* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download EnergyBasicsand1stLaw05
Efficient energy use wikipedia , lookup
Dark energy wikipedia , lookup
Kinetic energy wikipedia , lookup
William Flynn Martin wikipedia , lookup
Open energy system models wikipedia , lookup
Energy subsidies wikipedia , lookup
100% renewable energy wikipedia , lookup
Energy storage wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Regenerative brake wikipedia , lookup
Zero-energy building wikipedia , lookup
World energy consumption wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Low-carbon economy wikipedia , lookup
Alternative energy wikipedia , lookup
Energy policy of Australia wikipedia , lookup
International Energy Agency wikipedia , lookup
Internal energy wikipedia , lookup
Distributed generation wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Energy policy of the United Kingdom wikipedia , lookup
Energy harvesting wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Conservation of energy wikipedia , lookup
Negawatt power wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
United States energy law wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
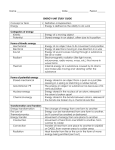
Energy Basics and the Laws of Thermodynamics Part 1: Energy Basics 1. Define energy and matter? • 2. Matter is stuff. • Energy is the capacity to take action and move matter around. How are energy and matter related? • • Energy moves matter. Energy is stored in matter. 3. Restate the Law of Conservation of Matter? • Matter cannot be created or destroyed. 4. What is the First Law of Law Thermodynamics (the Law of Conservation of Energy)? • Energy cannot be created or destroyed, but it can be transformed from one form into another. Part 2: Various Forms of Energy Mechanical Energy: The energy of movement or position. PE/KE Potential Energy: Stored energy that is ready to be used due to an objects position or condition. For example, the energy stored in a spring, the stored energy of a rock on a ledge, the energy stored in the chemical bonds of two particles, the energy stored in the nucleus of an atom, and energy stored in a conductor of electricity. Kinetic Energy: The energy of motion. The pattern of movement can be classified as random, wave, linear or rotational. For example, the energy of a thrown baseball, the energy carried by vibrating particles like sound. Heat/Thermal Energy: The energy of moving particles that can change the temperature of something. For example, the heat you feel when you rub your hands together. KE Light/Radiant Energy: The energy from electromagnetic waves. For example, light waves, solar radiation, radio waves, infrared radiation (lasers), microwaves, and x-rays. KE Electrical Energy: This is the energy of electrons stored in or flowing through a conductor in a controlled motion. The flow of electrons is conducted through wires made of materials whose properties make this type of motion possible. For example, the energy transferred through electrical wires made of copper. KE Chemical Energy: The energy stored in molecular bonds. This is the energy found trapped between the atoms that make up a given molecule. For example, photosynthesis - where the radiant energy from the sun is used to combine water (H2O) and carbon dioxide (CO2) into glucose sugar (C6H12O6) - most of the energy used is trapped within the bonds of the newly formed glucose. PE Nuclear Energy: The energy stored in the nuclei of atoms. The energy can be released by: 1) radioactive decay: the spontaneous decay of large nuclei to smaller ones, which releases energy in the process; 2) fission: the purposeful breaking down of nuclei into smaller ones; 3) fusion: the process of small nuclei fusing together to make bigger ones. PE Part 2: Various Forms of Energy Mechanical Energy The total energy an object has due to position, movement, or both Potential Energy Kinetic Energy Energy that is stored and ready to be used, due to an object’s position or condition. Energy that is released due to an objects movement or motion Examples: Examples: - Chemical - Electrostatic - Sound (wave) - Nuclear - Magnetic - Heat/Thermal (random) - Gravitational - Elastic - Electrical - Light/Radiant (wave) - Linear/Rotational Movement PE and KE are related b/c potential energy can be transformed into kinetic energy (and vice versa). Part 3: Energy Transformations: First Law of Thermodyn. Device Initial Form of Energy chemical Converted Form of Energy electricity heat light nuclear Heat, electricity Energy Transformation Demonstrations Energy cannot be created or destroyed. But, it can be transformed from one form to another. OBSERVE THE DEMONSTRATIONS: - Diagram the apparatus - Label the forms (and transformations) of energy - THE WAY THINGS GO