* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download chpt7QuantTheory
Molecular Hamiltonian wikipedia , lookup
History of quantum field theory wikipedia , lookup
Hidden variable theory wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Particle in a box wikipedia , lookup
Molecular orbital wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Matter wave wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Tight binding wikipedia , lookup
Astronomical spectroscopy wikipedia , lookup
Hydrogen atom wikipedia , lookup
Atomic orbital wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Wave–particle duality wikipedia , lookup
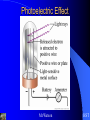
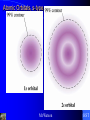
Chapter 7 Quantum Theory of the Atom Mr.Watson HST Electromagnetic Radiation Electromagnetic wave A wave of energy having a frequency within the electromagnetic spectrum and propagated as a periodic disturbance of the electromagnetic field when an electric charge oscillates or accelerates. Mr.Watson HST Electromagnetic Radiation Electromagnetic wave wavelength frequency amplitude Mr.Watson HST Electromagnetic Radiation nl= c where n => frequency l => wavelength c => speed of light Mr.Watson HST Wave Nature of the Electron Mr.Watson HST Electromagnetic Spectrum Mr.Watson HST Line Emission Spectrum Mr.Watson HST Line Spectrum A spectrum produced by a luminous gas or vapor and appearing as distinct lines characteristic of the various elements constituting the gas. Mr.Watson HST Emission Spectrum The spectrum of bright lines, bands, or continuous radiation characteristic of and determined by a specific emitting substance subjected to a specific kind of excitation. Mr.Watson HST Ground State The state of least possible energy in a physical system, as of elementary particles. Also called ground level. Mr.Watson HST Excited State Being at an energy level higher than the ground state. Mr.Watson HST Photoelectric Effect the emission of electrons by substances, especially metals, when light falls on their surfaces. Mr.Watson HST Photoelectric Effect Mr.Watson HST Quantum Mechanics Quantum theory the theory of the structure and behavior of atoms and molecules. Mr.Watson HST Black Body Radiation http://www.cbu.edu/~mcondren/C11599/BBvis.mov Mr.Watson HST Electromagnetic Radiation Ehi - Elo = hc/l where E => energy h => Planck's constant c => speed of light l => wavelength Mr.Watson HST Photons The quantum of electromagnetic energy, generally regarded as a discrete particle having zero mass, no electric charge, and an indefinitely long lifetime. Mr.Watson HST Dispersion of White Light Mr.Watson HST The Atomic Spectrum of Hydrogen and the Bohr Model Bohr Model for the Hydrogen Atom mnr = nh/2p Mr.Watson HST Bohr Atom Mr.Watson HST Bohr Model E = -B/n2 where n => quantum number 1, 2, 3, 4, 5, 6, 7, etc Mr.Watson HST Bohr Model for hydrogen ground state: n = 1 excited state: n > 1 Mr.Watson HST Bohr Model E = (-2.179 X 10-18 J/part.) (6.022 X 1023 part./mole) (1 kJ/103 J)/n2 = (-1312 kJ/mol)(1/n2) Mr.Watson HST Electron Transition in a Hydrogen Atom Lyman series => ultraviolet n > 1 ==> n = 1 Balmer series => visible light n > 2 ==> n = 2 Paschen series => infrared n > 3 ==> n = 3 Mr.Watson HST Line Spectra See CHEMWORKS software Mr.Watson HST Absorption Spectrum Light shinning on a sample causes electrons to be excited from the ground state to an excited state wavelengths of that energy are removed from transmitted spectra Mr.Watson HST Knowing diamond is transparent, which curve best represents the absorption spectrum of diamond (see below)? A, B, C Mr.Watson HST According to the energy diagram below for the Bohr model of the hydrogen atom, if an electron jumps from E1 to E2, energy is absorbed emitted not involved Mr.Watson HST Heisenberg, Werner 1901–76, German physicist 1932 Nobel Prize in physics A founder of QUANTUM MECHANICS, he is famous for his uncertainty principle, which states that it is impossible to determine both the position and momentum of a subatomic particle (such as the electron) with arbitrarily high accuracy. Mr.Watson HST Heissenberg Uncertainty Principle “it is impossible to determine both the position and momentum of a subatomic particle (such as the electron) with arbitrarily high accuracy” The effect of this principle is to convert the laws of physics into statements about relative, instead of absolute, certainties. Mr.Watson HST Orbitals region of probability of finding an electron around the nucleus 4 types => s p d f maximum of 2 electrons per orbital Mr.Watson HST Pure Atomic Orbitals s p d f shape # of orbitals / energy level spherical 1 dumbbell 3 complex 5 very complex 7 Mr.Watson HST Atomic Orbitals, s-type Mr.Watson HST Atomic Orbitals, ptype Mr.Watson HST Atomic Orbitals, d-type Mr.Watson HST Atomic Orbitals, f-type Mr.Watson HST Shapes of Orbitals http://www.colby.edu/chemistry/OChem/DEMOS/Orbitals.html Mr.Watson HST