micropor. Mesopor. Mater., 89, (2006) 251-259. [23] M.M. Rashad, A.M. Hassan, A.M. Nassar, N.M. Ibrahim, A. Mourtada, Appl. Phys. A117 (2013) 877-890. [24] M.M. Rashad, A.O. Turky, A.T. Kandil, J. Mater. Sci.: Mater. Electron. 24 (2013) 3284-3291. [25] K. Orubite-Okorosaye, N. C. Oforka, J. Appl. Sci. Environ-Mgt. 8(1) (2004) 57– 61. [26] I.A. Mohammed-Dabo, S.A.Yaro, G. Abubakar, S.I. Ayilara, T.U. Apugo-Nwosu, S.A. Akuso, J. Basic. Appl. Sci. Res. 1 (2011) 1989– 1999. [27] R. A. Lat, S. Adhikari, A. Kumar, In. J. Chem. 36A (1997) 1063-1067. [28] K. Burger, I. Ruff, F. Ruff, J. Inorg. Nucl. Chem., 27 (1965)179-190. 19 [29] O. B. Chanu, A. Kumar, A. Ahmed, R. A. Lal, J.Mol. Struct. 1007 (2012) 257–274. [30] S. Das, R.A. Lal, Indian J. chem. 27A(1988) 225-230. [31] S. Salapathy, B. Sahoo, J. Inorg. Nucl. Chem. 32 (1970) 2224-2227. [32] A. Kumar, R. A. Lal, O. B. Chanu, R. Borthakur, A. Koch, A. Lemtur, S. Adhikari, S. Choudhury, J. Coord. Chem. 64(10) (2011) 1729–1742. [33] J.R. Ferraro, Low Frequency Vibration of Inorganic and Coordination Compounds, Plenum, New York, 1971. [34] A.M. Ali, A.H. Ahmed, T.A. Mohamed, B.H. Mohamed, J.App. Sci. Res. 3(2) (2007)109-118. [35] A.M Ali, A.H Ahmed, T.A Mohamed, B.H Mohamed, Transit. Met. Chem., 32 (2007) 461-467. [36] D. Sutton, Electronic Spectra of Transition Metal Complexes, McGraw Hill, Londan, 1968. [37] A.B.P. Lever, Inorganic Electronic Spectroscopy, Elsevier Publishing Company, Amsterdam, 1984. [38] X.T. Tao, H. Suzuki, T. Watanabe, S.H. Lee, S. Miyata, H. Sasabe, Appl. Phys. Lett.70 (1997) 1503-1505. [39] F. Karipcin, B. Dede, Y. Caglar, D. Hur, S. Ilican, M. Caglar, Y. Sahin, Opt. Commun. 272 (2007) 131–137 [40] N. Turan, B. Gündüz, H. Körkoca, R. Adigüzel, N. Çolak, K. Buldurun, J. Mex. Chem. Soc. 58(1) (2014) 65-75. [41] S.K. Sengupta, O.P. Pandey, B.K. Srivastava, V. Sharma, Transit. Met. Chem. 23 (1998) 349-353. [42] M.L. Fu, G.C. Guo, X. Liu, L.Z. Cai, J.S. Huang, Inorg. Chem. Commun., 8 (2005) 18-21. 20 [43] J.I. Gittleman, E.K. Sichel, Y. Arie, Sol. Energy Mater. 1 (1979) 93-104. [44] F. Yakuphanoglu, H. Erten, opt. Appl. 4 (2005) 969-976. [45] M. Abdallah, A. S. Fouda, S. A. Shama, E. A. Afifi, African Journal of Pure and Applied Chemistry 2 (9) (2008) 83-91. [46] Karakus, M., M. Sahin and S. Bilgic, Mater. Chem. Phys. 9 (2005) 561. [47] J. Rodosevic, M. Kliskic, L.J. Aljinovic, S. Vuko,.In the Proceedings of the 8th European Symposium on Corrosion inhibition, Ann Univ. ferrara, Italy, 1995, pp: 817. [48] R. Mohammad, M. Hadj, S. Mina, B. Pourya, J. appl. Sci. enviro. Sanit. 6 (2011)113. [49] A.L. Ahmad, S. Bhatia, N. Ibrahim and S. Sumathi, Braz. J. Chem. Eng. 22 (2005) 1-13. [50] H. Shockry, M. Yuasa, I. Sekine, R.M. Issa, H.Y. El-baradie, G.K. Gomma, 40 (998) 2173-2186. [51] N.O. Eddy, P. Ekwumemgbo, S. A. Odoemelam, Inter. J. Phy. Sci., 3 (2008)1-6. [52] H. Ashassi-Sorkhabi, B. Shaabani, B. Aligholipour, D. Seifzadeh, Appl. Surf. Sci. 252 (2006) 4039-4047. [53] S. Bilgic, M. Sahin, Mater. Chem. Phys. 70 (2001) 290-295. [54] N.O. Eddy, A. S. Ekop, 4 (2008)10-16. 21 Table 1. The characteristic specifications for C-steel coupons used for weight loss measurements. Specimen C Si C- steel 0.07 0.01 As Sn - - Mn P S Cr Ni 0.39 0.022 0.0001 0.04 0.04 Pb Zn 0.003 - Cu Al 0.073 0.03 Fe Mo - Co Nb Ti V W 0.009 0.016 0.0006 0.003 0.017 .013 Table 2. Analytical, physical and spectroscopic data of the oxaloyldihydrazones and their related platinum(IV) complexes. Compound. Symbol C16H14N4O4 L1 C24H18N4O4 L2 C18H18N4O4 L3 C18H18N4O4 L4 [Pt2(L1-4H)(H2O)2(Cl)4] I [Pt2(L2-2H)(Cl)6] II [Pt2(L3-4H)(H2O)2(Cl)4] III [Pt2(L4-2H)(Cl)6] .0.5H2O IV M.p(oC) Colour >300 Yellow >300 Yellow >300 Pale Yellow >300 White >300 black >300 black >300 black >300 black Found(Calcd.)% C H N M 59.1 5.4 16.2 (58.9) (4.3) (17.2) 68.1 5.3 12.9 (67.7) (4.3) (13.2) 59.5 6.1 14.7 (61) (5.1) (15.8) 59.2 5.8 15.7 (61) (5.1) (15.8) 21.8 1.9 6.8 42.8 (21.6) (1.6) (6.3) (43.8) 28.8 1.4 6.1 39.1 (28.1) (1.6) (5.5) (38.1) 24.0 2.3 7.0 41.9 (23.5) (2.0) (6.1) (42.5) 23.1 2.2 6.5 41.4 (22.6) (1.7) (5.9) (4.8) 1 H-NMR μeff. Chemical shift (δppm) 12.6(NH, s), 10.98(OH, s), 8.75(CH=N, s), 6.6-8.40 (aromatic protons, m) 12.76(NH, s), 12.57(OH, s), 9.74(CH=N, s), 7.0-8.8 (aromatic protons, m 12.85(NH, s), 11.85(OH, s), 6.6-8.0(aromatic protons, m), 2.48(CH3, s) 12.3(NH, s), 11.9(OH, s), 8.95(CH=N, s), 6.8-8.5(aromatic protons, m), 3.93(OCH3, s) no bands (V.P.S compound) Diamg M+ Found/Calcd. 326.0/ 326.0 Eg (eV) 3.10 427.1/426.4 3.06 356.4/354.2 3.32 355.0/354.4 3.42 - 2.93 no bands (V.P.S compound) Diamg. - 2.52 no bands (V.P.S compound) Diamg - 2.90 no bands (V.P.S compound) Diamg - 3.20 22 Table 3. Significant IR and electronic absorption data of oxaloyldihydrazones and their platinum (IV) complexes. Symbol ν(OH) ν(H2O) ν(NH) phenolic (enolic) L1 L2 L3 L4 I II ν(C-H) ν(C=O) ν(C=N) ν(C-O) ν(C-OMe) aromatic (aliphatic) phenolic δ(C-H) ν(M-O) aromatic phenolic/enolic out of plane (carbonyl/methoxy) 3149 (3278) - 3204 3062 (2924) 1666 1620 1275 - 756 -/(-/-) 3476 (-) - 3166 3043 (2926) 1705 1660 1621 1287 - 741 -/(-/-) 3448 (-) - 3293 3048 (2923) 1689 1651 1607 1246 - 746 -/(-/-) (3227) - 3202 3041 (2940) 1653 1600 - 964 760 -/(-/-) (-) 3439 (-) III (-) IV (-) 3455 - 3043 (2923) 1653 1619 1284 - 744 518/(500/-) - - 3060 (2922) - 1599 1620 1289 - 755 522/(-/-) 3421 - 3010 (2923) 1629 1607 ( merged) 1290 - 756 490/(480/-) 3010 (2922) - 3431 - 1600 1615 - 1000 (merged) 758 -/-525 (-/460) λmax,nm (assignments) 390(n-π*, C=N), 350(n-π*, C=O), 340(π -π*, C=N), 303 (π -π*, C=O), 292(π -π*, Phenyl) 392(n-π*, C=N), 375(n-π*, C=O), 330(π -π*, C=N), 315(π -π*, C=O), 303(π -π*, Phenyl) 372(n-π*, C=N), 352(n-π*, C=O), 336(π -π*, C=N), 305 (π -π*, C=O), 290(π -π*, Phenyl) 350(n-π*, C=N), 338(n-π*, C=O), 306(π -π*, C=N), 292 (π -π*, C=O), 280(π -π*, Phenyl) 570(1A1g→3T1g) 520 (1A1g→1T1g) 450 (1Ag→1T2g) 540(1A1g→3T1g) 480 (1A1g→1T1g 525(1A1g→3T1g) 425 (1A1g→1T1g) 525(1A1g→3T1g) 410 (1A1g→1T1g 23 Table 4. Corrosion parameters for carbon steel coupons in (1M HCl) Solution in presence and absence of different concentrations of hydrazone ligands obtained from weight loss measurement at room temperature after 4 hrs immersion time. Inhibitor Weight Loss (g) Conc. (M) Rate of corrosion mg cm-2 h- Inhibition efficiency % θ 1 L1 Blank 5 10-4 1 10-4 5 10-5 1 10-5 0.0180 0.0120 0.0126 0.0131 0.0132 0.0046 0.0031 0.0033 0.0034 0.0034 0.32 0.28 0.27 0.26 32 28 27 26 L2 Blank 5 10-4 1 10-4 5 10-5 1 10-5 0.0180 0.0073 0.0081 0.0089 0.0096 0.0028 0.0019 0.0021 0.0023 0.0025 0.31 0.23 0.17 0.11 31 23 17 11 L3 Blank 5 10-4 1 10-4 5 10-5 1 10-5 0.0167 0.0094 0.0104 0.0110 0.0124 0.0043 0.0025 0.0027 0.0028 0.0032 0.43 0.37 0.34 0.26 43 37 34 26 L4 Blank 5 10-4 1 10-4 5 10-5 1 10-5 0.0110 0.0057 0.0059 0.0064 0.0069 0.0028 0.0015 0.0015 0.0016 0.0018 0.47 0.45 0.42 0.36 47 45 42 36 Table 5. Values of ΔGads (kJ/mol), adsorption constant (K), Freundlich heterogeneity factor (n), lateral interaction term describing the interaction in adsorbed layer (α), and correlation coefficient (R2) of inhibitors for carbon steel coupons. Freundlich isotherm Ligands Kads ∆Gads Langmuir isotherm R2 Kads ∆Gads (molL-1) (kJ mol-1) 1/n n L1 0.463 2.16 2.2530 -12.08 0.9110 0.4434 L2 0.269 3.72 2.5420 -12.39 0.9742 2.5281 L3 0.129 7.77 1.1710 -10.45 0.9693 1.1682 L4 0.064 15.5 1.2680 -10.65 0.9246 0.7870 (molL-1) (kJ mol-1) Frumkin isotherm Kads ∆Gads (molL-1) (kJ mol-1) 5.1 0.0001 -12.68 0.9433 6.8 0.0001 -13.17 0.8372 0.9993 4.3 0.0002 -10.79 0.9263 0.9996 3.1 0.0006 -8.49 0.9524 R2 α -8.02 0.9998 -12.37 0.9964 -10.44 -9.45 R2 24 L1 m/e L2 m/e Fig. 1. Mass spectra of L1 and L2. Y O H N C O N R X R C C O X N N H O C Y Staggered-configuration Y N R Y H N C H N C X O O C N C O R X O syn-cis-configuration Y H N O C N R C X O O X C O R N N H C Y anti-cis-configuration Fig. 2. Proposed structures of oxaloyldihydrazone ligands: where L1 (R=H, X=H, Y=H), L2 (R=ph, X=H, Y=H), L3 (R=H, X=H, Y=CH3) and L4 (R=H, X=CH3, Y=H). 25 Cl Cl Cl Cl Pt O O N N HC N N H N H CH Pt OH2 N H2O N O O Cl O Pt O O Cl Cl H Cl Pt Cl Cl O I Pt(IV)-L1 N H II Pt(IV)-L2 Cl Cl Cl Pt Cl O O N N H N H3C H N C N Pt OH2 O Pt O CH3 O Pt Cl Cl CH3 2.5 III Pt(IV)-L3 Pt(IV)-L4 1.2 L1 2 L2 II 1.0 2.0 0.8 2 (h) (eV/m) x10 4 I 1.5 0.6 1.0 0.4 0.5 0.2 0.0 0.0 1.5 2.0 2.5 h (eV) 3.0 3.5 1.0 1.6 2.0 2.2 2.4 h (eV) 2.6 2.8 3.0 3.2 L4 0.8 2 III 0.6 0.6 0.4 0.4 0.2 0.2 0.0 0.0 1.5 1.8 1.0 L3 2 0.8 2.0 2.5 3.0 h (eV) O Cl Fig. 3. Suggested structures of the PtIV –oxaloyldihydrazone complexes. 4 H2O O Cl Cl IV (h) (eV/m) x10 N N O Cl N 3.5 4.0 1.5 IV 2.0 2.5 h (eV) 3.0 3.5 Fig. 4. The plots of (αhʋ)2 vs. hʋ of Pt(IV) oxaloyldihydrazone complexes. CH3 C 26 20 L1 L2 I II 15 10 5 R% 0 20 15 L3 L4 III IV 10 5 0 300 400 500 600 700 300 400 500 600 700 800 (nm) Fig. 5. The optical reflectance spectra (R) for oxaloyldihydrazone ligands and their Pt(IV) complexes 2.5 L1 L2 I II 2.0 1.5 n 1.0 L3 2.5 L4 III IV 2.0 1.5 1.0 300 400 500 600 700 300 400 500 600 700 800 (nm) Fig. 6. The variation of the refractive index (n) with wavelength (λ) for oxaloyldihydrazone ligands and their Pt(IV) complexes. 27 0.0 L1 R2 = 0.9742 -0.5 (a) L1 L1 2 L2 R = 0.9110 -1.0 L2 L2 -1.5 L3 L3 ln ln θ -2.0 L3 R2 = 0.9693 L4 R2 = 0.9246 (a) L4 L4 -2.5 -12.0 -11.0 -10.0 lnlnC C -9.0 -8.0 -7.0 -2.5 (b) L1 R2 = 0.9998 Log (C/θ) -3.0 L2 R2 = 0.9964 L1L1 L3 R2 = 0.9993 -3.5 L2L2 L4 R2 = 0.9996 L3L3 -4.0 L4L4 -4.5 -5.0 -5.5 -5.0 -4.5 -4.0 -3.5 -3.0 Log C -3.0 (c) Log [C(θ/1-θ)] -3.5 L1L1 -4.0 L2L 2 -4.5 L1 R2 = 0.9433 -5.0 L3 L3 2 L2 R = 0.8372 -5.5 L4 L4 L3 R2 = 0.9263 -6.0 2 L4 R = 0.9524 -6.5 0.2 0.3 0.4 0.5 0.6 0.7 θ Fig. 7. Freundlich (a), Langmuir (b) and Frumkin (c) isotherms for C-steel coupons in HCl solution containing oxaloyldihydrazone ligands as inhibitors.