* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter02a
100% renewable energy wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Cogeneration wikipedia , lookup
World energy consumption wikipedia , lookup
Regenerative brake wikipedia , lookup
Kinetic energy wikipedia , lookup
Energy storage wikipedia , lookup
International Energy Agency wikipedia , lookup
Zero-energy building wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Low-carbon economy wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
Compressed air energy storage wikipedia , lookup
Alternative energy wikipedia , lookup
Negawatt power wikipedia , lookup
Gibbs free energy wikipedia , lookup
Distributed generation wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Solar air conditioning wikipedia , lookup
Environmental impact of electricity generation wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
Internal energy wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Micro combined heat and power wikipedia , lookup
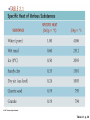
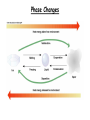
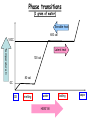
Energy, Temperature, Heat Recap • The atmosphere may be divided into layers (or regions) according to its • • ♦ vertical profile of temperature; ♦ gaseous composition; ♦ electrical properties. We live at the bottom of the troposphere, which is an atmospheric layer where the air temperature normally decreases with height, and is a region that contains all of the weather we are familiar with. The study of the atmosphere and all of its related phenomena is called meteorology, a term whose origin dates back to the days of Aristotle. End of the chapter study material • Key terms: atmosphere, nitrogen, oxygen, water vapor, carbon dioxide, ozone, ozone hole, aerosol, pollutants and acid rain • • • • (Chapter 18), outgassing, density, pressure, air pressure, lapse rate (6), temperature inversion (3), troposphere, radiosonde, stratosphere, tropopause, mesosphere, thermosphere, exosphere, homosphere, heterosphere, ionosphere, weather, climate, meteorology, middle latitudes and middle latitude cyclonic storm (12), hurricane (15), thunderstorms and tornadoes (14), wind (8), wind direction (9), front (11) ♦ Must learn now ♦ Will discuss later (in Chapter xyz) Questions for review Questions for thought Problems and exercises Questions for exploration • Energy: • Energy and Work ♦ Definition: The ability or the capacity to do work on some form of matter ♦ Units: J (Joule), cal, erg Work: ♦ Work is done when a body with a non-zero mass is moved (horizontally or vertically) over a distance as a result of an acting force. ♦ Units: same as energy, usually J What is NOT work??? Kinetic/Potential Energy • Kinetic Energy: E (J) k ♦ Any moving object has a kinetic energy ♦ High velocity = large Ek ♦ Large mass = large Ek ♦ Fast winds closer to the ground correspond to bigger Ek. (Why?) • Potential Energy: E (J) p ♦ The potential to do work. Gravitational energy (height) Elastic energy (deformation) • • 1 2 E K mv 2 H E p mgH Energy can produce electricity • Gravitational potential energy: hydropowerplants • Kinetic energy: wind turbines, tidal harnesses • Radiant (solar) energy: solar panels • Chemical energy: batteries • Nuclear energy: nuclear powerplants • Heat: steam engines Conservation of Energy • Energy conservation law: ♦ The total energy of a closed system remains constant. ♦ Energy cannot be created nor can it be destroyed! ♦ Energy can only change from one form to another, e.g. kinetic to potential and vice versa. • Mechanical system: ♦ The loss of Ek in a given process equals the gain in Ep in the same process! ♦ The work is done by gravity or elastic forces. Radiant Energy • The energy emitted from the Sun is radiant energy. It is the most important source of energy for the Earth’s atmosphere. • Every body emits radiant energy (electromagnetic emission). ♦ Infrared, Visible, ultraviolet, X-rays, … Thermal Energy and Temperature • Definition: E is kinetic energy on a microscopic (molecular) level. • The temperature is proportional to the average molecular kinetic energy. • The temperature is a measure of the thermal energy. T ET T • The Absolute ZERO! ♦ No thermal energy. ♦ The atoms do not move. ♦ The absolute zero practically cannot be achieved. 1 2 E K mv 2 Temperature Scales • Kelvin (absolute scale) K • • ♦ No negative values ♦ Starts with THE O! ♦ Used in science ♦ Freezing point 273K ♦ Boiling point 373K Fahrenheit F ♦ Freezing point 32F ♦ Boiling point 212F ♦ 180 intervals Celsius (Centigrade) C ♦ Freezing point 0C ♦ Boiling point 100C ♦ 100 intervals Scale conversions • • • • • Fahrenheit -> Kelvin • Kelvin -> Celsius K - 273 =C Celsius -> Kelvin C + 273 =K Fahrenheit -> Celsius (F-32) x 5/9 = C Celsius Cx9/5+32 Kelvin -> Fahrenheit -> Fahrenheit ??? ??? =F Heat: Q • from one object to another. Heat is energy in the process of being transferred • • The amount of heat is equal to the change of energy that results from the process of energy transfer. Processes of heat transfer: ♦ Conduction; ♦ Convection; ♦ Radiation. Heat Capacity • Heat capacity: the amount of heat energy that is required to change the temperature of a body by 1 K. ♦ Heat capacity= Heat energy/Temperature change Q C T • > > ♦ It depends on the material and on the mass of the body Specific heat capacity: the amount of energy that is required to change the temperature of 1 gram of substance by 1 degree C. Q C m T = ♦ It does not depend on the mass of the body. ♦ It depends only on the material of the body. > Table 2-1, p. 30 Thermal inertia • Bodies with a large heat capacity cool and/or heat up very slowly. ♦ Analogy with a heavy body (a big truck) ♦ Water has a high heat capacity (large thermal inertia) 1cal/gram/degree ♦ Regions near large bodies of water (rivers, lakes, oceans) do not experience sharp temperature changes. Their climate is mild. ♦ Air and land have smaller specific heats than water. Figure 3.23 Phase Changes Phase transitions (1 gram of water) Sensible heat 600 cal 100C Temperature C Latent heat 100 cal 80 cal 0C ICE melting water HEAT IN boiling vapor Latent heat • Latent heat: the heat required to change a substance from one state to another (phase change) ♦ Evaporation/Melting (cools the environment) ♦ Condensation/Freezing (heats the environment) The importance of latent heat