* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Energy
Dark energy wikipedia , lookup
Efficient energy use wikipedia , lookup
Open energy system models wikipedia , lookup
William Flynn Martin wikipedia , lookup
Energy storage wikipedia , lookup
100% renewable energy wikipedia , lookup
Energy subsidies wikipedia , lookup
Potential energy wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Zero-energy building wikipedia , lookup
World energy consumption wikipedia , lookup
Kinetic energy wikipedia , lookup
Low-carbon economy wikipedia , lookup
Regenerative brake wikipedia , lookup
Gibbs free energy wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Alternative energy wikipedia , lookup
Energy policy of Australia wikipedia , lookup
International Energy Agency wikipedia , lookup
Distributed generation wikipedia , lookup
Energy harvesting wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Internal energy wikipedia , lookup
Energy policy of the United Kingdom wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Negawatt power wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Conservation of energy wikipedia , lookup
United States energy law wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
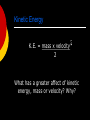
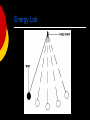
SWBAT to explain why fossil fuels are important as an energy source List as many forms of energy as you can Energy: Forms and Changes Overview Nature of energy Kinetic vs. potential energy intro States of energy Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical Nature of Energy Energy is all around you! You can hear energy as sound. You can see energy as light. And you can feel it as wind. Nature of Energy You use energy when you: hit a softball. lift your book bag. compress a spring. Nature of Energy Living organisms need energy for growth and movement. Nature of Energy What is energy that it can be involved in so many different activities? Nature of Energy What is energy that it can be involved in so many different activities? Energy: the ability to do work. Work: when you exert a force Nature of Energy What is energy that it can be involved in so many different activities? Energy: the ability to do work. Work: when you exert a force If an object or organism does work (exerts a force over a distance to move an object) the object or organism uses energy. Nature of Energy Because of the direct connection between energy and work, energy is measured in the same unit as work: joules (J). Nature of Energy Because of the direct connection between energy and work, energy is measured in the same unit as work: joules (J). Objects use energy to do work and gain energy when work is done on them Energy: the ability to do work Energy is all around us- it can be seen, felt, and heard Two types of energy: kinetic and potential Measured in joules Energy: the ability to do work We know that energy is flowing freely all around us. Are there any places where energy cannot flow freely? Energy: the ability to do work Black holes are regions of space where gravity is so strong that matter and light energy can't escape. Black Holes Black Holes Black Holes Black Holes Black Holes Black Holes Black Holes http://www.youtube.com/watch?v= zTXW9aRO23Y Overview Nature of energy Kinetic vs. potential energy intro States of energy Energy conversions within the body Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical States of Energy The most common energy conversion is the conversion between potential and kinetic energy. All forms of energy can be in either of two states: Potential Kinetic Kinetic vs. Potential Energy Intro All energy is divided into kinetic or potential energy Kinetic energy: the energy of motion Potential energy: the energy of position (stored energy) Kinetic Energy K.E. = mass x velocity 2 What has a greater affect of kinetic energy, mass or velocity? Why? Kinetic energy formula: KE=1/2 x m x v2 M= mass v= velocity (speed) A 3 kg ball is rolling 2 m/s. How much kinetic energy does it have? Kinetic Energy The energy of motion is called kinetic energy. The faster an object moves, the more kinetic energy it has. The greater the mass of a moving object, the more kinetic energy it has. Kinetic energy depends on both mass and velocity. Potential Energy Potential Energy is stored energy. Stored chemically in fuel, the nucleus of atom, and in foods. Or stored because of the work done on it: Stretching a rubber band. Winding a watch. Pulling back on a bow’s arrow. Lifting a brick high in the air. Gravitational Potential Energy Potential energy that is dependent on height is called gravitational potential energy. Gravitational Potential Energy A waterfall, a suspension bridge, and a falling snowflake all have gravitational potential energy. Gravitational Potential Energy If you stand on a 3-meter diving board, you have 3 times the G.P.E, than you had on a 1-meter diving board. Gravitational Potential Energy “The bigger they are the harder they fall” is not just a saying. It’s true. Objects with more mass have greater G.P.E. The formula to find G.P.E. is G.P.E. = Weight X Height. Kinetic-Potential Energy Conversions Roller coasters work because of the energy that is built into the system. Initially, the cars are pulled mechanically up the tallest hill, giving them a great deal of potential energy. From that point, the conversion between potential and kinetic energy powers the cars throughout the entire ride. Kinetic-Potential Energy Conversions Where is KE and PE maximum and minimum? Kinetic-Potential Energy Conversions As a basketball player throws the ball into the air, various energy conversions take place. Ball slows down Ball speeds up Energy Lab Today we are going to look at how a pendulum demonstrates how kinetic and potential energy are interconverted Energy Lab Today we are going to look at how a pendulum demonstrates how kinetic and potential energy are interconverted A pendulum is a weight hung from a fixed point so that it can swing freely backward and forward Energy Lab Energy Lab When the pendulum is pulled back to maximum height or at its highest point, there is only potential energy. Energy Lab When the pendulum is pulled back to maximum height or at its highest point, there is only potential energy. Once the pendulum is released that potential energy begins changing to kinetic energy Energy Lab When the pendulum is pulled back to maximum height or at its highest point, there is only potential energy. Once the pendulum is released that potential energy begins changing to kinetic energy At the very bottom or lowest point of the pendulum, the energy is all kinetic. Energy Lab When the pendulum is pulled back to maximum height or at its highest point, there is only potential energy. Once the pendulum is released that potential energy begins changing to kinetic energy At the very bottom or lowest point of the pendulum, the energy is all kinetic. As the pendulum then rises, that kinetic begins to be converted into potential energy. Overview Nature of energy Kinetic vs. potential energy intro States of energy Energy conversions within the body Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical Overview Nature of energy Kinetic vs. potential energy intro States of energy Energy conversions within the body Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical Kinetic-Potential Energy Conversions in the Human Body KWL Chart Forms of Energy (ATP Production) (BIO) www.youtube.com/watch?v=c_adi3nrg 5Y Kinetic-Potential Energy Conversions in the Human Body KWL Chart K= what you already know Forms of Energy (ATP Production) (BIO) www.youtube.com/watch?v=c_adi3nrg 5Y Kinetic-Potential Energy Conversions in the Human Body KWL Chart K= what you already know W= what you want to know Forms of Energy (ATP Production) (BIO) www.youtube.com/watch?v=c_adi3nrg 5Y Kinetic-Potential Energy Conversions in the Human Body KWL Chart K= what you already know W= what you want to know L= what you learned Forms of Energy (ATP Production) (BIO) www.youtube.com/watch?v=c_adi3nrg 5Y https://www.youtube.com/watch?v=00 jbG_cfGuQ Kinetic-Potential Energy Conversions in the Human Body K: What do we already know about energy use in the body? Kinetic-Potential Energy Conversions in the Human Body K: What do we already know about energy use in the body? Energy comes in two forms: kinetic (energy of movement) and potential (stored energy) Kinetic-Potential Energy Conversions in the Human Body K: What do we already know about energy use in the body? Energy comes in two forms: kinetic (energy of movement) and potential (stored energy) ATP is a molecule used to store energy in the body Kinetic-Potential Energy Conversions in the Human Body K: What do we already know about energy use in the body? Energy comes in two forms: kinetic (energy of movement) and potential (stored energy) ATP is a molecule used to store energy in the body Energy cannot be created or destroyed Kinetic-Potential Energy Conversions in the Human Body W: What do we want to know about energy in the body? Where is potential energy found in the body? Where is kinetic energy found in the body? How is energy from food transferred to potential and kinetic energy in the body? How is energy balanced in the body? Kinetic-Potential Energy Conversions in the Human Body W: Where is potential energy found in the body? Kinetic-Potential Energy Conversions in the Human Body W: Where is potential energy found in the body? Potential energy in the body is stored in the chemical bonds of ATP. When the bonds in ATP are broken, that energy is released. Kinetic-Potential Energy Conversions in the Human Body W: Where is kinetic energy found in the body? Kinetic-Potential Energy Conversions in the Human Body W: Where is kinetic energy found in the body? Kinetic energy is the energy that is released from ATP. It is used for day to day activities like: Cellular and molecular transport Mechanical work (muscle contraction) Production of other high energy molecules (tissues, proteins, etc.) Kinetic-Potential Energy Conversions in the Human Body W: How is energy from food transferred to potential and kinetic energy in the body? Kinetic-Potential Energy Conversions in the Human Body W: How is energy from food transferred to potential and kinetic energy in the body? Catabolic processes of the body break food down to release the energy from food molecules so it can be stored in the form of ATP Anabolic processes of the body: use energy stored in ATP to perform day to day activities Kinetic-Potential Energy Conversions in the Human Body W: How is energy balanced in the body? Kinetic-Potential Energy Conversions in the Human Body W: How is energy balanced in the body? Energy that comes in through food has two options: it can be converted to chemical energy (stored as ATP) or it can be used for kinetic energy (day to day activities) Kinetic-Potential Energy Conversions in the Human Body W: How much energy is released when one ATP molecule is converted to ADP + P? How much ATP is produced per hour on a 2,000 Calorie diet? How much ATP does a person have in their body at any given moment? Kinetic-Potential Energy Conversions in the Human Body W: How much energy is released when one ATP molecule is converted to ADP + P? How much ATP is produced per hour on a 2,000 Calorie diet? 30.5 kJ 4.4 kg/hr (105 kg/ day) How much ATP does a person have in their body at any given moment? 50-200g (ATP IS RECYCLED HEAVILY!) Kinetic-Potential Energy Conversions in the Human Body L: List 7 bullet points. Turn this in before you leave! Intro Label the following as potential or kinetic. Electricity Nuclear energy Chemical energy Heat energy Light energy Mechanical energy Overview Nature of energy Kinetic vs. potential energy intro States of energy Energy conversions within the body Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical Forms of Energy Five main forms of energy are: Heat Chemical Electromagnetic Nuclear Mechanical Heat Energy Heat is a form of energy that is transferred by a difference in temperature Heat is caused by the internal motion of atoms Heat energy can be produced by friction. Heat energy causes changes in temperature and phase of any form of matter. Chemical Energy Chemical energy is what forms and breaks chemical bonds Chemical energy is required to bond atoms together. When bonds are broken, energy is released. Chemical Energy Fuel and food are forms of stored chemical energy. Electromagnetic Energy… …is a form of energy emitted and absorbed by charged particles that exhibits wave-like behavior as it travels through space Power lines carry electromagnetic energy into your home in the form of electricity. Electromagnetic Energy Light is a form of electromagnetic energy. Each color of light (Roy G Biv) represents a different amount of electromagnetic energy. Electromagnetic Energy is also carried by X-rays, radio waves, and laser light. Nuclear Energy The nucleus of an atom is the source of nuclear energy. Nuclear Energy When the nucleus splits (fission), nuclear energy is released in the form of heat energy and light energy. Nuclear energy is also released when nuclei collide at high speeds and join (fuse). Nuclear Energy The sun’s energy is produced from a nuclear fusion reaction in which hydrogen nuclei fuse to form helium nuclei. Nuclear Energy Nuclear energy is the most concentrated form of energy. Mechanical Energy The energy possessed by an object due to its motion or its position When work is done to an object, it acquires energy. The energy it acquires is known as mechanical energy. Mechanical Energy When you throw a balling ball, you give it energy. When that bowling ball hits the pins, some of the energy is transferred to the pins (transfer of momentum). Exit Survey: Classify as Kinetic or Potential Heat energy Electromagnetic energy (light) Mechanical energy Chemical energy Nuclear energy Gravitational energy Kinetic vs. Potential Energy Kinetic energy: the energy of motion Heat energy Electromagnetic energy (light) Mechanical energy Potential energy: the energy of position (stored energy) Chemical energy Nuclear energy Gravitational energy Overview Nature of energy Kinetic vs. potential energy intro States of energy Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical Energy conversions Laws of energy Energy Conversion Energy can be changed from one form to another. Changes in the form of energy are called energy conversions. Energy conversions All forms of energy can be converted into other forms. The sun’s energy through solar cells can be converted directly into electricity. Green plants convert the sun’s energy (electromagnetic) into starches and sugars (chemical energy). Other energy conversions In an electric motor, electromagnetic energy is converted to mechanical energy. In a battery, chemical energy is converted into electromagnetic energy. The mechanical energy of a waterfall is converted to electrical energy in a generator. Energy Conversions In an automobile engine, fuel is burned to convert chemical energy into heat energy. The heat energy is then changed into mechanical energy. Chemical --> Heat --> Mechanical The Law of Conservation of Energy Energy can be neither created nor destroyed by ordinary means. It can only be converted from one form to another. If energy seems to disappear, then scientists look for it – leading to many important discoveries. Overview Nature of energy Kinetic vs. potential energy intro States of energy Energy conversions within the body Forms of energy Heat Chemical Electromagnetic Nuclear Mechanical