* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Hypothalamic huntingtin-associated protein 1 as a mediator of feeding behavior
Survey
Document related concepts
Transcript
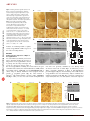
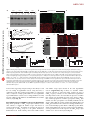
ARTICLES © 2006 Nature Publishing Group http://www.nature.com/naturemedicine Hypothalamic huntingtin-associated protein 1 as a mediator of feeding behavior Guoqing Sheng1, Guo-qing Chang2, John Y Lin3, Zhao-Xue Yu1, Zhi-Hui Fang1, Juan Rong1, Stuart A Lipton3,4, Shi-Hua Li1, Gang Tong3,4, Sarah F Leibowitz2 & Xiao-Jiang Li1 The hypothalamus responds to circulating leptin and insulin in the control of food intake and body weight. A number of neurotransmitters in the hypothalamus, including c-aminobutyric acid (GABA), also have key roles in feeding. Huntingtinassociated protein 1 (Hap1) is expressed more abundantly in the hypothalamus than in other brain regions, and lack of Hap1 in mice leads to early postnatal death. Hap1 is also involved in intracellular trafficking of the GABAA receptor. Here, we report that fasting upregulates the expression of Hap1 in the rodent hypothalamus, whereas intracerebroventricular administration of insulin downregulates Hap1 by increasing its degradation through ubiquitination. Decreasing the expression of mouse hypothalamic Hap1 by siRNA reduces the level and activity of hypothalamic GABAA receptors and causes a decrease in food intake and body weight. These findings provide evidence linking hypothalamic Hap1 to GABA in the stimulation of feeding and suggest that this mechanism is involved in the feeding-inhibitory actions of insulin in the brain. The hypothalamus integrates central and peripheral signals, such as leptin and insulin, to regulate food intake and energy balance1–4. As the receptors for insulin and leptin are expressed in various regions of the hypothalamus, the neuronal interactions are more likely crucial for the proper actions of insulin and leptin in their control of feeding behavior. Although the complex hypothalamic circuitry that controls food intake and energy homeostasis remains to be fully characterized, it is apparent that this circuitry is crucially dependent on neurotransmitters and synaptic transmission. For example, a number of transmitters in the hypothalamus, including glutamate, GABA and serotonin, have been found to have key roles in feeding5–7. Likewise, injection of GABA or its agonist into the hypothalamus or third ventricle of the brain can stimulate eating behavior in animals8–11. This phenomenon thus raises the question of whether circulating insulin or leptin can influence the activity of GABA or other neurotransmitter receptors in hypothalamic neurons. Understanding the mechanism for the hypothalamic control of feeding behavior may also depend on our knowledge about proteins that are crucial for the normal function of hypothalamic neurons. As an example, Hap1, which was initially identified as an interacting partner for the Huntington disease protein12, seems to be crucial for the normal function of the hypothalamus, while also perhaps playing a role in feeding. Firstly, Hap1 is very abundant in the hypothalamus13–16. Secondly, gene-targeting knockout experiments show that lack of the Hap1 gene in mice causes retarded growth and postnatal death, possibly owing to a feeding defect17–19. Thirdly, the deletion of Hap1 causes the selective degeneration of hypothalamic neurons18. The role of Hap1 in hypothalamic function is also suggested by the hypothalamic neuropathology in Huntington disease. Although neurodegeneration preferentially occurs in the striatum in the brains of individuals with Huntington disease, it also extends to other brain regions including the hypothalamus during late stages of the disease20,21. The hypothalamic dysfunction in Huntington disease may contribute to the progressive loss of body weight22,23. Similarly, loss of body weight and degeneration of hypothalamic neurons are characteristics of some mice with transgenically induced Huntington disease that express N-terminal fragments of mutant Huntington disease protein huntingtin (Htt)18,24. Because mutant Htt binds more tightly to Hap1 than does normal Htt13,25, it is likely that dysfunction of Hap1 is involved in the hypothalamic pathology in Huntington disease. It remains unclear, however, whether and how Hap1 is involved in feeding behavior under normal conditions. Growing evidence suggests that Hap1 participates in intracellular trafficking25–27. A more relevant finding for the potential role of Hap1 in feeding is that Hap1 interacts with b subunits of the GABAA receptor and stabilizes the internalized GABAA receptors in cultured cells, thereby increasing GABAA receptor activity28. Here, we focused on the role of Hap1 in the hypothalamus. We show that fasting can increase the level of Hap1 in the hypothalamus, whereas insulin decreases Hap1 through ubiquitination. Suppressing expression of Hap1 in the hypothalamus through RNA interference reduces food intake and body weight of mice. Moreover, reducing Hap1 decreases the level and activity of GABAA receptors. Given the stimulating effect of GABA on eating behavior, we propose that Hap1 functions as a 1Department of Human Genetics, Emory University School of Medicine, 615 Michael Street, Atlanta, Georgia 30322, USA. 2Laboratory of Behavioral Neurobiology, The Rockefeller University, 1230 York Avenue, New York, New York 10021, USA. 3Center for Neuroscience and Aging, The Burnham Institute for Medical Research, 10901 North Torrey Pines Road, La Jolla, California 92037, USA. 4Department of Neurosciences, University of California San Diego, 9500 Gilman Drive, La Jolla, California 92093, USA. Correspondence should be addressed to X.-J.L.([email protected]). Received 23 January; accepted 23 February; published online 9 April 2006; doi:10.1038/nm1382 NATURE MEDICINE ADVANCE ONLINE PUBLICATION 1 Control Fasted DMH VMH ARC Fasted b Control 1 2 LC Fasted 3 1 2 HC LC 3 1 2 3 1 2 * 1.4 c HC 1.2 3 1 Hap1/tubulin Control Hap1 0.8 * 0.6 0.4 0.2 Tubulin 0 Control Fasted LC e 0.15 0.075 0.1 0.05 0.05 0.025 Insulin (ng/ml) 0 DMH VMH ARC Control Fasted ** 3 0 LC 2 HC * 3 2 1 1 0 0 Control Fasted LC HC mice that were previously established by us18. Fasting caused an increase in the level of soluble Hap1, as indicated by a more diffuse cytoplasmic distribution of Hap1 in high-magnification photomicrographs (Fig. 1a). Western blotting of the soluble fraction of hypothalamic tissue and densitometry analysis confirmed these immunocytochemical results (Fig. 1b,c). Conversely, consumption of a high-carbohydrate diet (70% carbohydrate) reduced the expression Glucose IP c Control DMH Glucose IP 3 G 1 G 2 G 3 G 4 Control DMH 0.1 HC ** C b Fasted ** 2 Glucose IP Hap1/Actb d 1 Control Hipp HC LH Control RESULTS Fasting increases expression of Hap1 in the hypothalamus We used immunocytochemistry and western DMH blots to determine whether the expression of VMH Hap1 in the mouse hypothalamus is altered ARC by fasting and diet. Hap1 transcripts and protein are expressed in various nuclei of the mouse hypothalamus, including the arcuate nucleus, the paraventricular nucleus (PVN), the ventromedial nucleus (VMH) and the dorsomedial nucleus (DMH; Fig. 1a and Supplementary Fig. 1 online). Hap1 is diffusely distributed in the cytoplasm and also presents as cytoplasmic puncta (Fig. 1a), whose function is unknown14. This immunostaining is specific to Hap1 (Supplementary Fig. 2 online), as it does not occur in brains of Hap1 knockout a LC C mediator for circulating insulin to regulate activity of hypothalamic GABA receptors in the control of feeding behavior. a C Figure 1 Fasting and diet regulate expression of Hap1 in the mouse hypothalamus. (a) Representative images (original magnification, upper panels, 100; lower panels, 630) of Hap1 immunostaining of hypothalamic regions from control (mice fed normal chow) and fasted mice and mice fed a high-carbohydrate (HC) or lowcarbohydrate (LC) diet. LH, lateral hypothalamus, DMH, dorsomedial hypothalamus; VMH, ventromedial hypothalamus; ARC, arcuate nucleus. (b) Western blotting analysis of the expression of hypothalamic Hap1. (c) Densitometry analysis of the relative level of Hap1 (ratio of Hap1 to tubulin, *P o 0.05) in the hypothalamus of mice in each group (mean ± s.e.m, n ¼ 3). (d) Radioactive in situ hybridization shows Hap1 transcript expression in the hypothalamus of fasted and control rats. (e) Quantitative RT-PCR analysis of Hap1 expression (mean ± s.e.m, n ¼ 10) in control and fasted rats and rats fed a high-carbohydrate (HC) or low-carbohydrate (LC) diet. Plasma insulin levels (ng/ml) of the rats are also shown (lower panel). *P o 0.05, **P o 0.01. Hap1 Tubulin Tha VMH LH Amg DMH VMH ARC VMH Hap1/tubulin © 2006 Nature Publishing Group http://www.nature.com/naturemedicine ARTICLES 1.4 1.2 1 0.8 0.6 0.4 0.2 0 ** Control Glucose IP Figure 2 Intraperitoneal injection of glucose reduces expression of Hap1 in the hypothalamus. (a) Hap1 immunostaining of brain sections from mice injected intraperitoneally (IP) with saline (Control) or 10% glucose for 4 h. Hap1 is selectively reduced in various hypothalamic nuclei. LH, lateral hypothalamus, DMH, dorsomedial hypothalamus; VMH, ventromedial hypothalmus; ARC, arcuate nucleus. Other brain regions shown include the hippocampus (Hipp), thalamus (Tha) and amygdala (Amg). (b) High-magnification images of the PVN and DMH showing that glucose injection also reduces the expression of Hap1. (c) Western blotting and densitometry analysis of hypothalamic tissues from control and glucose-injected mice (mean ± s.e.m., **P o 0.01). Antibodies to Hap1 and tubulin were used to probe the blots. 2 ADVANCE ONLINE PUBLICATION NATURE MEDICINE ARTICLES Insulin ICV a ACSF 2 h 5 mU 10 min 5 mU 1 h 20 mU 2 h e Glucose 2 h Control 1 Insulin 2 3 1 2 3 Hap1 DMH Tubulin 5 0.4 0 AC In s Control c 4 tro l 1 0.8 on Glucose 3 * 0.8 SF Tubulin 2 0.6 0.4 Glucose (mM) 5 0.2 0 1 2 1 1.2 10 2 15 1 2 Tubulin Insulin 0.8 0.4 0 5 10 15 30 Glucose (mM) Control DMH 1 2 Insulin PVN DMH 1 0.5 0 pt Le C on tro l DMH Hap1/tubulin Tubulin Control PVN Leptin 2 Hap1 VMH ARC d 2 Hap1 1 LH 30 1 in Hap1 1 C ACSF 1.2 6 Hap1/tubulin 5 AC SF G lu co se 4 Hap1/tubulin Insulin 3 ul in 2 Hap1/tubulin © 2006 Nature Publishing Group http://www.nature.com/naturemedicine ACSF 1 * ul in ARC b 0.8 0.6 0.4 0.2 0 Hap1/tubulin VMH In s LH Figure 3 Insulin reduces expression of Hap1 in the hypothalamus. (a,b) Artificial cerebrospinal fluid (ACSF; 5 ml), insulin (5 or 20 mU/5 ml) or glucose (100 mg/5 ml) was injected into the third cerebral ventricle (ICV) of male mice. Hap1 immunostaining of hypothalamic region was performed 10 min, 1 h or 2 h after injection (a). LH, lateral hypothalamus, DMH, dorsomedial hypothalamus; VMH, ventromedial hypothalmus; ARC, arcuate nucleus. Hypothalamic tissues were analyzed by western blotting to obtain the relative level of Hap1 (Hap1/tubulin mean ± s.e.m., n ¼ 3, *P o 0.05) with densitometry (b). (c,d) Hap1 immunostaining of mouse brain slices that were incubated with ACSF (control) or insulin (0.03 mM) for 2 h. Images at low (c) and high (d) magnification are shown. PVN, paraventricular nucleus; DMH, dorsomedial hypothalamus. (e) Western blotting and densitometry analysis (mean ± s.e.m., n ¼ 2–3, *P o 0.05) of the relative Hap1 expression level (Hap1/tubulin) show that insulin, but not glucose (5–30 mM) or leptin (0.1 mM), decreased Hap1 expression in the hypothalamic region of brain slices. of soluble Hap1 compared to a low-carbohydrate diet (20% carbohydrate; Fig. 1a–c). Similar results were obtained with measurements of Hap1 mRNA using in situ hybridization (Fig. 1d) and real-time quantitative PCR (Fig. 1e). The higher expression of Hap1 in fasted rats was accompanied by lower levels of insulin than in satiated rats, whereas reduced expression of Hap1 in rats consuming a highcarbohydrate diet was accompanied by higher levels of insulin (Fig. 1e). Together, these data suggest that fasting and increasing carbohydrate consumption have opposite effects on Hap1, both in transcript and protein expression; these changes are associated with alterations in insulin levels, suggesting an inverse relationship between Hap1 and circulating insulin. Insulin inhibits the hypothalamic expression of Hap1 Fasting seems to have a more marked effect on the expression of Hap1 than the high-carbohydrate diet, perhaps because it produces a more rapid and greater change in plasma levels of insulin. To begin to test NATURE MEDICINE ADVANCE ONLINE PUBLICATION this notion, we administered 10% glucose by intraperitoneal injection, which rapidly increases pancreatic secretion of insulin29,30. This treatment decreased the expression of Hap1 in the hypothalamus as shown by immunohistochemistry (Fig. 2a,b) and western blots (Fig. 2c). To investigate whether insulin directly regulates the expression of Hap1 in the hypothalamus, we performed intracerebroventricular (into the third ventricle) injection of this hormone in mice. Immunocytochemistry and western blots showed that a single intracerebroventricular infusion of insulin (5, 10 or 20 mU/5 ml over 10 min) substantially reduced protein expression of Hap1 in the mouse hypothalamus (Fig. 3a). The largest dose (20 mU) of intracerebroventricular insulin almost eliminated Hap1 in hypothalamic areas along the third ventricle, suggesting that the insulin-induced reduction of Hap1 was dosage dependent. Western blotting verified the decrease in Hap1 expression in response to the injection of 5 mU insulin (Fig. 3b). In contrast to insulin, central injection of glucose (100 mg/5 ml) had no effect on Hap1 (Fig. 3a,b), indicating that the 3 ARTICLES on © 2006 Nature Publishing Group http://www.nature.com/naturemedicine C 4 In tro l su lin Hap1-A/tubulin change in expression of Hap1 can be attriba b Control Insulin c Control Insulin d Control Insulin uted specifically to insulin itself. Also, we Hap1-A Merged Hap1 observed no change in hypothalamic Hap1 kDa Hap1 after intracerebroventricular injection of Lysate either 5 mg/5 ml leptin or the orexigenic 172 Tubulin kDa peptide neuropeptide Y (NPY, 5 mg/5 ml; 0.4 Supplementary Fig. 3 online). 172 Ubi – – – + Insulin 0.3 – + + + 1A To confirm the direct effect of insulin on 110 + – + + Ubi the level of Hap1 in hypothalamic neurons, 0.2 * Anti-Ubi 110 we isolated hypothalamic slices from mouse 0.1 79 Antibrain and incubated them with insulin Hap1 0 Hap1 IP Hap1 IP (5 mU/ml or 0.03 mM) for 2 h. This treatment Hap1 IP Ubiquitin blot Ubiquitin blot reduced the level of Hap1, as evidenced by both immunocytochemistry (Fig. 3c,d) and Figure 4 Insulin increases ubiquitination and degradation of Hap1. (a) Colocalization of transfected western blots (Fig. 3e). It has been reported ubiquitin-HA (red) and Hap1-A puncta (green) in HEK293 cells. Hap1-A immunoprecipitates (IP) that incubation with 20 mM glucose com- showed increased ubiquitin immunoreactivity after the cells were treated with insulin (0.25 mM) for pared with 5 mM increases electrophysiolo- 2 h. (b) Immunoprecipitation of endogenous Hap1 from PC12 cells also shows increased Hap1 gical activity of neurons in hypothalamic ubiquitination (lower panel) by insulin treatment. (c) Male mice aged 8–12 weeks received slices31. Similar to intracerebroventricular intracerebroventricular injections of insulin (1 mU/5 ml). After 2 h, the hypothalamic tissues were glucose injection, various concentrations of subjected to Hap1 immunoprecipitation to obtain the ratio (mean ± s.e.m., *P o 0.05, n ¼ 3) of glucose (5–30 mM) did not significantly precipitated Hap1 to tubulin. (d) Hap1 immunoprecipitates from hypothalamic tissue were also probed with an antibody to ubiquitin. Arrowheads indicate Hap1 monomer. affect Hap1 expression in hypothalamic slices (Fig. 3e). Incubation in leptin (0.1 mM) also had no effect on Hap1 expression (Fig. 3e). Thus, insulin able to infect neurons diffusely in several nuclei of the hypothalamus seems to specifically and directly inhibit the level of Hap1 in the on the injected side, the noninjected contralateral region did not hypothalamus, whether administered intracerebroventricularly or manifest infected or GFP+ neurons (Fig. 5b). Notably, GFP+ neurons incubated directly with hypothalamic slices. showed much weaker Hap1 staining than neurons in the noninjected contralateral region, indicating that Hap1-specific siRNA efficiently Insulin increases degradation of Hap1 by ubiquitination inhibited the expression of Hap1. The diet-induced change in hypothalamic Hap1 may be mediated by Having confirmed that Hap1-specific siRNA could reduce the insulin at both the transcriptional and protein levels. Whereas long- expression of Hap1 in the hypothalamus, we conducted bilateral term regulation of central feeding-related activity by insulin could injections of either adenoviral Hap1-specific siRNA or GFP into the involve transcriptional regulation through many pathways32,33, insulin PVN of the mouse hypothalamus, a nucleus involved in the control of can also reduce the level of its receptor substrates and other proteins feeding behavior5. Examination of brain sections that clearly showed by targeting them to the ubiquitin-proteasome pathway34,35. The the PVN in both sides of the hypothalamus confirmed the presence of rapid and direct action of insulin in decreasing expression of Hap1 a large number of GFP+ neurons (Fig. 5c). Whereas food intake was led us to focus on the effect of insulin on the ubiquitination of Hap1. transiently suppressed (40% reduction) on day 1 after hypothalamic We found that ubiquitin colocalizes with Hap1 in transfected HEK293 injection of the control adenoviral GFP, the injection of adenoviral cells and that insulin can increase ubiquitin conjugation to Hap1, Hap1-specific siRNA caused a greater decrease (up to 80% reduction) representing its ubiquitination (Fig. 4a). Insulin treatment reduced in food intake, which was statistically significant (P o 0.05) for as the level of endogenous Hap1 in PC12 cells and increased the amount long as 14 d after injection (Fig. 5d). This decrease in food intake of ubiquitinated Hap1 in Hap1 immunoprecipitates (Fig. 4b). Exam- caused a significant reduction in body weight for 3 weeks (Fig. 5d). ination of Hap1 in the hypothalamus of mice that had been injected The food intake of Hap1-specific siRNA–injected mice gradually intracerebroventricularly with insulin showed a decrease in expression returned to a normal level after 2 weeks, possibly because expression of Hap1 and increase in its ubiquitination (Fig. 4c,d). Thus, similar to of the siRNA decreased over time. the effect of insulin on the degradation of insulin receptor substrate It is of interest whether the increased expression of Hap1 in the and other proteins34,35, the downregulation of Hap1 by insulin seems hypothalamus can also regulate food intake. Because early postnatal death of Hap1 knockout mice18 precludes rescue experiments through to be mediated by the ubiquitin-proteasome pathway. stereotaxic injection, we instead overexpressed Hap1-A, an isoform of Decreasing hypothalamic Hap1 suppresses food intake Hap1 that is able to stabilize membrane receptors28, in the hypothaTo further establish a role for endogenous Hap1 in feeding, we used lamus of adult mice through injection of adenoviral vectors. We RNA interference (short interfering RNA, siRNA) to suppress expres- confirmed the expression of adenoviral Hap1-A using western blotting sion of Hap1 in the hypothalamus. We generated a mouse Hap1- (Fig. 5e) and immunocytochemistry (Fig. 5f). Compared to control specific siRNA that effectively inhibits the expression of endogenous mice that were injected with adenoviral GFP alone, mice injected with Hap1 in PC12 cells (Fig. 5a). This siRNA was expressed from an adenoviral Hap1-A showed an increase in food intake and body adenoviral vector that also independently expressed green fluorescent weight after viral injection for 4–8 d (Fig. 5g). Although this result protein (GFP). Thus, cells labeled with GFP should also express Hap1- also supports the role of Hap1 in stimulating feeding behavior, the specific siRNA. An adenoviral vector expressing GFP alone served as a expression of transgenic Hap1-A was not significantly increased after control. We first performed unilateral injection of adenoviral Hap1- 8 d as compared with endogenous Hap1 (data not shown) so that specific siRNA to ensure that this injection could reduce the expres- exogenously expressed Hap1 through adenovirus temporally augmension of Hap1 in the hypothalamus. Although the injected virus was ted the effect of the abundant endogenous Hap1 in hypothalamic ADVANCE ONLINE PUBLICATION NATURE MEDICINE ARTICLES on C b Day 4 tro l H s i ap R 1 N A Day 3 Double fluorescent Noninjected GFP on Day 2 tro l H s i ap R 1 N A C on tro l H s i ap R 1 N A C on tro l H s i ap R 1 N A Day 1 C a c Injected GFP Hap1 PVN Hap1 Tubulin Hap1 Hap1-specific siRNA Ad-GFP 5 4 3 * ** Noninjected f Injected HEK293 l tro on Size Time (d) C 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 ap 1 ** ** 12 0 e * * ** * ** * -H 1 * ** * Ad 2 PC Food intake (g/d) 6 (kDa) Body weight change (percent of preinjection) 182 10 116 0 3V 82 Noninjected Injected 63 –10 49 37 –20 26 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 Time (d) * 4 3 2 1 0 0 2 4 6 8 Time (d) Ad-Hap1 10 12 14 h 10 * * 5 0 * –5 –10 0 2 4 6 8 10 Time (d) Cortex + GFP 12 14 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 ** 0 Hypo + GFP Cortex + Hap1 siRNA 4 8 Time (d) * 12 Body weight change (percent of preinjection) * Body weight change (percent of preinjection) ** 5 Food intake (g/d) Ad-GFP g Food intake (g/d) © 2006 Nature Publishing Group http://www.nature.com/naturemedicine d PVN Hypo + Hap1 siRNA 5 0 –5 –10 –15 ** –20 0 4 ** 8 Time (d) ** 12 Figure 5 Altering Hap1 expression in the mouse hypothalamus through adenoviral infection regulates food intake and body weight. (a) Western blotting of PC12 cells infected with adenoviral GFP or adenoviral Hap1-specific siRNA for 1–4 d. (b) Hap1 immunostaining shows that the adenoviral Hap1-specific siRNA–injected hypothalamic area with GFP signal has markedly reduced Hap1 expression as compared to the noninjected region. (c) A representative image of the hypothalamus shows that both PVN of a mouse bilaterally injected with Hap1-specific siRNA contain GFP signal. (d) Food intake (g/mouse/ 24 h) and body-weight change ((postinjection – preinjection)/(preinjection 100)) of mice after hypothalamic injection of adenoviral GFP or Hap1-specific siRNA. **P o 0.01, *P o 0.05 compared to adenoviral GFP. (e) Hap1 western blotting of adenoviral Hap1-A–infected HEK293 cells. PC12 cells and uninfected 293 cells (control) served as controls. (f) Hap1 immunostaining of the DMH of mouse hypothalamus injected with adenoviral Hap1-A and the noninjected contralateral region. 3V, third ventricle. Enlarged images of insets are shown in the lower panel. (g) Food intake and body weight of mice after intrahypothalamic injection of adenoviral Hap1-A (Ad-Hap1) or GFP (Ad-GFP). (h) Food intake and body weight of mice that had been injected with adenoviral Hap1-specific siRNA or GFP into the DMH in hypothalamus (Hypo+) or cerebral cortex (Cortex+). *P o 0.05, **P o 0.01 compared to adenoviral GFP injection. neurons. Thus, suppressing endogenous Hap1 is more likely to reveal the role of Hap1 in hypothalamic neurons. Along these lines, to confirm the specific effect of Hap1-specific siRNA, we compared the effects of injection of Hap1-specific siRNA into the hypothalamic region DMH and the cerebral cortex, a nonhypothalamic area. Only injection of Hap1-specific siRNA into the DMH inhibited food intake and body weight (Fig. 5h). Hap1 stabilizes the level of GABAA receptors in the hypothalamus Hap1 binds specifically to GABAA receptor b2/3 subunits, but not other subunits, to stabilize the GABAA receptor and increase its activity in cultured cells28. b2 subunits are crucial for the endocytosis and transport of GABAA receptors to the plasma membrane36. Immunofluorescent double labeling showed that HAP1 localizes NATURE MEDICINE ADVANCE ONLINE PUBLICATION with GABAA receptor b2/3 subunits in the same hypothalamic neurons (Supplementary Fig. 4 online). To determine whether decreased expression of endogenous Hap1 regulates the level of GABAA receptor b subunits, we first examined the hypothalamus from Hap1 knockout mice. We found that the expression of GABAA b2/3 subunits was substantially decreased in the absence of Hap1. Also, immunoprecipitation of HAP1 coprecipitated GABAA b2/3 subunits, and this coprecipitation was markedly diminished in the absence of Hap1 (Fig. 6a). We then examined hypothalamic Hap1 and GABAA b2/3 subunits in mice that had received hypothalamic injection of adenoviral Hap1-specific siRNA or GFP. Hap1-specific siRNA, which clearly inhibited the expression of Hap1 in the hypothalamus, caused a parallel decrease in the expression of GABAA receptor b2/3 subunits at 2, 4 and 6 d after injection (Fig. 6b). 5 2d b C 4d S S C C S Size (kDa) Size (kDa) 115 60 β2/3 e 6d S Hap1 79 37 β2/3 90 80 70 60 50 40 30 20 10 0 B max (fmol/mg protein) IP WT KO Control Insulin 20 10 0 0 5 10 15 20 0 3 C Hap1 β2/3 Tubulin 2h 4h Insulin C C 2h 4h 2h 4h Insulin C 2h 4h 0.8 * 50 pA Control Insulin 1s 20 pA * 80 60 40 20 20 ms Control Insulin h 0 Control Insulin 1.0 0.8 0.6 Control Insulin 0.4 0.2 0 0 100 200 300 mIPSC amplitude (pA) 0.6 0.4 * Cumulative frequency Insulin Insulin Cortex Insulin g Control Bicuculline d Hypothalamus f mIPSC amplitude (pA) c Relative expression (ratio to tubulin) © 2006 Nature Publishing Group http://www.nature.com/naturemedicine Tubulin Control 10 H-Muscimol (nM) Hap1 * 20 Kd (nM) Input WT KO KO a Specific binding of 3H-muscimol (fmol/mg protein) ARTICLES Plasma insulin HAP1 expression Anorexic effect GABAA receptor level 0.2 0 Hap1 β2/3 Figure 6 Decreasing Hap1 expression reduces functional GABAA receptors in the hypothalamus. (a) Hap1 immunoprecipitation of hypothalamic GABAA receptor b2/3 subunits in wild-type (WT) and Hap1 knockout (KO) mice. (b) Western blotting shows that intrahypothalamic injection of Hap1-specific siRNA (S) reduces the expression of Hap1 and GABAA receptor b2/3 subunits. Control (C) is adenoviral GFP injection. (c) Intraperitoneal injection of insulin (1 U/ kg) reduces expression of Hap1 and GABAA receptor b2/3 subunits in the hypothalamus, but not in the cortex, at 2 or 4 h after the injection. Control (C) represents 4 h after saline injection. (d) Densitometric quantification of the ratio (mean ± s.e.m., n ¼ 3, *P o 0.05) of Hap1 or GABA receptor b2/3 subunits to tubulin in the hypothalamus. (e) 3H-muscimol binding to GABAA receptor in hypothalamic tissue of mice 2 h after intracerebroventricular injection of insulin (10 mU/5 ml) or ACSF (control). Dose-dependent binding (left panel), Bmax and Kd (right panel) are shown (*P o 0.05, n ¼ 4–5). (f) Representative mIPSCs of hypothalamic slices incubated with and without insulin. Bicuculline is a competitive antagonist at GABAA receptors. mIPSC amplitudes recorded from insulin-treated slices (n ¼ 14) are significantly (*P o 0.05) smaller than those of control slices (n ¼ 13). (g) Averaged cumulative probability plots of mIPSC amplitude in control and insulin-treated slices. (h) Proposed role of Hap1 in feeding behavior. Examination of insulin receptor, NPY and orexin A showed that their levels were not changed in the hypothalamus of HAP1 knockout mice (Supplementary Fig. 5 online), suggesting that the effect of Hap1 on GABAA receptors is specific. We also found that ghrelin did not alter the level of HAP1 in cultured hypothalamic slice (Supplementary Fig. 5). We therefore focused on the effect of insulin on Hap1 and we asked whether circulating insulin, which is able to decrease hypothalamic Hap1, also produces changes in the expression of GABAA receptors. We administered insulin (1 U/kg) to mice by intraperitoneal injection and examined the expression of Hap1 and GABAA b subunits in the hypothalamus 2 and 4 h later. The level of Hap1 was reduced in the hypothalamus but not in the cortex (Fig. 6c), consistent with the specific effect of plasma insulin on hypothalamic neurons. Notably, insulin specifically reduced the level of GABAA b2/3 subunits in the hypothalamus, but not in the cortex, suggesting that this reduction depends on Hap1, which is more abundant in the hypothalamus than in other brain regions. Densitometric quantification of the relative amount of Hap1 and GABAA b2/3 subunits validated this observation (Fig. 6d). To confirm the direct action of insulin on the function of hypothalamic GABAA receptors, we measured the binding of GABAA receptor to its agonist, 3H-muscimol, in the hypothalamus of mice after intracerebroventricular injection of insulin. Insulin treatment significantly (P o 0.05) decreased the number of 3H-muscimol binding sites (Bmax) but only slightly reduced the binding affinity (Kd; Fig. 6e). This finding is consistent with the notion that the number of GABAA receptors decreased. 6 To investigate the effect of insulin on GABA inhibitory synaptic transmission, we recorded miniature inhibitory postsynaptic currents (mIPSCs) from neurons of ventromedial hypothalamus in brain slices. Insulin treatment significantly reduced (P o 0.05) the mean amplitude of mIPSCs of hypothalamic neurons (Fig. 6f) but had no significant effect on the rise time of mIPSCs (20–80% rise time in ms; control, 0.48 ± 0.03; insulin, 0.52 ± 0.04), or the decay time constant (ms) of mIPSCs (control, 7.33 ± 0.82; insulin, 7.19 ± 0.57). These results suggest that insulin decreased density of synaptic GABAA receptors. In addition, insulin treatment significantly decreased (Kolmogorov-Smirnov test, P o 0.01) the amplitude distribution of mIPSCs (Fig. 6g). DISCUSSION It has been reported that a short treatment of cultured hippocampal neurons with a large dose of insulin (0.5 mM for 10 min) causes a rapid recruitment of GABAA receptors to postsynaptic domains37. Here we show that treatment of brain slices with a lower concentration of insulin for a longer period of time (0.03–0.1 mM for 42 h) reduces the expression or activity of hypothalamic GABAA receptors through Hap1. The effect of insulin on GABAA receptors may be determined by cell type and dosage. Given the abundance of Hap1 in hypothalamic neurons and its function to stabilize b subunits of the GABAA receptor28, Hap1 probably has an important role in regulating the level of GABAA receptors and their related functions in the hypothalamus. It needs to be pointed out that Hap1 is expressed in other brain regions including the brain stem, which regulate feeding behavior as ADVANCE ONLINE PUBLICATION NATURE MEDICINE © 2006 Nature Publishing Group http://www.nature.com/naturemedicine ARTICLES well. It remains to be investigated whether HAP1 in the extrahypothalamic regions also participates in the control of food intake. GABA provides dominant inhibitory input to the hypothalamus, as 50% of the synapses are GABAergic38. Hap1 is localized to presynaptic terminals13,14,39 and colocalizes with GABAA b2/3 subunits in hypothalamic neurons. Moreover, intrahypothalamic or intracerebroventricular injection of GABA or the GABAA receptor agonist muscimol causes an increase in food intake8–11. Hypothalamic GABAergic activity is increased in obese Zucker rats40, in which hypothalamic insulin signaling is impaired41. Although increased GABAergic activity often elicits fast synaptic transmission, the network connectivity and properties of the intrinsic voltage-gated currents might permit inhibitory interneurons to control the rhythmic output of other cells42,43, thereby stimulating food intake. In addition to the neural circuitry regulated by GABAergic transmission, a variety of molecules in the hypothalamus are involved in regulating food intake1–8. We examined several peptides including insulin receptor, NPY, orexin A and ghrelin but did not find that their levels were regulated by or that they affect Hap1 expression. Accordingly, we propose that elevated insulin in the circulation, by inhibiting the expression of hypothalamic Hap1, decreases the expression of functional GABAA receptors in hypothalamic neurons, which in turn crucially influences hypothalamic activity and feeding behavior (Fig. 6h). The finding that insulin increases the ubiquitination and degradation of Hap1 is similar to the effect of insulin on other proteins through the ubiquitin-proteasome pathway34,35. But the mechanism by which insulin regulates Hap1 expression remains to be investigated. Despite this, our study shows that peripheral signals from insulin can regulate expression of GABAA receptor in the hypothalamus through Hap1 to influence the central control of feeding. This model may help in the development of therapeutic strategies to treat eating disorders associated with abnormal energy homeostasis. METHODS Antibodies. We generated rabbit antibodies to Hap1 (Hap1-A and Hap1-B) in our previous studies13,14,39. We also used the following antibodies: mouse monoclonal antibodies to g-tubulin (Sigma), b2/3 subunits of GABAA receptors (bd17, Chemicon) and antibodies against NPY (22940, ImmunoStar), insulin receptor (sc8080, Santa Cruz) and orexin A (sc8070, Santa Cruz). Animals and feeding studies. We housed male C57BL/6J mice aged 8–12 weeks and Sprague-Dawley rats (275–300 g) aged 6 weeks in a light (12-h dark (18:00–6:00) 12-h light (6:00–18:00) cycle) and temperature (22 1C)-controlled chamber. We fed animals ad libitum (Lab Diet 5001, 12% fat, 28% protein, 60% carbohydrate) or fasted them for 48 h with free access to water. We also fed male mice a diet (Research Diets) containing high carbohydrate (D12450B, 70% carbohydrate, 20% protein, 10% fat) or low carbohydrate (D12492, 20% carbohydrate, 60% fat, 20% protein) for 7 weeks and fed rats for 3 weeks. We generated Hap1 knockout mice in our previous study18. We bred these mice and maintained them in the animal facility under specific pathogen-free conditions in accordance with institutional guidelines. All animal experiments were approved by the Animal Care Committees of Emory University, the Rockefeller University and the Burnham Institute. Immunocytochemistry and western blots. We performed immunostaining and western blotting of brain sections and cultured cells as described previously13,18,39. We used mouse antibody against the influenza hemagglutin epitope (12CA5) and rabbit antibody to Hap1-A for immunofluorescence double labeling. For immunoprecipitation, we precipitated Hap1 in the brains of mouse pups at postnatal days 1 and 2 with antibodies to Hap1. We then subjected the precipitates to western blotting with antibodies to b2/3 subunits of GABAA receptors. We also used antibody to tubulin to probe the same blots in western blot experiments. The methods for in situ hybridization and realtime quantitative RT-PCR are described in Supplementary Methods online. NATURE MEDICINE ADVANCE ONLINE PUBLICATION Preparation of adenoviral Hap1-specific siRNA and HAP1-A. By testing siRNA sequences, we found that one (1695–1713, 5¢-GAAGTATGTCCT CCAGCAA-3¢) was able to effectively inhibit expression of HAP1. This siRNA was inserted into an adenoviral vector that independently expresses GFP under the control of the cytomegalovirus promoter27. The cDNA encoding Hap1-A was also inserted into the same vector to overexpress Hap1-A in the brain. A vector that expresses GFP alone served as the control. Recombinant adenoviruses were generated and purified by Welgen, Inc. We determined the viral titer by measuring the number of infected HEK293 cells expressing GFP. We adjusted all viral stocks to 1 108 plaque-forming units/ml before their use. For injection of adenoviral vectors into the hypothalamus, we anesthetized male C57BL/6J mice aged 8–12 weeks (n ¼ 6–14 per group) with 2.5% avertin. We performed standard surgical procedures to inject adenoviral vectors expressing either Hap1-specific siRNA with GFP or GFP alone using a stereotaxic table (David Kopf Instruments; Supplementary Methods online). Insulin treatment. We injected insulin (10-20 mU/5 ml), leptin (5 mg/ 5 ml), NPY (5 mg/ 5 ml), glucose (100 mg/5 ml or 2% and 110 mM), or 5 ml artificial cerebrospinal fluid (ACSF), which consists of NaCl (120 mM) , KCl (3.3 mM), NaH2PO4 (1.13 mM), NaHCO3 (26 mM), CaCl2 (1.8 mM) and glucose (11 mM), into the third ventricle with the infusion pump over 10 min. After 60 or 120 min following intracerebroventricular administration, we subjected mouse brains to western blotting and immunohistochemistry. We performed the GABA receptor binding experiment using standard methods (Supplementary Methods online). We intraperitoneally injected male C57BL/6J mice aged 8–12 weeks with insulin (1 U/kg). We then isolated brains at 2 h and 4 h after the injection for western blotting and immunohistochemical analyses. We prepared the brain slices (400 mm) containing the hypothalamus of mice and incubated them in ACSF buffer containing insulin (0.03 mM), glucose (5–30 mM) or leptin (0.1 mM) for 2 h. We either fixed the brain slices in 4% paraformaldehydePBS for immunocytochemical study or used them for western blotting. Electrophysiological studies. We obtained coronal hypothalamic slices using standard methods from 9–10-week-old male C57BL/6J mice44,45. We prepared hypothalamic slices (200 mm thick) and incubated them in ACSF solution containing insulin (0.03 mM) and glucose (11 mM) for 3–4 h before recordings. Neurons of the ventromedial hypothalamus were visualized using an upright infrared DIC video microscope with a 40 water-immersion objective (Axioskop, Carl Zeiss). We recorded mIPSCs at 32 1C in the whole-cell recording configuration using a MultiClamp 700A (Molecular Devices) amplifier in the presence of 0.5–1 mM tetrodotoxin, 1.3 mM extracellular Mg2+ and 10 mM CNQX, a selective inhibitor of AMPA/kainate receptor, with and without insulin. All mIPSCs were blocked by 25 mM bicuculline, a competitive antagonist at GABAA receptors. Holding potential was –60 mV. Patch pipettes were filled with a CsCl-based solution and had a resistance of 3–6 MO. Statistical analysis. Statistical significance (P o 0.05) was assessed using the Student t-test. Calculations were performed with SigmaPlot 4.11 and Prism (version 4) software. Note: Supplementary information is available on the Nature Medicine website. ACKNOWLEDGMENTS We thank V. Jaswani for technical assistance and the members of the Li laboratory for comments and critical reading. This work was supported by US National Institutes of Health grants NS36232 and AG19206 (to X.J.L.), NS045016 (to S.H.L.), P01 HD29587 (to S.A.L.), MH43422 to (S.F.L.) and AG00975 (to G.T.). COMPETING INTERESTS STATEMENT The authors declare that they have no competing financial interests. Published online at http://www.nature.com/naturemedicine/ Reprints and permissions information is available online at http://npg.nature.com/ reprintsandpermissions/ 1. Friedman, J.M. & Halaas, J.L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998). 7 © 2006 Nature Publishing Group http://www.nature.com/naturemedicine ARTICLES 2. Saper, C.B., Chou, T.C. & Elmquist, J.K. The need to feed: homeostatic and hedonic control of eating. Neuron 36, 199–211 (2002). 3. Schwartz, M.W. & Porte, D., Jr. Diabetes, obesity, and the brain. Science 307, 375–379 (2005). 4. Benoit, S.C., Clegg, D.J., Seeley, R.J. & Woods, S.C. Insulin and leptin as adiposity signals. Recent Prog. Horm. Res. 59, 267–285 (2004). 5. Kalra, S.P. et al. Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr. Rev. 20, 68–100 (1999). 6. Leibowitz, S.F. & Wortley, K.E. Hypothalamic control of energy balance: different peptides, different functions. Peptides 25, 473–504 (2004). 7. van den Pol, A.N. Weighing the role of hypothalamic feeding neurotransmitters. Neuron 40, 1059–1061 (2003). 8. Olgiati, V.R., Netti, C., Guidobono, F. & Pecile, A. The central GABAergic system and control of food intake under different experimental conditions. Psychopharmacology (Berl.) 68, 163–167 (1980). 9. Tsujii, S. & Bray, G.A. GABA-related feeding control in genetically obese rats. Brain Res. 540, 48–54 (1991). 10. Stratford, T.R. & Kelley, A.E. GABA in the nucleus accumbens shell participates in the central regulation of feeding behavior. J. Neurosci. 17, 4434–4440 (1997). 11. Pu, S. et al. Interactions between neuropeptide Y and gamma-aminobutyric acid in stimulation of feeding: a morphological and pharmacological analysis. Endocrinology 140, 933–940 (1999). 12. Li, X.J. et al. A huntingtin-associated protein enriched in brain with implications for pathology. Nature 378, 398–402 (1995). 13. Li, X.J. et al. Huntingtin-associated protein (HAP1): discrete neuronal localizations in the brain resemble those of neuronal nitric oxide synthase. Proc. Natl. Acad. Sci. USA 93, 4839–4844 (1996). 14. Gutekunst, C.A. et al. The cellular and subcellular localization of huntingtin-associated protein 1 (HAP1): comparison with huntingtin in rat and human. J. Neurosci. 18, 7674–7686 (1998). 15. Page, K.J., Potter, L., Aronni, S., Everitt, B.J. & Dunnett, S.B. The expression of Huntingtin-associated protein (HAP1) mRNA in developing, adult and ageing rat CNS: implications for Huntington’s disease neuropathology. Eur. J. Neurosci. 10, 1835–1845 (1998). 16. Fujinaga, R. et al. Neuroanatomical distribution of Huntingtin-associated protein-1 mRNA in the male mouse brain. J. Comp. Neurol. 478, 88–109 (2004). 17. Chan, E.Y. et al. Targeted disruption of Huntingtin-associated protein-1 (Hap1) results in postnatal death due to depressed feeding behavior. Hum. Mol. Genet. 11, 945–959 (2002). 18. Li, S.H. et al. Lack of huntingtin-associated protein-1 causes neuronal death resembling hypothalamic degeneration in Huntington’s disease. J. Neurosci. 23, 6956–6964 (2003). 19. Dragatsis, I., Zeitlin, S. & Dietrich, P. Huntingtin-associated protein 1 (Hap1) mutant mice bypassing the early postnatal lethality are neuroanatomically normal and fertile but display growth retardation. Hum. Mol. Genet. 13, 3115–3125 (2004). 20. Vonsattel, J.P. et al. Neuropathological classification of Huntington’s disease. J. Neuropathol. Exp. Neurol. 44, 559–577 (1985). 21. Kremer, H.P., Roos, R.A., Dingjan, G., Marani, E. & Bots, G.T. Atrophy of the hypothalamic lateral tuberal nucleus in Huntington’s disease. J. Neuropathol. Exp. Neurol. 49, 371–382 (1990). 22. Sanberg, P.R., Fibiger, H.C. & Mark, R.F. Body weight and dietary factors in Huntington’s disease patients compared with matched controls. Med. J. Aust. 1, 407–409 (1981). 23. Pratley, R.E., Salbe, A.D., Ravussin, E. & Caviness, J.N. Higher sedentary energy expenditure in patients with Huntington’s disease. Ann. Neurol. 47, 64–70 (2000). 8 24. Petersen, A. et al. Orexin loss in Huntington’s disease. Hum. Mol. Genet. 14, 39–47 (2005). 25. Gauthier, L.R. et al. Huntingtin controls neurotrophic support and survival of neurons by enhancing BDNF vesicular transport along microtubules. Cell 118, 127–138 (2004). 26. Li, X.J. & Li, S.H. HAP1 and intracellular trafficking. Trends Pharmacol. Sci. 26, 1–3 (2005). 27. McGuire, J.R., Rong, J., Li, S.H. & Li, X.J. Interaction of Huntingtin-associated protein-1 with kinesin light chain: implications in intracellular trafficking in neurons. J. Biol. Chem. 281, 3552–3559 (2006). 28. Kittler, J.T. et al. Huntingtin-associated protein 1 regulates inhibitory synaptic transmission by modulating gamma-aminobutyric acid type A receptor membrane trafficking. Proc. Natl. Acad. Sci. USA 101, 12736–12741 (2004). 29. MacDonald, P.E. et al. Impaired glucose-stimulated insulin secretion, enhanced intraperitoneal insulin tolerance, and increased b-cell mass in mice lacking the p110gamma isoform of phosphoinositide 3-kinase. Endocrinology 145, 4078–4083 (2004). 30. Chang, G.Q., Karatayev, O., Davydova, Z., Wortley, K. & Leibowitz, S.F. Glucose injection reduces neuropeptide Y and agouti-related protein expression in the arcuate nucleus: a possible physiological role in eating behavior. Brain Res. Mol. Brain Res. 135, 69–80 (2005). 31. Yang, X.J., Kow, L.M., Funabashi, T. & Mobbs, C.V. Hypothalamic glucose sensor: similarities to and differences from pancreatic b-cell mechanisms. Diabetes 48, 1763–1772 (1999). 32. O’Brien, R.M. & Granner, D.K. Regulation of gene expression by insulin. Physiol. Rev. 76, 1109–1161 (1996). 33. Samson, S.L. & Wong, N.C. Role of Sp1 in insulin regulation of gene expression. J. Mol. Endocrinol. 29, 265–279 (2002). 34. Rui, L., Fisher, T.L., Thomas, J. & White, M.F. Regulation of insulin/insulin-like growth factor-1 signaling by proteasome-mediated degradation of insulin receptor substrate-2. J. Biol. Chem. 276, 40362–40367 (2001). 35. Matsuzaki, H., Daitoku, H., Hatta, M., Tanaka, K. & Fukamizu, A. Insulin-induced phosphorylation of FKHR (Foxo1) targets to proteasomal degradation. Proc. Natl. Acad. Sci. USA 100, 11285–11290 (2003). 36. Herring, D. et al. Constitutive GABAA receptor endocytosis is dynamin-mediated and dependent on a dileucine AP2 adaptin-binding motif within the beta 2 subunit of the receptor. J. Biol. Chem. 278, 24046–24052 (2003). 37. Wan, Q. et al. Recruitment of functional GABA(A) receptors to postsynaptic domains by insulin. Nature 388, 686–690 (1997). 38. Decavel, C. & Van den Pol, A.N. GABA: a dominant neurotransmitter in the hypothalamus. J. Comp. Neurol. 302, 1019–1037 (1990). 39. Li, S.H., Li, H., Torre, E.R. & Li, X.J. Expression of huntingtin-associated protein-1 in neuronal cells implicates a role in neuritic growth. Mol. Cell. Neurosci. 16, 168–183 (2000). 40. Coscina, D.V., Castonguay, T.W. & Stern, J.S. Effects of increasing brain GABA on the meal patterns of genetically obese vs. lean Zucker rats. Int. J. Obes. Relat. Metab. Disord. 16, 425–433 (1992). 41. Carvalheira, J.B. et al. Selective impairment of insulin signalling in the hypothalamus of obese Zucker rats. Diabetologia 46, 1629–1640 (2003). 42. Staley, K.J., Soldo, B.L. & Proctor, W.R. Ionic mechanisms of neuronal excitation by inhibitory GABAA receptors. Science 269, 977–981 (1995). 43. McBain, C.J. & Fisahn, A. Interneurons unbound. Nat. Rev. Neurosci. 2, 11–23 (2001). 44. Emmerson, P.J. & Miller, R.J. Pre- and postsynaptic actions of opioid and orphan opioid agonists in the rat arcuate nucleus and ventromedial hypothalamus in vitro. J. Physiol. (Lond.) 517, 431–445 (1999). 45. Vogt, K., Mellor, J., Tong, G. & Nicoll, R. The actions of synaptically released zinc at hippocampal mossy fiber synapses. Neuron 26, 187–196 (2000). ADVANCE ONLINE PUBLICATION NATURE MEDICINE