* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download + Cl
Gas chromatography–mass spectrometry wikipedia , lookup
Crystallization wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Acid–base reaction wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Coordination complex wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Gaseous detection device wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Scanning electrochemical microscopy wikipedia , lookup
Implicit solvation wikipedia , lookup
Metalloprotein wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Electrochemistry wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Electrolysis of water wikipedia , lookup
History of electrochemistry wikipedia , lookup
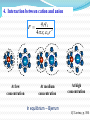
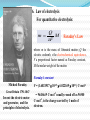
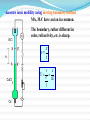
Chapter 7 Electrochemistry A science that studies the relation between electric and chemical phenomena and the disciplines that govern the conversion between electric and chemical energies. Chapter 7 Electrochemistry Main contents • Section 1: Electrolyte and electrolytic solution • Section 2: Electrochemical Thermodynamics: • Section 3: Irreversible electrochemical system • Section 4: Applied electrochemistry §7.1 Electrolyte and electrolytic solution Main contents: 1) Electrolyte: origin of the concept 2) Existence of ions in solution 3) Ion-dipole interaction--Hydration theory 4) Interionic interaction 5) Motion under electric field 6) Conducting mechanism 7) Faraday’s law and its application 1. Origin of the concept – electrolyte 1) Definition of electrolyte An electrolyte is a substance that, when dissolved in solvent, produces a solution that will conduct electricity. Progress of the definition: (1) molten salt; (2) solid-state electrolyte (3) room-temperature ionic liquids (RTIL). 2) Dissociation of substance In 1886, van’t Hoff published his quantitative research on the colligative properties of solution. For sucrose, the osmotic pressure () can be expressed as: =cRT But for some other kind of solvates such as NaCl, the osmotic pressure had to be modified as: =icRT i , van’t Hoff factor, is larger than 1. In the paper written in Achieves Neerlandaises (1885) and Transactions of the Swedish. Academy (1886), van't Hoff showed analogy between gases and dilute solutions. 3) Dissociation theory for weak electrolytes In 1887, Svant A. Arrhenius postulated that, when dissolved in adequate solvent, some substances can split into smaller particles, the process was termed as dissociation. + AB molecule + A+ cation positive ion + B– anion negative ion The charged chemical species are named as ions and the process is termed as ionization. New definitions: Ion, cation, anion; Dissociation, ionization Weak / strong electrolyte? True / potential electrolyte? Theory of Electrolytic Dissociation Acid-base theory Cf. Levine p.295 2. Ions in solution In what state do ions exist in solution? Solvated (hydrated) ion Primary hydration shell ion secondary hydration shell Disordered layer Bulk solution Solvation shells Hydration of ion The water molecules in the hydration sphere and bulk water have different properties which can be distinguished by spectroscopic techniques such as nuclear magnetic resonance (NMR), infrared spectroscopy (IR), and XRD etc. Coordination number: Li+: 4, K+: 6 Primary solvation shell: 4-9, 6 is the most common number Secondary slovation shell: 6-8, for Al3+ and Cr3+: 10-20 3. Hydration Theory / Solvation Theory Why does NaCl only melt at higher temperature, but dissolve in water at room temperature? H / kJ mol-1 Na+(g) + Cl(g) hydration energy: 784 kJ mol-1 788 784 Na+(aq) + Cl(aq) NaCl(s) 4 1948, Robinson and Storks F q1 q 2 4 r 0 r 2 Long-range forces The interionic distance for NaCl crystal is 200 pm, while for 0.1 moldm-3 solution is 2000 pm. To draw Na+ and Cl apart from 200 nm to 2000 nm, the work is: W (/kJ) = 625 / r for melting: r =1, W = 625 kJ, m.p. = 801 oC。 for dissolution in water: r = 78.5, W = 8 kJ. Therefore, NaCl is difficult to melt by easy to dissolve in water at room temperature. 4. Interaction between cation and anion F + At low concentration q1 q 2 4 r 0 r 2 + At medium concentration In equilibrium -- Bjerrum + At high concentration Cf. Levine, p. 304 Owing to the strong interaction, ionic pair forms in concentrated solution. ionic pair vs free ion In an ionic pair, the cation and anion are close to each other, and few or no solvent molecules are between them. Therefore, HCl does not ionize and NaCl does not dissociate completely in solvents. Some facts about strong electrolytes solution present species 0.52 mol·dm-3 KCl 95% K+ + 5% KCl 0.25 mol·dm-3 Na2SO4 76 % Na+ + 24% NaSO4¯ 0.1 mol·dm-3 CuSO4 44% CuSO4 For concentration-dependence of ion pair, see Levine p. 305, Figure 10.10 Degree of association Activity coefficient is essential for quite dilute solutions 5. Conducting mechanism of electrolyte + I + + + + + + + E + •Electric transfer of ion in solution under electric field How can current cross the electrode / solution interface ? Motion of ions in the solution: 1) diffusion: due to difference in concentration 2) convection: due to the difference in density 3) transfer: due to the effect of electric field e e e Cl Cl H+ Cl e H+ Cl H+ Cl Cl Cl e H+ H+ Cl H+ e H+ H+ Cl H+ Cl H+ At anode: At cathode: 2Cl 2e Cl2 2H+ + 2e H2 Conducting mechanism: 1) Transfer of ion in solution under electric field; 2) electrochemical reaction at electrode/solution interface. 6. Law of electrolysis For quantitative electrolysis: Q m M zF Faraday’s Law where m is the mass of liberated matter; Q the electric coulomb, z the electrochemical equivalence, F a proportional factor named as Faraday constant, M the molar weight of the matter. Faraday’s constant Micheal Faraday Great Britain 1791-1867 Invent the electric motor and generator, and the principles of electrolysis. F = (1.6021917 10-19 6.022169 1023 ) C·mol-1 = 96486.69 C·mol-1 usually round off as 96500 C·mol-1, is the charge carried by 1 mole of electron. Current efficiency () Qtheoretical Qeffective meffective mtheoretical Current efficiency is lower than 100% due to side-reactions. For example, evolution of hydrogen occur during electrodeposition of copper. Application of Faraday’s law 1) Definition of ampere: IUPAC: constant current that would deposit 0.0011180 g of silver per second from AgNO3 solution in one second: 1 ampere. 2) Coulometer: copper / silver / gas (H2, O2) coulometer 3) Electrolytic analysis – electroanalysis Q ↔m ↔ n ↔ c 7. Transfer of ion under electric field How do we describe the motion of ions under electric field? 1) Ionic mobility Rate of electric transfer: Ionic velocity dE dl dE U dl Ionic mobility (U) : the ionic velocity per unit electric field, is a constant. measure ionic mobility using moving boundary method MA, MA’ have an ion in common. The boundary, rather different in color, refractivity, etc. is sharp. x v t v x U V tE l 8. Transference number Transference number (transfer/ transport number), is the fraction of the current transported by an ion. plane A I = I+ + I- Q = Q+ + Q- I + Q+ t+ I Q I- I - Qt- I Q I+ tj I Supporting electrolyte? Ij I Qj Q t+ + t- = ? (1) Principle for measuring transference number C A B For time t: Q+ = A U+t c+ Z+ F I+ Q = A Ut c Z F U t Owing to electric migration, on the left side of plane A, there are more anions, while on the right side, more cations. Is this real? (1) Principle of Hittorf method (1853) Example: Electrolysis of HCl solution + =1F + + + + + + + + + + + + + + + + + + bulk solution cathodic region anodic region When 4 Faraday pass through the electrolytic cell + + + + + + + + + + + + + + + + + + 4Cl- -4e- 2Cl2 3 mol H+ 1 mol Cl- 3 mol H+ 1 mol Cl- 4H+ +4e- 2H2 + + + + + + + + + + + + + + + + + + 4Cl- -4e- 2Cl2 final result 3 mol H+ 1 mol Cl- 3 mol H+ 1 mol Cl- 4H+ +4e- 2H2 + + + + + + + + + + + + + + anodic region bulk solution cathodic region For anodic region: cresidual cinitial creacted ctransfered EXAMPLE Cock stopper Pt electrode, FeCl3 solution: In cathode compartment: Initial: FeCl3 4.00 mol·dm-3 Final: FeCl3 3.150 mol·dm-3 FeCl2 1.000 mol·dm-3 Calculate the transference number of Fe3+ Anode chamber Cathode chamber Hittorf’s transference cell What factors will affect the accuracy of the measurement? (2) Principle for the moving-boundary method When Q coulomb passes, the boundary moves x, the cross-sectional area of the tube is A, then: xAcZ+F = Q+ = t+Q Why is the moving-boundary method more accurate that the Hittorf method? Are there any other methods for measuring transfer number? (3) Influential factors (1) Temperature and (2) Concentration Transference number of K+ in KCl solution at different concentration and temperature c /mol·dm-3 0.000 0.005 0.01 0.02 15 0.4928 0.4926 0.4925 0.4924 25 0.4906 0.4903 0.4902 0.4901 35 0.4889 0.4887 0.4886 0.4885 T /℃ (3) Co-existing ions Table transference number on co-existing ions Electrolyte KCl KBr KI KNO3 t+ 0.4902 0.4833 0.4884 0.5084 Electrolyte LiCl NaCl KCl HCl t– 0.6711 0.6080 0.5098 0.1749 Problem: Why does the transference number of certain ion differ a lot in different electrolytes? Outside class reading Ira N. Levine, Physical Chemistry, 5th Ed., McGraw-Hill, 2002. pp. 294-310 Section 10.6 solutions of electrolytes Section 10.9 ionic association pp. 512-515 Section 16.6 electrical conductivity of electrolyte solutions.