* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Dr Ziai chronic visual loss_compressed
Survey
Document related concepts
Contact lens wikipedia , lookup
Photoreceptor cell wikipedia , lookup
Retinal waves wikipedia , lookup
Fundus photography wikipedia , lookup
Blast-related ocular trauma wikipedia , lookup
Vision therapy wikipedia , lookup
Eyeglass prescription wikipedia , lookup
Visual impairment wikipedia , lookup
Keratoconus wikipedia , lookup
Cataract surgery wikipedia , lookup
Corneal transplantation wikipedia , lookup
Idiopathic intracranial hypertension wikipedia , lookup
Retinitis pigmentosa wikipedia , lookup
Macular degeneration wikipedia , lookup
Transcript
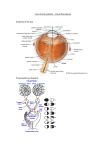
Chronic Visual Disturbance and Visual Loss Setareh Ziai April 2nd, 2009 [email protected] QUICK review Basic Anatomy Where is the problem? LMCC Objectives Pre-retinal: cornea (dystrophy, scarring, edema) lens (age-related, traumatic, steroid-induced) glaucoma Retinal: DM (diabetic retinopathy, macular edema) vascular insufficiency (arterial or venous occlusion) tumours macular degeneration Post-retinal: anterior to optic chiasm (if optic nerve = monocular) • compressive optic neuropathy (intracranial masses, thyroid eye disease) • toxic/nutritional (nutritional deficiencies, alcohol/tobacco amblyopia) optic chiasm lesions (pituitary adenoma) Where is the problem? Pre-retinal: cornea (dystrophy, scarring, edema) lens (age-related, traumatic, steroid-induced) glaucoma Retinal: DM (diabetic retinopathy, macular edema) vascular insufficiency (arterial or venous occlusion) tumours macular degeneration Post-retinal: anterior to optic chiasm (if optic nerve = monocular) • compressive optic neuropathy (intracranial masses, thyroid eye disease) • toxic/nutritional (nutritional deficiencies, alcohol/tobacco amblyopia) optic chiasm lesions (pituitary adenoma) Diagnosis based on: - focused ophthalmological history - - exam: … start with gross examination - - monocular vs. binocular acute vs. chronic painful vs. painless VA slit lamp biomicroscopy +/- fluorescein dilated fundus examination VF testing fluorescein angiography +/- other tests **Remember for exam: sometimes, chronic visual loss in ONE eye is noted incidentally some time later due to occlusion of normal eye…: CHRONIC LOSS OF VISION CAN PRESENT ACUTELY!! Corneal Causes - dystrophy - scarring - edema The Cornea - allows light to enter the eye - provides most of the eye’s optical power - 0.5-0.8 mm thick - transparent due to its uniformity, avascularity and deturgescence Epithelium Stroma Endothelium Corneal Dystrophies - rare inherited disorders - progressive, usually bilateral - can affect any of the three layers of the cornea - affect transparency - age at presentation: first to fourth decades Corneal Dystrophies - divided into: - anterior dystrophies: - epithelium - may present with recurrent corneal erosions - stromal dystrophies: - usually present with visual loss - if very anterior, can cause erosions and pain - posterior dystrophies: - endothelium - vision loss secondary to edema (endothelial dysfx) Corneal Scarring - multiple causes: - trauma - infectious (eg., herpes) - post-surgical Corneal Edema - most often caused by dysfunction of the corneal endothelium: - dystrophy - trauma - infectious (eg., herpes) - post-surgical Corneal Transplantation If the corneal stroma opacifies due to trauma or infection, or if there is swelling or an irregularity of the surface of the cornea, light cannot properly reach the retina. In some cases, a cornea from a deceased donor can be transplanted. Corneal Transplantation Lens-Related Causes (cataract) age-related - traumatic - steroid induced - The Lens - biconvex, avascular, transparent structure - sits inside a thin capsule, attached to the ciliary body by the zonules - provides the remainder of the eye’s optical power (along with the cornea) Lens cataracts are due to the opacification of this normally clear structure Age-Related Cataract - - often affect the nucleus of the lens first: - yellowing, followed by a browning of the lens - eventually, liquefaction causes myopic changes (increased refractive index of the lens) Traumatic Cataract - most common cause of unilateral cataract in young individuals - most often caused by direct penetrating injury to the lens - can also be caused by: - concussion - ionizing radiation to ocular tumours - infrared radiation (glassblowers) Steroid-Induced Cataract - both systemic and topical steroids can be the culprits - posterior part of lens affected first - children may be more susceptible - if lens changes develop, dose should be reduced to the minimum necessary - early opacities may regress with discontinuation of therapy Glaucoma Glaucoma disease of the optic nerve, often caused by an increase in intraocular pressure due to poor drainage of aqueous from the trabecular meshwork… Glaucoma if left untreated, glaucoma can lead to permanent damage to the optic nerve and resultant visual field loss can progress to blindness Glaucoma by definition, glaucoma is a trimodal disease, characterized by: increased IOP optic nerve changes visual field changes Goldmann Applanation Tonometer Glaucoma classification: primary: open-angle, angle-closure secondary: inflammatory, traumatic, neovascular, steroid-induced etc… congenital Risk Factors for Glaucoma age african-american high IOP family history myopia heritage Symptoms of Glaucoma often asymptomatic with late disease, constriction of peripheral, and later central visual field with very high IOP, can have blurry vision and halos around lights Glaucoma: Optic Nerve Changes increased cup:disc ratio thinning of neural rim progressive loss of nerve fiber layer flame hemorrhages on disc Primary Open Angle Glaucoma most common (90%) usually bilateral (can be asymmetric) prevalence increases with age angle is open, eye is quiet increased resistance to aqueous drainage at the level of the trabecular meshwork is thought to be the main pathophysiologic feature Treatment options goal is to stabilize the IOP to protect the optic nerve against further damage options: drops laser surgery Glaucoma - Medications mechanism of action: decrease aqueous production: • beta blockers: timolol • alpha agonists: brimonidine • carbonic anhydrase inhibitors: diamox increase aqueous outflow: • miotics: pilocarpine • epinephrine • prostaglandin analogs: latanoprost Glaucoma - Lasers usually when medical management fails ALT (argon laser trabeculoplasty), SLT (selective laser trabeculoplasty): for open angle glaucomas peripheral iridotomy: for angle-closure glaucomas high success rate Glaucoma - Surgery usually when medical management and laser treatments fail trabeculectomy: sub-conjunctival shunt of aqueous drainage devices (valves) cyclodestruction: last resort – destruction of ciliary body Where is the problem? Pre-retinal: cornea (dystrophy, scarring, edema) lens (age-related, traumatic, steroid-induced) glaucoma Retinal: DM (diabetic retinopathy, macular edema) vascular insufficiency (arterial or venous occlusion) tumours macular degeneration Post-retinal: anterior to optic chiasm (if optic nerve = monocular) • compressive optic neuropathy (intracranial masses, thyroid eye disease) • toxic/nutritional (nutritional deficiencies, alcohol/tobacco amblyopia) optic chiasm lesions (pituitary adenoma) THE RETINA - neural tissue lining the inside of the eye - converts the visual image into a neurochemical message and sends it to the brain - is made up of 10 anatomic layers Diabetes diabetic retinopathy - diabetic macular edema - Diabetic Retinopathy microangiopathy affects pre-capillary arterioles, capillaries and post-capillary venules features of: microvascular occlusion leakage clinically, can be divided into: background DR (nonproliferative) preproliferative DR proliferative DR Diabetic Retinopathy: Epidemiology 239 million people by 2010 doubling in prevalence since 1994 diabetes will affect: 28 million in western Europe 18.9 million in North America 138.2 million in Asia 1.3 million in Australasia • #1 cause of blindness in patients 20-64 yrs • prevalence increases with duration of diabetes and patient age rare to find DR in children < 10 yrs, regardless of duration risk of developing DR increases after puberty Epidemiology Wisconsin Epidemiologic Study of Diabetic Retinopathy Between 1979-1980 1210 patients with Type 1 1780 patients with Type 2 predominantly white population After 20 yrs, DR present in: 99% of Type 1 60% of Type 2 WESDR: Frequency of retinopathy in subjects with type 1diabetes WESDR: Frequency of retinopathy in subjects with type 2 diabetes Diabetic Retinopathy: Risk Factors duration of diabetes: most important risk factor poor metabolic control pregnancy: can be associated with rapid progression HTN nephropathy smoking obesity hyperlipidemia Classification of Diabetic Retinopathy Classified into 2 stages Nonproliferative Diabetic Retinopathy (NPDR) early stage also known as background DR (BDR) further categorized based upon extent of DR mild, moderate, severe, very severe Proliferative Diabetic Retinopathy (PDR) more advanced stage • ***Macular edema • May be present at any stage of DR NPDR typically asymptomatic fluctuating visual acuity: fluctuating blood sugar decreased visual acuity: CSME macular ischemia review these patients annually Mild NPDR Moderate NPDR Severe NPDR Proliferative Diabetic Retinopathy more likely to become symptomatic than early NPDR may have decreased vision, sudden vision loss, floaters, cobwebs, flashes, dull eye ache PDR can also affect visual function by affecting the macula with resulting macular ischemia and/or edema Proliferative DR affects 5-10% of the diabetic population neovascularization is the hallmark NVD: neovascularization of the disc NVE: neovascularization elsewhere new vessels are not only extremely fragile (intraretinal or vitreous hemorrhage), but often associated with fibrous proliferation, leading to an increased risk of tractional retinal detachment Advanced PDR Tractional retinal detachment resulting from contraction of the fibrovascular proliferative tissue on the retina Panretinal Photocoagulation for High-risk PDR goal is to induce involution (or at least arrest) of new vessels by creating areas of retinal ischemia 1200-3000 burns 4 sessions Vitrectomy for Vitreous Hemorrhage / TRD Diabetic Macular Edema (DME) retinal edema threatening or involving the macula diagnosis is made by slit-lamp exam, confirmed by fluorescein angiography and/or OCT important observations include: location of retinal thickening relative to the fovea presence and location of exudates DME and CSME Treatment of CSME argon laser application intravitreal steroid injection intravitreal anti-VEGF injection pars plana vitrectomy Ophthalmological Follow-Up Diabetic Screening Type 1 diabetics: Dilated funduscopic exam (DFE) 5 yrs after diagnosis Newly diagnosed patients with Type 1 diabetes rarely have retinopathy during the first 5 yrs Type 2 diabetics: Type 2 diabetics typically diagnosed yrs after initial onset DFE at the time of diagnosis Significant portion of newly diagnosed Type 2 diabetics have established DR at the time of diagnosis Vascular Insufficiency arterial occlusions (CRAO, BRAO) - venous occlusions (CRVO, BRVO) - CRAO CRAO most of the retina is supplied by the central retinal artery (branch of the ophthalmic artery, which is the first branch of the ICA) if this supply is interrupted (embolus, thrombosis, inflammation, vasculitis or compression), the retina becomes ischemic irreversible damage occurs after approximately 90 minutes CRAO presentation is with sudden and profound loss of vision RAPD is present orange reflex from the choroid stands out at the fovea, and contrasts with the surrounding pale retina (cherry-red spot) must r/o temporal arteritis CRAO most commonly the result of atherosclerosis (thrombosis) but may also be caused by calcific emboli often in older patients, with a hx of arteriosclerosis may have had a hx of amaurosis fugax (transient visual loss) CRAO OPHTHALMOLOGIC EMERGENCY!! treatment: decrease IOP paracentesis ocular massage goal: to send the embolus distally **remember to r/o giant cell arteritis! (ESR, CRP, plt) poor prognosis: 60% 20/400 BRAO BRAO sudden and profound altitudinal or sectoral visual field loss similar causes as CRAO identify and treat associated medical conditions (HTN, DM, hypercholesterolemia, smoking, vasculitis etc…) BRAO retinal cloudiness in ischemic area +/- visible embolus also has a poor prognosis, unless the obstruction can be dislodged within a few hours CRVO CRVO thrombosis of the central retinal vein sudden loss of vision in affected eye severity of symptoms varies… non-ischemic: 75% Ischemic most characteristic finding: retinal hemorrhages CRVO underlying associations advancing age systemic conditions: HTN, DM, smoking, obesity, hyperlipidemia glaucoma inflammatory diseases: sarcoidosis, Behcet disease thrombophilic disorders: hyperhomocysteinaemia, antiphospholipid antibody syndrome CRVO Treatment: treat associated medical conditions decrease IOP if elevated pan-retinal photocoagulation (laser) if: • neovascularization (iris, angle, retina) • …especially if ischemic CRVO BRVO BRVO thrombosis of a branch of the central retinal vein visual loss depends on the amount of macular drainage compromised by the occlusion (peripheral occlusions may be asymptomatic) characteristic findings in one sector of the retina: dilatation and tortuosity of veins retinal hemorrhages retinal/macular edema BRVO obstruction often at arterio-venous crossings: arteries and veins share adventitial sheath… thickening of the arteriole (arteriosclerosis) compresses the vein, eventually causing an occlusion often associated with: hypertension (75%) diabetes (10%) BRVO prognosis: depends on amt of venous drainage involved by the occlusion and severity of macular ischemia: within 6 mos, about 50% of eyes have a VA of 20/30 or better main complications: chronic macular edema neovascularization laser photocoagulation may be helpful in above cases Retinal Tumours ocular tumours: ciliary body: • melanoma choroid: • melanoma • hemangioma • metastases primary ocular lymphoma retina and optic nerve: • retinoblastoma • astrocytoma • hemangioma Choroidal Melanoma most common primary intraocular tumour in adults presentation usually in 6th decade: asymptomatic vs. visual field defect and/or decreased visual acuity signs: raised, usually pigmented lesion visible at the back of the eye may be associated with retinal detachment optic nerve may be involved Choroidal Melanoma treatment: consider size, location, activity of tumour, state of fellow eye, general health/age of pt, pt’s wishes/fears • brachytherapy • external radiotherapy • transpupillary thermotherapy • local resection • enucleation • exenteration • palliative (may include chemo) Choroidal Metastases …with choroidal melanoma, don’t forget general medical investigations! mets TO the choroid: • most frequently from bronchus in both sexes and the breast in women, rarely kidney or GI CXR, rectal exam, mammography mets FROM the choroid: • liver hepatic u/s, GGT, ALP • lungs (rarely affected before liver) CXR Choroidal Metastases usually present with visual impairment only IF tumour is near the macula signs: fast-growing, creamy coloured lesion most often in posterior pole usually not very elevated (infiltrates laterally) Choroidal Metastases treatment: observe: if asxic or receiving systemic chemo radiation: external beam or brachy transpupillary thermotherapy systemic therapy for the primary enucleation: for painful blind eye prognosis is poor… median survival: 8-12 mos for all pts, 15-17 mos for those with breast ca Retinoblastoma most common malignant tumour of the eye in childhood (1:20 000) mean age of presentation: 8 mos if inherited, 25 mos if sporadic 60% present with leukocoria (white pupillary reflex) strabismus (20%) occasionally: painful, red eye if inherited: often bilateral Retinoblastoma malignant transformation of primitive retinal cells before their final differentiation can be caused by germinal mutations (can be passed on to the next generation), or can be sporadic (66% of cases) Retinoblastoma this is a clinical diagnosis, but CSF and bone marrow should be examined to check for metastatic disease if ON involved or if there is evidence of extraocular extension rx: small: cryotherapy, photocoagulation medium: brachytherapy, external beam, chemo large/advanced cases: chemoreduction + local treatment, enucleation metastatic disease: chemo (intrathecal if cells in CSF) Retinoblastoma prognosis: depends on extent of disease at diagnosis overall mortality ~ 5-15% ~ 50% of children with the germinal mutation will eventually develop a second primary tumour (eg., osteosarcoma of the femur or pinealoblastoma) Macular Degeneration Macula 1.5 mm in diameter central vision: BEST VISUAL ACUITY colour vision progressive destruction of the macular area: MACULAR DEGENERATION Macular Degeneration most common cause of irreversible visual loss in the developed world exists in two forms: non-exudative (dry) macular degeneration exudative (wet) macular degeneration Non-exudative Macular Degeneration lipid products arising from photoreceptor outer segments are found under retina can be seen with ophthalmoscope! called « drusen » Exudative Macular Degeneration new vessels from the choroid grow into the sub-retinal space; form a subretinal neovascular membrane subsequent hemorrhage into the subretinal space or even through the retina into the vitreous is associated with profound loss of vision Macular Degeneration symptoms: since fovea is responsible for fine visual resolution, any disruption will cause severe visual impairment • blurry/reduced vision • distorted vision (metamorphopsia) • reduction (micropsia) or enlargement (macropsia) of objects • VF loss (scotoma) Macular Degeneration rx: non-exudative (usually slowly progressive): • no actual medical treatment • use low vision aids • high dose antioxidants MAY be beneficial (eg., vitalux) Macular Degeneration rx: exudative (can be rapidly progressive and devastating): • intravitreal injections of anti-VEGF factors: bevacizumab, ranibizumab • photodynamic therapy (injection of photosensitizer into systemic circulation followed immediately by laser targeting new vessels in macular area) • combination of above treatments Where is the problem? Pre-retinal: cornea (dystrophy, scarring, edema) lens (age-related, traumatic, steroid-induced) glaucoma Retinal: DM (diabetic retinopathy, macular edema) vascular insufficiency (arterial or venous occlusion) tumours macular degeneration Post-retinal: anterior to optic chiasm (if optic nerve = monocular) • compressive optic neuropathy (intracranial masses, thyroid eye disease) • toxic/nutritional (nutritional deficiencies, alcohol/tobacco amblyopia) optic chiasm lesions (pituitary adenoma) OPTIC NERVE 1.2 million cells 80 % visual fibres 20 % pupillary fibres carries visual information from the eye to the brain OPTIC CHIASM crossover above of nasal fibers the pituitary internal carotids are just lateral from optic chiasm: optic tract to the lateral geniculate body optic radiation to the primary visual cortex Anterior to Optic Chiasm compressive optic neuropathies - toxic/nutritional optic neuropathies - Compressive Optic Neuropathies INTRACRANIAL MASSES: optic nerve glioma • typically affects young women, end of first decade • associated with NF-1 optic nerve sheath meningioma • most frequent in middle-aged women • unilateral, gradual visual impairment any other orbital or chiasmal tumour compressing any part of the optic nerve THYROID EYE DISEASE Thyroid Eye Disease may occur in the absence of biochemical evidence of thyroid dysfx autoimmune reaction (IgG Abs) causing: inflammation of EOMs: pleiomorphic cellular infiltration associated with increased secretion of GAGs and osmotic imbibition of water • muscles can become up to 8 times their original size!! no relation to severity of thyroid dysfx! Thyroid Eye Disease main findings: (not all are always present!) soft tissue involvement lid retraction proptosis optic neuropathy restrictive myopathy Thyroid Eye Disease vision loss from: exposure keratopathy • due to severe proptosis resulting in incomplete lid closure → chronically exposed cornea → corneal ulceration & exposure keratopathy optic neuropathy • affects 5% of pts • compression of ON or its blood supply by congested (enlarged) EOMs • can lead to severe, permanent visual impairment • rx with steroids, surgery if needed Toxic/Nutritional Optic Neuropathies nutritional deficiencies alcohol-tobacco amblyopia Nutritional Deficiencies pts with extremely poor diets, often in association with alcohol-tobacco amblyopia usually due to B12 deficiency in combination with cyanide toxicity symmetrical VF loss if early, can be treated with high-dose vitamins and restoration of « well-balanced diet » eventually leads to optic atrophy and permanent vision loss Alcohol-Tobacco Amblyopia affects heavy drinkers, cigar and pipe smokers: deficient in protein and the B vitamins symptoms: insidious, bilateral, progressive visual impairment + dyschromatopsia signs: symmetrical VF defect, may have pale (or normal) discs rx: 1000 units of hydroxocobalamin qweekly X 10 wks + multivitamins + « well-balanced diet » px: good in early cases if comply with rx advanced cases: optic atrophy and permanent visual loss Optic Chiasm Lesions - pituitary adenoma Pituitary Adenoma presentation usually in early adult life or middle age symptoms: h/a visual symptoms: very gradual onset (often not noticed by pt until very well-established) • VF defect: usually, bitemporal hemianopia, worst in the superior field, and extending inferiorly • colour desaturation across vertical midline • optic atrophy: in 50% of cases with field defects caused by pituitary lesions Pituitary Adenoma investigations: MRI: coronal, axial and sagittal sections before and after gadolinium injection CT: demonstrates enlargement or erosion of the sella endocrinological investigation: PRL, FSH, TSH, GH Pituitary Adenoma treatment options: observation medical: dopamine agonists (bromocriptine) surgery radiotherapy: often used as an adjunct gamma knife stereotactic radiotherapy Visual Field Defects Merci