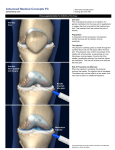
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Arthrocentesis Skills: Shoulder and Wrist
Survey
Document related concepts
Transcript
Apply Evidence Based Medicine to Arthrocentesis Skills: Shoulder and Knee Gregory C. Gardner, MD, FACP Gilliland-Henderson Professor of Medicine University of Washington, Seattle, WA Evidence Based References Evidence based references Courtney and Doherty. Best Practice & Research Clinical Rheumatology 2009; 23:161–192 (2013 update) Crawshaw DP et al. Exercise therapy after corticosteroid injection for moderate to severe shoulder pain. BMJ 2010;340:c3037 doi:10.1136/bmj.c3037 Raynauld JP, et al. Safety and efficacy of long-term intraarticular steroid injections for osteoarthritis of the knee. Arthritis Rheum 2003;48:370-377 Habib GS, et al. Local effectes of intra-articular corticosteroids. Clin Rheumatol 2010;29:347-356 Gardner GC. Teaching arthrocentesis and injection techniques: what is the best way to get our point across. J Rheumatol 2007 vol. 34 (7) pp. 1448-1450 Outline of Workshop Didactics Procedure pearls Anatomy review Clinical Issues and technique review Surface anatomy Present evidence based best practices Current status of how we do and teach arthrocentesis and injection therapy Dangers of “Evidence based Medicine” Analytic rubric Procedure distilled into discrete steps: 1. Patient counseling 2. Patient preparation 1. 5. Able to verbalize anatomy and Discuss reasons for doing the procedure 2. Discuss details of doing the procedure mark appropriate location 6. Patient positioned properly for site of procedure 3. Discuss potential side effects 7. Skin cleansed properly 4. Inquire about potential contra-indications 8. Appropriate application of anesthesia Analytic rubric continued 3. Needle insertion 4. Post-procedure wrap-up 9. Choose appropriate needle 13. Skin re-cleansed if necessary, and syringe for procedure 10. Needle inserted at appropriate angle/depth 11. Needle/syringe stabilized during procedure 12. Aseptic technique site bandaged 14. Post-procedure counseling provided 15. Sharps disposed of properly 16. Operator showed concern for patient comfort Utility of Rubric Framework for teaching arthrocentesis skills Can be used to think about and applying evidenced-based principles Useful as assessment tool for physicians in training Useful as a documentation of procedure Procedure Pearls Textbook complications of arthrocentesis & injection therapy; How much should we worry? Charcot arthropathy – very rare; why would it develop and how often can we safely inject an joint? Periarticular calcifications – 4% give or take may develop Infection – very rare (TBD) Post-injection synovitis – uncommon; 6-12 hrs following procedure and resolves in 48 hrs Tendon rupture – rare; avoid injecting tendon especially high tension tendons i.e. Achilles Skin depigmentation – 5%; may develop 8 wks after injection and resolve by 16 wks Subcutaneous fat atrophy – up to 8%; avoid placing corticosteroid in subcutaneous fat Other – AVN, hyperglycemia, flushing Textbook contraindications to injecting corticosteroids into joints and soft tissue Infection or suspected infection around the joint Hypersensitivity to injectables Avoid injecting through psoriatic plaque Most “hypersensitivity” is to epinephrine; no need to use Upcoming surgery on the joint TBD Suspected intra-articular fracture/joint instability Anticoagulation Caution not contraindication; TBD Joint procedures and infection Seror et al Rheumatology 1999 Retrospective study involving 69 French rheumatologists Mean number of years in practice 20.9 Mean number of corticosteroid injections per year 809 Total injections 1,160,000 in 20 years with 15 instances of post injection infection Overall rate of infection was 1/77,300 When pre-packaged CS syringes used: 1/162,000 When multi-dose vial used: 1/21,000 (7.7 X higher!) 1/4.6 rheumatologists had post-injection septic complication over 20 years How often can we inject a knee? 68 knee OA pts randomized to either saline or 40 mg of triamcinolone acetate injections every 3 months for 2 years Double blind study After 2 years of therapy, no difference in joint space width between groups (figure) Joint pain at night and stiffness significantly better for steroid group using AUC analysis at 2 years Conclusion: JS not affected Clinical improvement especially at 1 year, less apparent at 2 yrs No significant SE Raynauld et al. Arthritis Rheum 2003;48:370 Steroid injection into hip prior to THA Chitre et al. JBJS 2007 99 pts had received steroid/local anesthetic into hip joint 4 to 50 mo prior to THA; mean 18 mo Follow-up 25.8 mo post-surgery with range 9-78 mo NO instances of post-operative joint infection or sepsis occurred Similar data from Sankar et al and Sreekumar et al in the hip and Desai et al in the knee Data from McIntosh et al raise concern about infection if given within 6 weeks of surgery (trend but NS) McIntosh et al. Clin Orthop Relat Res 2006 Best way of cleansing the skin? Current evidence based guidelines for inserting IV catheters suggests the following is best practice according to EPIC 2, a mega- systematic review of best practices for prevention of catheter associated infections Decontaminate the skin site with a single patient use application of alcoholic chlorhexidine gluconate solution (preferably 2% chlorhexidine gluconate in 70% isopropyl alcohol) prior to the insertion of a central venous access device. (Class A) Use a single patient use application of alcoholic povidone-iodine solution for patients with a history of chlorhexidine sensitivity. Allow the antiseptic to dry before inserting the catheter (Class D) Journal of Hospital Infection (2007) 65S, S1–S64 Arthrocentesis and anticoagulation? Ahmed and Gertner Am J Med 2012 Retrospective review of complication in 640 arthrocentesis in 514 anticoagulated patients Compared the incidence of significant bleeding in patients by INR (>2 or < 2) Conclusion: no need to reduce anticoagulation level prior to procedures Complication INR >2 N=456 INR < 2 N=184 Early Bleeding 1 (0.2%) 0 Late Bleeding 0 0 Infection 1 (0.2%) 0 Pain 3 (0.7%) 0 Comparisons between groups NS Patient with bleeding also had pain Should I use anesthesia prior to a procedure? Park et al Rheumatol International 2009 99 patients underwent 2 arthrocenteses One without and one with anesthesia Procedures performed with: Conventional syringe or Reciprocating procedure device Pain measured on 10 cm VAS (0 no pain; 10 unbearable pain) Conventional syringe Reciprocating syringe 49 50 Pain w/o anesthesia 9.22 9.39 NS Pain of anesthesia 6.18 3.96 .006 Pain after anesthesia 4.10 1.84 .003 100% 100% NA # of subjects % Pt wanting anesthesia P Benefit of buffering lidocaine Lidocaine maintained at acidic pH to increase shelf life; pH 6.2 preventing photodegradation/aldehyde formation; lidocaineepinephrine has even lower pH (5.98) Sodium bicarbonate neutralizes pH (7.2) but does not affect onset of action; increases efficacy via increasing uncharged form of drug Buffering: 1 part (1mEq/ml) sodium bicarbonate to 10 parts lidocaine Consistent literature favoring buffering: Vasectomy Bone marrow Bx IV cannula insertion Laceration repair Etc.. Cristoph et al. Ann Emerg Med 1988;17:117-120 Pain scores on VAS following intradermal infiltration of unbuffered and buffered local anesthesia 6 Unbuffered Buffered 5 4 3 2 1 6.21 0 7.22 Lidocaine 5.98 7.16 Lidocaine with Epi Cristoph et al. Ann Emerg Med 1988;17:117-120 6.18 7.20 Mepivacaine pH noted Needles: Primum non nocere or Gardner’s rule 23 “use the smallest needle necessary to get the job done” 25 Gauge 22 Gauge 18 Gauge 18 gauge in articular cartilage John Clark, MD, PhD Dual Injector Three Way Stop Cock Simkin Method New ways of doing procedures Reciprocating Procedure Device Which is he best corticosteroid preparation? Triamcinolone (Aristospan, Kenalog) - easily goes through 26-30 gauge needles, Methylprednisone Acetate (Depomedrol) floculant, may require larger than 30 gauge needle. Betamethasone Acetate (Celestone Soluspan) mixture of short and long acting preparation Dexamethasone Acetate (Decadron-LA) - Long acting, frequently mixed with Decadron for short and long acting combination Only head to head trial of TCA 20 mg vs MPA 40 mg in knee OA - TCA better pain relief, MPA lasted longer Pyne D, et al Clin Rheumatol 2004;23:116-120 In 2 small trials of TCA vs MPA in RA, TCA lasted Can we mix anesthetic and steroids? Dogma about not “mixing” local anesthetic with steroids especially methylprednisolone; 74% of us do mix Percent drug remaining after time noted by HPLC Formula 1 Triamcinolone Bupivicaine Iohexol 4 hours 93% 101 103 24 hours 97 99 101 Formula 2 Triamcinolone Lidocaine Iohexol Preservative parabens may cause steroid to appear flocculant 4 hours 85 101 91 24 hours 85 101 97 Formula 3 Methylprednisolone Bupivicaine Iohexol HPLC study to determine stability of mixed contents for epidural injections 4 hours 88 103 104 24 hours 86 103 98 Formula 4 Methylprednisolone Lidocaine Iohexol 4 hours 101 102 98 Conclusion: mix away! 24 hours 98 99 93 Shat et al. BJ Radiology 2009;82:109-111 Stored at 370 Why do patients with acute onset joint effusions keep their joint at 30-450 of flexion? Boyles Law of course! Boyles Law: Pressure is inversely proportional to volume when temperature is constant in a closed system pV=C www.grc.nasa.gov/WWW/K-12/airplane/aboyle.html Joint position and intra-articular pressure: Maximum joint volume between 30-450 of flexion Hochberg et al. Rheumatology 3rd edition Practice Point: Three causes of severe joint pain There are three causes of joint pain so severe that the patient will guard the joint and protect against movement BUGS BLOOD CRYSTALS BBC Joint Patients hold their joint 300 of flexion as this represents maximum joint volume; flexion or extension results in reduced volume thus increased pressure Slowly developing joint effusions allow time for the capsule to distend and thus do not cause the same degree of pain Convex vs Concave Joint Surfaces Electronic Textbook of Hand Surgery www.eatonhand.com Carpus Direction of Needle Convex Concave Ulna Radius Radius Ulna Making your own polarizing microscope Synovial Fluid Findings Only a small amount of synovial fluid is necessary to make the diagnosis of crystalline forms of arthritis; important to crystal prove at least once Scope and Kit Placing Polarizer and Analyzer Crystals Shoulder Subacromial Region Shoulder Anatomy Osseous structures Scapula Scapular spine Acromium Glenoid Coracoid Clavicle Humerus Shoulder Anatomy Important muscles Important joints Deltoid Rotator Cuff Scapular stabilizers Glenohumeral Acromioclavivular “Scapulothoracic” Other Redundant capsule Subdeltoid bursa Glenohumeral Joint: Circle of Stability Long head of biceps tendon Note pear shaped glenoid that allows humeral head to be depressed by RTC muscles Shoulder Joint Capsule Capsule distended Grays Anatomy Suggested X-rays: Internal rotation External rotation Axillary view External rotation view Mike Richardson, M.D. Axillary view Dx in 101 Patients with Shoulder Pain Over 18 Months in Internal Medicine Clinic Diagnosis Percent Rotator Cuff Disease 62 Myofascial Pain 22 Adhesive Capsulitis AC Joint OA 10 4 Bicepital Tendonitis RA/OA/RSD/PMR 3 1 each Anderson, Kaye. West J Med 1983; 138:268 Impingement Syndrome Impingement is caused by compression of the rotator cuff tendons and subacromial bursa between the greater tubercule of the humerus and the lateral edge of acromion; direct trauma vs impairment of blood supply? 3 stages of impingement syndrome described by Neer Stage 1 - Edema and hemorrhage Stage 2 - Cuff fibrosis, thickening, and partial cuff tearing Stage 3 - Full thickness tendon tears, bony changes, and tendon rupture. Neer CS, 2d. Impingement lesions. Clin Orthop 1983; 173:70 Posterolateral Injection of the Subacromial Space Behind humeral head, under the acromium, directed toward the AC joint 25 gauge, 1 1/2 inch needle Anesthesia with 3-4 cc 1-2% lidocaine useful as diagnostic test Inject with 2 cc bupivicaine & 20-40 mg of depomedrol or triamcinalone Lateral Subacromial Injection Lateral Palpate AC joint Identify acromion 1 cm below acromion; angle under AC joint Subacromial Injection: Lateral vs Posterolateral Note how patient is elevating arm? Injection + exercise vs exercise alone for RTC tendonitis 227 pts randomized to injection + exercise vs exercise alone for impingement syndrome; mean 16 wks of pain 20 mg of triamcinolone plus lignocaine Exercise individualized; attended as many PT sessions as necessary Outcome: 1 & 6 wks pain/disability scores significantly better for injection At 12 & 24 wks NO difference between groups At later assessments, exercise only group taking more pain meds & 32% were injected at some time between 12 and 52 wks Conclusion: Injection therapy provides early pain improvement in impingement syndrome Crawshaw et al BMJ 2010 One-third of exercise only patients go on to injection Do repeated injections lead to RTC tears? 230 patients who had had an MRI scan for impingement symptoms retrospectively evaluated for frequency of RTC tears by number of subacromial steroid injections 128 had 0-2 injections, 102 had 3 or more Exclusions: age > 70, trauma, RA, Diabetes Results: 0-2 injections 3 or more 65 (50.8%) 48 (47%) Partial thickness 12 21 Full thickness 50 24 Massive tear 3 3 RTC tears Conclusion: repeated injections NOT associated with RTC tears Ann R Coll Surg Engl 2009; 91: 414– More full thickness tears in fewer injection group?? Does a corticosteroid injection help in adhesive capsulitis and if so does it matter where we put it? 191 pts with adhesive capsulitis randomized to 4 groups; all received progressive PT 1. 2. 3. 4. Subacromial steroid injection (40 mg TCA) Glenohumeral steroid injection Combination injection using same total steroid dose NSAID At 16 weeks steroid groups significantly less pain and improved motion compared to NSAID only At 24 weeks no difference b/w groups Did not matter where the steroid was placed (US guided) Shin SJ, Lee SY. J Shoulder Elbow Surg 2013;22:521-5 Knee Knee anatomy Important points: Synovial space extends above patella; large target Synovial space posterior to knee joint as well; may explain why patients have posterior pain with an effusion Gray’s Anatomy Menisci Bursae ACL Menisci PCL Infrapatellar Fat Pad Joint Capsule Patella, Lateral Patellar Groove (Femoral Sulcus) Femoral Condyles MultiMedia Group, used with permission Note “keel” of the patella Purpose of patella? Shape of Patella Axial View of Knee Visible Human Lateral Medial Suprapatella Lateral Medial Lateral Medial Lateral Medial Lateral Medial Midpatella Lateral Medial Lateral Medial Reasons to aspirate or inject a knee Aspirate Inject Confirm inflammatory arthritis/crystalline arthritis R/O septic arthritis (knee most affected joint) Relieve pressure in OA/inflammatory arthritis Treat inflammatory arthritis with IA steroids Inject steroids/hyaluronic acid for OA Equipment 21-22 gauge needle for aspiration (18??) 25 gauge to inject only 20-40 mg of triamcinolone or depomedrol Like celestone soluspan for gouty arthritis when available Knee approaches There are at least 4 different routes utilized to enter the knee joint Medial mid-patellar Lateral supra-patellar Anteromedial or anterolateral Lateral mid-patellar Each route has their proponents but it is important to be be familiar with several as some patients will not be easily approached via your most comfortable route Medial knee approach Medial approach is particularly useful when there is a small effusion Identify the superior and inferior poles of the patella Mark a location 1/3-1/2 down from the superior pole and just below the medial margin of the patella Angle needle slightly inferior and a bit superior to miss the patellar keel Make sure you look at the patient’s foot; the needle should be generally perpendicular to the foot Courtney & Doherty: Best Practice & Research Clinical Rheumatology. 2009 Superolateral knee approach This approach is useful for large effusions Mark the area where the superior and lateral margins of the patella meet Angle the needle down and medially into the joint space Courtney & Doherty: Best Practice & Research Clinical Rheumatology. 2009 Anterior approaches for knee injections This approach can be done from either the medial or lateral surface Identify the patellar ligament then the tibial plateau Generally there is a “soft spot” that identifies the area adjacent to the ligament and just above the tibial plateau Angle the needle toward the center of the Patella Medial edge of patellar ligament Tibial plateau Anterior approach “Soft spot” Needle directed toward center of knee Not recommended for aspiration Lateral Midpatellar Approach John Clark MD, PhD Remember MRI scan Suggest inserting needle no further than 1/2 the way down the lateral patellar margin Tissue to traverse is thin laterally and can use 1” needle in normal sized person John Clark is using butterfly needle in this picture Bilateral knee effusions Note loss of medial concavity and bulging suprapatellar pouch Anesthesia with 1-2% lidocaine without epinephrine (what is wrong with this picture?) Note marks on knee Aspiration with 21-22 gauge needle and 20-30 cc syringe Using mosquito clamp to change from aspiration to injection syringe Injecting with 40 mg of triamcinolone or depomedrol +/mixed with small amount of local anesthetic Clean, bandage, bed rest x 24 hr Accuracy of knee injection into non-effused joints by location of injection 240 knee injected for OA done by one orthopaedic surgeon into knees without effusions Three approaches; anteromedial, anterolateral, and lateral mid-patellar Placement of injected material confirmed by fluoroscopy Accuracy: Anteromedial: 60/80 in joint space (75% accuracy) Anterolateral: 57/80 in joint space (71% accuracy) Lateral mid-patellar: 74/80 in joint space (93% accuracy) Jackson et al. JBJS 2002;84-A:1522-27 Aspirate or not aspirate RA knee before steroid injection? 191 swollen RA knees randomized to receive 20 mg of triamcinolone following: Complete aspiration of fluid No aspiration of fluid Followed 6 mo primary end point recurrent knee swelling Groups balanced with regard to gender, medications, prednisone use, ESR etc. Primary end point: Relapses in 23% of aspirated knees Relapses in 47% of non-aspirated knees (p = .001) Medications changed in similar % of pts during study period Weitoft & Uddenfeldt. Ann Rheum Dis 2000; 59:233 Should an RA knee be rested following injection? Old dogma: bed for 2-3 days and then use crutches for 2-3 wks Chakravarty et al Br J Rheum 1994 Randomized 91 Pts with inflammatory knee arthritis to 24 hr of in hospital rest or usual activity after 40 mg of triamcinolone hexacetonide & 2 cc of lignocaine in knees aspirated to dryness VAS pain, knee circumference, 50 foot walk time, CRP followed at 3, 6, 12 and 24 months Results - both improved at 3 wks; RG showed highly significant improvement through 24 wks compared to NRG in all of the study variables; 8 NRG & 2 RG Pts required reinjection Conclusion - 24 hr of bed rest improves clinical outcome for inflammatory arthritis patients (OA pts as well?) Conclusion Make sure our patients are well informed about the procedure process If unsure, review basic anatomy before procedure Be careful but remember that serious adverse events are very rare Evidence based approach will hopefully turn a “wild west” approach to arthrocentesis and injection into a more orderly process!